Hairycellleukemia.org
CLINICAL TRIALS AND OBSERVATIONS
Eradication of minimal residual disease in hairy cell leukemia
Farhad Ravandi, Jeffrey L. Jorgensen, Susan M. O'Brien, Srdan Verstovsek, Charles A. Koller, Stefan Faderl, Francis J. Giles,Alessandra Ferrajoli, William G. Wierda, Shirley Odinga, Xuelin Huang, Deborah A. Thomas, Emil J. Freireich, Dan Jones,Michael J. Keating, and Hagop M. Kantarjian
Although the nucleoside analogs cladrib-
newly diagnosed HCL (n ⴝ
11) or after
CD8 lymphocyte number after rituximab.
ine and pentostatin produce high re-
failure of one prior chemotherapy (n ⴝ
2)
We conclude that eradication of MRD in
sponse rates in patients with hairy cell
with cladribine (5.6 mg/m2 given intrave-
HCL is possible. Whether this leads to a
leukemia (HCL), a significant number of
nously over 2 hours daily for 5 days)
reduced risk of relapse would need to be
patients eventually relapse. Several stud-
followed by 8 weekly doses of rituximab
evaluated in a larger number of patients
ies have demonstrated that patients with
(375 mg/m2). All patients achieved a CR
and with longer follow-up. Disease char-
complete remission (CR) have a longer
and minimal residual disease (MRD) as-
acteristics may potentially be used to
disease-free survival. Therefore, strate-
sessed by consensus primer polymerase
identify patients that are more likely to
gies to improve on the initial response to
chain reaction (PCR) or flow cytometry
benefit from such additional therapy.
nucleoside analog therapy are likely to be
was eradicated in 11 (92%) of 12 and in 12
beneficial, at least for a proportion of
(92%) of 13 of patients, respectively. There
patients. We have treated 13 patients with
was no decline in the absolute CD4 and
2006 by The American Society of Hematology
Hairy cell leukemia (HCL) is an indolent lymphoproliferative
achieved CR, with 7 (30%) achieving PR. The overall survival after
malignancy characterized by infiltration of the bone marrow, liver,
12 years was 87%.13 Other studies confirm a high but not universal
spleen, and occasionally lymph nodes with neoplastic B cells with
response rate as well as a relapse-free survival of 70% to 80% at
cytoplasmic hairlike projections.1 Before the availability of effec-
about 4 years.14,15
tive agents, splenectomy was the standard treatment option leading
Similar results have been achieved using pentostatin.16 Overall
to significant improvements in cytopenias but of limited duration.2
CR rates of 44% to 89% are seen with pentostatin given at a dose of
More recently, interferon-␣ (IFN-␣) and nucleoside analogs such
2 to 4 mg/m2 every 2 weeks.16 Dearden et al17 evaluated 165
as 2-chlorodeoxyadenosine (2-CDA or cladribine) and 2-deoxyco-
patients treated with pentostatin and 45 patients treated with
formycin (DCF or pentostatin) have been successfully used to treat
cladribine. Relapse rates were 24% for pentostatin and 29% for
patients with HCL.3-9
cladribine after a median follow-up of 71 and 45 months, respec-
Cladribine has been highly effective in treating patients with
tively.17 Flinn et al9 reported their long-term follow-up of 241
HCL, with overall response rates ranging from 75% to 100% after a
patients treated with pentostatin either as initial therapy or after
single course of the drug administered by continuous infusion daily
failure of IFN-␣. The 5- and 10-year event-free survival rates were
for 7 days.10 However, responses are not universal, and a significant
85% and 67%, respectively.9
proportion of patients relapse.10 Saven et al11 reported the long-
Therefore, there is a definite relapse rate associated with therapy
term follow-up of 358 patients treated at the Scripps Clinic who
of HCL with both cladribine and pentostatin, and the relapse-free
were followed for a median of 52 months. Twenty-six percent of
survival curve does not appear to reach a plateau. Recently, a
patients relapsed at a median of 29 months.11 Goodman et al12
number of reports have demonstrated the efficacy of the monoclo-
described the long-term follow-up of 209 patients treated with
nal antibody rituximab in treating patients with relapsed HCL.18-21
cladribine. Although the overall response rate was 100%, 76
Rituximab targets the pan–B-cell antigen CD20, which is ex-
patients (37%) relapsed, with a median time to relapse of 42
pressed at high levels on the surface of hairy cells.22,23 Nieva et al19
months.12 In a recent report from Northwestern University,13 86
reported a CR rate of 13% and a PR rate of 13% in 24 patients with
consecutive patients were treated with cladribine; a complete
relapsed HCL who received rituximab 375 mg/m2 once weekly for
remission (CR) rate of 79% and a partial remission (PR) rate of
4 weeks. Lauria et al21 treated 10 patients with relapsed or
21% were noted. The progression-free survival after 12 years was
progressed HCL with a similar regimen of rituximab and noted 1
54% and with a median follow-up of 9.7 years (range, 0.3-13.8
CR and 4 PR [overall response (OR), 50%]. In a study by Hagberg
years), 31 patients (36%) relapsed. Twenty-three relapsed patients
et al,20 11 patients with HCL (including 3 previously untreated
were treated with a second course of cladribine and 12 (52%)
patients) received rituximab 375 mg/m2 weekly for 4 weeks. The
From the Department of Leukemia, University of Texas, M.D. Anderson Cancer
Reprints: Farhad Ravandi, University of Texas, M.D. Anderson Cancer Center,
Center, Houston, TX; the Department of Hematopathology, University of Texas,
Department of Leukemia, Unit 428, 1515 Holcombe Blvd, Houston, TX 77030;
M.D. Anderson Cancer Center, Houston, TX; and the Department of
Biostatistics, University of Texas, M.D. Anderson Cancer Center, Houston, TX.
The publication costs of this article were defrayed in part by page chargepayment. Therefore, and solely to indicate this fact, this article is hereby
Submitted November 21, 2005; accepted February 14, 2006. Prepublished
marked ‘‘advertisement'' in accordance with 18 U.S.C. section 1734.
online as
Blood First Edition Paper, February 23, 2006; DOI 10.1182/blood-2005-11-4590.
2006 by The American Society of Hematology
BLOOD, 15 JUNE 2006 䡠 VOLUME 107, NUMBER 12
BLOOD, 15 JUNE 2006
ERADICATING MRD IN HCL
䡠 VOLUME 107, NUMBER 12
OR rate was 64% with 6 CRs and 1 PR (including 1 CR in a
treatment-naive patient).20 Thomas et al18 used an extended dosing
Between June 2004 and August 2005, 13 consecutive patients with HCL
regimen and administered rituximab 375 mg/m2 weekly for 8 to 12
were treated. The study was approved by the institutional review board of
doses to 15 patients with relapsed or refractory HCL. The overall
the University of Texas M.D. Anderson Cancer Center. All patients signed
response rate was 80%, including 8 CRs (52%), 2 CRs with
an informed consent to participate in the study. Eleven patients had newly
residual marrow disease (13%), and 2 PRs (13%).18
diagnosed disease and 2 had relapsed from prior therapy. One patient had
Rituximab has been successfully combined with nucleoside
received 2CDA 9 years ago and relapsed with progressive cytopenias;
analogs in the treatment of other indolent lymphoproliferative
another patient had received chlorambucil for 8 years and had generalized
disorders.24,25 These combinations have generally been well toler-
adenopathy, and a variant HCL histology. The median age of the patientswas 53 years (range, 31-73 years). Fluorescent in situ hybridization (FISH)
ated. We have treated 13 patients with HCL, either newly diag-
was positive for the presence of a clone with p53 deletion or monosomy 17
nosed or in relapse after one prior therapy, with cladribine followed
in 2 patients. One patient with variant HCL had a complex karyotype, but no
by rituximab to eradicate residual HCL. Minimal residual disease
cytogenetic abnormalities were noted in the other 12 patients. Six of 10
(MRD) was evaluated using flow cytometry for HCL-specific
evaluable patients had mutated immunoglobulin heavy chain variable gene
marker expression patterns and by an immunoglobulin heavy chain
(IgVH), whereas IgVH was unmutated in 4 patients. Patient characteristics
(IgH) polymerase chain reaction (PCR) assay using framework-1,
are summarized in Table 1.
-2, and -3 primer sets.
Cladribine 5.6 mg/m2 was administered intravenously over 2 hours daily for5 days. Approximately 28 days after initiation of cladribine, a repeat bone
Patients and methods
marrow examination was evaluated for the presence of MRD. This wasfollowed by 8 weekly doses of rituximab 375 mg/m2. At completion of
therapy with rituximab a repeat bone marrow examination for evaluation of
Patients were eligible if they had a new diagnosis of HCL with active
MRD was performed. Patients are then followed with peripheral blood
disease or if they had relapsed after one prior therapy. Diagnosis of HCL
evaluations every 3 months, including flow cytometry assays for MRD.
was based on morphologic evaluation of peripheral blood, bone marrowaspirates, and core biopsies, in combination with a characteristic flow
cytometric immunophenotype (bright positivity for CD22, CD11c, and
Prophylactic antibiotics, including levofloxacin, valacyclovir, and flucon-
CD103). Most cases were also stained for tartrate-resistant acid phospha-
azole (or equivalent agents), were administered at the discretion of the
tase (TRAP) on aspirate smears, and all tested cases were positive. Active
treating physician. Growth factors such as granulocyte-macrophage colony-
disease was defined as one or more of the following: (1) hemoglobin (Hgb)
stimulating factor (GM-CSF) and granulocyte colony-stimulating factor
level less than 100 g/L or transfusions of at least 2 units of packed red blood
(G-CSF) were administered at the discretion of the treating physician.
cells per month, absolute neutrophil count (ANC) less than 1.5 ⫻ 109/L,
Transfusion support with irradiated and filtered packed red blood cells,
platelet count less than 100 ⫻ 109/L, or greater than 25% decline from
platelets, or both was provided when indicated.
baseline over 3 months in one or more cell lines; (2) circulating hairy cellsat least 1 ⫻ 109/L or extramedullary HCL; and/or (3) bone marrow hairy
Criteria for response
cells at least 10% (on aspirates and/or biopsy sections); (4) recurrentinfections, progressive decline in performance status, or symptomatic
CR was defined as the absence of hairy cells on BM aspirate smears or the
splenomegaly. Patients were required to have an Eastern Cooperative
presence of less than 1% atypical cells (not definitively called hairy cells) in
Oncology Group (ECOG) performance status of 2 or less and adequate
bone marrow and blood and the disappearance of all evidence of HCL on
organ function with a serum bilirubin level of no more than 3.5 mg/dL,
physical examination. Achievement of CR required an ANC of at least
aspartate aminotransferase/alanine aminotransferase (AST/ALT) less than 3
1.5 ⫻ 109/L; Hgb at least 120 g/L (at least 110 g/L for women); and platelet
times the upper limit of normal, and creatinine no more than 2.0 mg/dL.
count at least 100 ⫻ 109/L without growth factor or transfusion support. CR
Higher values were acceptable if they were directly related to the disease.
with residual disease (CR-RD) was defined as for CR but with persistence
Table 1. Patient characteristics
Hgb level,
Age, y/sex
ⴛ
109/L
ⴛ
109/L
homology, %
17 by FISH
Rx indicates medication; WBC, white blood count; ANC, absolute neutrophil count; Hgb, hemoglobin; FISH, fluorescent in situ hybridization; 2CDA, 2-chlorodeoxyade-
nosine; NA, not available; Chl, chlorambucil.
*Variant HCL.
BLOOD, 15 JUNE 2006 䡠 VOLUME 107, NUMBER 12
of 1% to 5% hairy cells in the marrow (but no circulating hairy cells). PRwas defined as (1) meeting the peripheral blood count criteria forCR/CR-RD but with more than 5% residual hairy cells in the marrow or (2)at least 50% improvement or correction of at least one cytopenia withoutdecrease in any of the other cell counts, reduction in palpable abnormalitieson physical examination by at least 50%, and reduction in circulating orbone marrow hairy cells by at least 50%. Responses were evaluated aftercompletion of therapy with rituximab.
Figure 1. Assessment of hairy cells in a patient with HCL by flow cytometry
Bone marrow histology and immunohistochemistry (IHC)
performed on bone marrow aspirate specimen. CD19⫹ lymphocytes are shown.
(A) Day 0, hairy cells are 10.7% of total cells. (B) Day 30 (after 2CDA), hairy cells are
Formalin-fixed, paraffin-embedded bone marrow core biopsies were stained
11.0% of total cells. (C) Day 90 (after rituximab), hairy cells are undetectable, with
with hematoxylin and eosin for morphologic evaluation, at all posttherapy
fewer than 1/200 000 cells CD19⫹ with CD103 expression significantly abovebackground.
time points. For most time points, IHC staining was performed on biopsysections by standard techniques,26 for a pan–B-cell marker other than
framework 3 (FR3) regions, in combination with either a consensus JH or
CD20. Antibodies used were PAX-5 (1:35; BD Biosciences Transduction
CH primer with detection by capillary electrophoresis.27
Laboratories, San Jose, CA) or CD79a (1:50; Dako, Carpinteria, CA).
Cases showing B-cell aggregates by IHC were scored as positive for
IgVH gene analysis
residual disease.
Total RNA was extracted from bone marrow aspirate or peripheral bloodusing Trizol, converted to cDNA using Superscript II (Invitrogen, Carlsbad,
MRD and immune status assessment by flow cytometry
CA) and used to amplify the clonal immunoglobulin heavy chain gene
Peripheral blood and bone marrow specimens for MRD assays were stained
rearrangements by a PCR method using V primers derived from the leader
with a 4-color panel of antibodies with the following combinations
region with either a consensus JH or CH primer.28 The dominant IgVH
clone product(s) was then sequenced by standard Sanger methods. Diver-
CD45/CD19, CD20/CD25/CD45/CD19, Ig/CD22/CD45/CD19, Ig/CD22/
gence from germ line IgVH segments29 was calculated using DNAPLOT
CD45/CD19, and Ig/Ig/CD19/CD22. All antibodies were from BD
software (VBASE Sequence Directory, I.M.; Tomlinson, MRC Center for
Biosciences (San Diego, CA). Antibodies were added to 106 mononuclear
Protein Engineering, Cambridge, United Kingdom); 2% or less changes
cells per tube in 100 L whole blood or marrow (diluted with phosphate-
over codons 1 to 94 of IgVH was regarded as unmutated. The VH segment
buffered saline [PBS] and 1% fetal bovine serum as needed), and incubated
used was also recorded.
for 10 minutes at room temperature. Erythrocyte lysis was performed usingBD PharmLyse (BD Biosciences), followed by a wash with PBS containing
0.1% sodium azide, using a Sorvall Cell Washer 2. Cells were resuspended
A maximum of 44 patients are planned for this trial with early stopping
for acquisition in PBS containing 1% formaldehyde. For tubes containing
rules to monitor futility and excess toxicity. These stopping boundaries have
anti- or anti- antibodies, lysis and washing were performed prior to
not been reached. The CD4 and CD8 counts during the treatment were
cell-surface staining. Data were acquired on FACSCalibur flow cytometers
analyzed by paired Wilcoxon and paired
t tests.
(BD Biosciences) using BD CellQuest Pro software. CD19⫹ B cells wereselectively gated, with from 40 000 to 600 000 total cells acquired per tube,depending on sample quality. Data were analyzed using CellQuest Pro (BD
Hairy cells were identified on the basis of bright coexpression of CD11c
Response to therapy
and CD22, positivity for CD103 and CD25 (moderate to bright), and clonalexpression of surface immunoglobulin light chains on CD22 bright B cells.
All 13 patients (100%) have achieved CR after completion of the
Hairy cell variants were negative for CD25. The limit of sensitivity was
planned therapy. Approximately 1 month after 2CDA therapy,
established using control specimens consisting of 10 bone marrow aspi-
morphologic, IHC, or both examinations of bone marrow core
rates, acquired for staging of patients with other B-lineage lymphomas, all
biopsies showed that 8 patients had 0 or less than 1% hairy cells in
negative for malignancy, stained with the same antibody panel. There weresmall subsets of CD19⫹ cells in most control marrows which were also
their BM (CR), whereas 5 patients still had 1% to 50% hairy cells.
positive for CD11cbright/CD22bright. Thus, this marker combination was
Examination of the bone marrow core biopsies at the completion of
noncontributory for MRD studies. However, the background of CD103⫹
treatment with rituximab did not show definitive evidence of HCL
cells in control marrows was low (⬍ 0.02% of total cells), and CD25bright⫹
in any patient. MRD assessed by flow cytometry was positive in 11
cells showed similarly low levels. Therefore, in cases with background
patients 1 month after 2CDA therapy but became negative in 12 of
populations of normal B cells, the limit of detection of residual HCL was
13 patients after rituximab. The flow cytometry (FC) assay
0.02%. In specimens with no normal background B cells, HCL populations
appeared more sensitive than IHC for pan–B-cell markers; no
with multiple phenotypic aberrancies could be detected at a level of 0.05%
specimen was positive by IHC but negative by FC, whereas several
to 0.003% (20 aberrant cells of 40 000-600 000 collected). The MRD
specimens showed low-level positivity by FC but indeterminate
assessment for a representative patient is demonstrated in Figure 1.
IHC results, with only rare scattered B cells. MRD by PCR assay
For immune status assessment, peripheral blood lymphocytes were
stained with the following antibody combinations (FITC/PE/PerCP-Cy5.5/
was positive in 5 of 11 evaluable patients 1 month after 2CDA
APC): CD8/CD4/CD45/CD3 and CD3/CD56/CD45/CD19. All antibodies
therapy and became negative in 11 of 12 evaluable patients
were from BD Biosciences. Erythrocytes were lysed using FACSLyse (BD
(equivocal in 1) after rituximab. With a median follow-up of 14
Biosciences). Data were acquired and analyzed as described above.
months (range, 6-16 months), no patients have relapsed (medianresponse duration, 9 months; range, 4-16 months). One patient has
MRD assessment by polymerase chain reaction
developed a second malignancy (pancreatic carcinoma).
Total DNA was extracted from PB or bone marrow aspirate samples using
an automated method (Autopure; Genta, Minneapolis, MN). B-cell clonal-ity was determined using a polymerase chain reaction (PCR) method using
The treatment was well tolerated with no unexpected toxicity. Nine
V primers derived from the framework 1 (FR1), framework 2 (FR2), and
episodes of grade 3 toxicity, including 3 episodes of grade 3
BLOOD, 15 JUNE 2006
ERADICATING MRD IN HCL
䡠 VOLUME 107, NUMBER 12
neutropenic fever thought to be related to therapy and 2 episodes of
of IFN-␣ produced response rates of greater than 80%; however,
grade 3 herpes zoster possibly related to therapy, were reported.
most were PRs and of limited duration. The nucleoside analogs
Four other grade 3 events included one episode each of cellulitis,
cladribine and pentostatin have been highly effective in producing
acute gouty arthritis in a patient with history of gout, renal
lasting responses in most patients with HCL. Despite this, single-
impairment related to an acute attack of gout in a patient with a
agent therapy with cladribine or pentostatin is not universally
known history of gout, and renal impairment due to antibiotic-
effective. Furthermore, relapses do occur in up to 40% of patients
induced interstitial nephritis thought not to be related to the
within 4 years of initial therapy. Several studies have demonstrated
treatment. Other adverse events were grade 1 and 2 and included
that the quality of the initial response is predictive of outcome, with
nausea, rash, fatigue, weight gain, weight loss, and fever.
a longer disease-free survival for those achieving CR with theirinitial therapy.9,17 Therefore, to realize "cure" in HCL, achievement
of CR without evidence of MRD is desirable.
To evaluate the effect of an extended course of rituximab on
Wheaton et al30 have previously noted a shorter relapse-free
immune parameters, peripheral blood CD4 and CD8 lymphocyte
survival in patients who had evidence of MRD (detected by IHC
subsets were evaluated at various time points during the therapy.
using anti-CD45RO, anti-CD20, and DBA.44 in paraffin-embed-
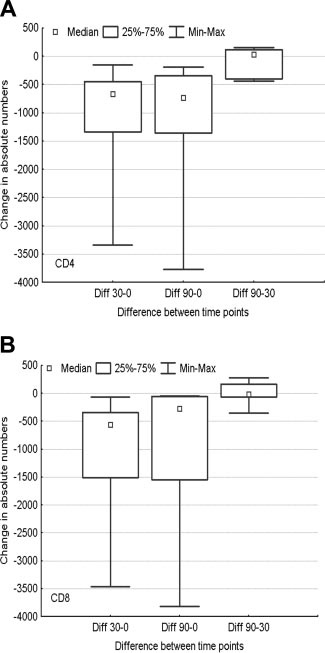
Treatment with cladribine led to a statistically significant decline in
ded bone marrow sections) compared with those who did not. More
the CD4 count, which remained stable after therapy with rituximab.
precise methods for detecting MRD using immunophenotyping by
Both CD4 and CD8 counts dropped significantly at day 30 (after
flow cytometry and consensus primer polymerase chain reaction
2CDA, P ⫽ .03 and P ⫽ .03, respectively) and day 90 (after
(cpPCR) analysis of immunoglobulin receptors are now avail-
rituximab, P ⫽ .03 and P ⫽ .03, respectively), compared with day
able.31 Whether eradication of MRD as detected by flow cytometry
0 (before starting therapy). However, neither CD4 (P ⫽ .84) nor
or cpPCR would lead to improvements in progression-free survival
CD8 (P ⬎ .999) counts dropped significantly from day 30 to day
(PFS) and overall survival (OS) is unclear. Furthermore, any agent
90 (by paired Wilcoxon tests) (Figure 2).
used for this purpose should have little additional associatedtoxicity. Rituximab has demonstrated efficacy in treating patientswith relapsed HCL and has been used successfully to treat patients
with purine analog–resistant disease.18-21 The combination ofrituximab with nucleoside analogs has been associated with an
Before the availability of the modern chemotherapeutic agents,
increased incidence of neutropenia.24,32 However, rituximab has
splenectomy was the standard therapy in patients with HCL and
relatively low toxicity and is not associated with significant
resulted in significant improvement in cytopenias. The introduction
immunosuppression. We have demonstrated the efficacy of anextended course of rituximab in eradicating MRD in most patientswith HCL treated in this study. Whether such MRD eradicationcould be achieved with nucleoside analog therapy alone is unclear,although previous studies have demonstrated that a significantnumber of patients in CR have evidence of MRD. Wheaton et al30reported that 5 (13%) of 39 patients in CR 3 months after therapywith cladribine had evidence of disease by IHC. We used signifi-cantly more sensitive techniques and found that only 1 (8%) of 13patients had disease detectable by flow cytometry and only 1 (8%)of 12 patients had an equivocal PCR at 3 months after therapy. Asexpected, treatment with rituximab was not associated with addi-tional toxicity, and there was no significant decline in the absolutenumbers of CD4 and CD8 lymphocyte subsets after extendedtherapy with rituximab.
Despite its limited toxicity, financial considerations in a disease
highly responsive to available therapy mandate the identification ofpatients at risk of relapse who would be candidates for additionaltherapy over nucleoside analogs alone. A different approach wouldbe to evaluate the marrow for MRD at 3 to 6 months after thecompletion of therapy with cladribine and administer monoclonalantibody only to those patients with evidence of MRD as has beendone by Cervetti et al.33 Alternatively, pretreatment features may beidentified that would predict the likelihood of relapse, therebyallowing selection of appropriate patients for additional therapy.
In most patients, the leukemic cells express mutated IgVH,
suggesting their origin from a postgerminal center (GC) memory Bcell.34,35 Nevertheless, we as well as other investigators haveidentified a number of patients with HCL with unmutated IgVHgenes, suggesting a pre-GC origin.36 However, gene expressionprofiling experiments have not demonstrated the existence ofdifferent subsets of HCL, suggesting that HCL has a uniform
Figure 2. Changes in lymphocyte subsets during therapy. Distribution of the (A)
phenotype.37 Furthermore, when compared with the gene expres-
CD4 count and (B) CD8 count changes among day 0 (before starting therapy), day 30(after 2CDA), and day 90 (after rituximab).
sion profiles of purified normal B-cell populations, including those
BLOOD, 15 JUNE 2006 䡠 VOLUME 107, NUMBER 12
of pre-GC (naive), GC, and post-GC (memory) B cells, the HCL
more extended regimen (8 weekly doses versus 4) appears to be
profile resembled that of memory B cells.37 The mutational status
associated with an improvement in the number and quality of
of IgVH did not influence the response to therapy in this cohort of
responses.18 This was the rationale for the selection of 8 doses of
patients. We have also identified 2 patients with deletion or
rituximab in our study. However, no definitive statements about the
mutation of p53, although they both responded to our regimen of
adequate number of doses of rituximab to achieve effective MRD
cladribine followed by rituximab. Whether the mutational status of
eradication can be made.
IgVH or p53 abnormalities can predict those patients who will
In conclusion, eradication of MRD in HCL using rituximab
relapse needs a larger cohort with longer follow-up.
after therapy with nucleoside analogs is safe and feasible. Longer
Another important consideration is the number of doses of
follow-up and larger patient numbers are needed to determine
rituximab that may be necessary for achieving disease eradication.
whether such eradication of MRD would translate to longer
Previously reported studies of rituximab in HCL have been in the
disease-free survival. Identification of predictors of relapse may
setting of relapse.18-21 Although the numbers of patients in each
allow the selection of patients who are most likely to benefit from
study are small and a definitive comparison cannot be justified, a
this extended regimen.
1. Kraut EH. Clinical manifestations and infectious
western University experience. Blood. 1996;88:
18) and 8q24/c-MYC translocation generally have
complications of hairy-cell leukaemia. Best Pract
a poor prognosis. Mod Pathol. 2006;19:25-33.
Res Clin Haematol. 2003;16:33-40.
15. Hoffman MA, Janson D, Rose E, Rai KR. Treat-
27. Theriault C, Galoin S, Valmary S, et al. PCR anal-
2. Jansen J, Hermans J. Splenectomy in hairy cell
ment of hairy-cell leukemia with cladribine: re-
ysis of immunoglobulin heavy chain (IgH) and
leukemia: a retrospective multicenter analysis.
sponse, toxicity, and long-term follow-up. J Clin
TcR-gamma chain gene rearrangements in the
diagnosis of lymphoproliferative disorders: results
3. Quesada JR, Reuben J, Manning JT, Hersh EM,
16. Grever MR, Doan CA, Kraut EH. Pentostatin in
of a study of 525 cases. Mod Pathol. 2000;13:
Gutterman JU. Alpha interferon for induction of
the treatment of hairy-cell leukemia. Best Pract
remission in hairy-cell leukemia. N Engl J Med.
Res Clin Haematol. 2003;16:91-99.
28. Campbell MJ, Zelenetz AD, Levy S, Levy R. Use
17. Dearden CE, Matutes E, Hilditch BL, Swansbury
of family specific leader region primers for PCR
4. Ratain MJ, Golomb HM, Vardiman JW, Vokes EE,
GJ, Catovsky D. Long-term follow-up of patients
amplification of the human heavy chain variable
Jacobs RH, Daly K. Treatment of hairy cell leuke-
with hairy cell leukaemia after treatment with pen-
region gene repertoire. Mol Immunol. 1992;29:
mia with recombinant alpha 2 interferon. Blood.
tostatin or cladribine. Br J Haematol. 1999;106:
29. European Bioinformatics Institute. ImMuno
GeneTics (IMGT) Database. http://www.ebi.
5. Piro LD, Carrera CJ, Carson DA, Beutler E. Last-
18. Thomas DA, O'Brien S, Bueso-Ramos C, et al.
ac.uk/imgt. Accessed October 2005.
ing remissions in hairy-cell leukemia induced by a
Rituximab in relapsed or refractory hairy cell leu-
single infusion of 2-chlorodeoxyadenosine.
kemia. Blood. 2003;102:3906-3911.
30. Wheaton S, Tallman MS, Hakimian D, Peterson
N Engl J Med. 1990;322:1117-1121.
19. Nieva J, Bethel K, Saven A. Phase 2 study of rit-
L. Minimal residual disease may predict bonemarrow relapse in patients with hairy cell leuke-
6. Estey EH, Kurzrock R, Kantarjian HM, et al.
uximab in the treatment of cladribine-failed pa-
mia treated with 2-chlorodeoxyadenosine. Blood.
Treatment of hairy cell leukemia with 2-chlorode-
tients with hairy cell leukemia. Blood. 2003;102:
oxyadenosine (2-CdA). Blood. 1992;79:882-887.
31. Sausville JE, Salloum RG, Sorbara L, et al. Mini-
7. Kraut EH, Bouroncle BA, Grever MR. Pentostatin
20. Hagberg H, Lundholm L. Rituximab, a chimaeric
mal residual disease detection in hairy cell leuke-
in the treatment of advanced hairy cell leukemia.
anti-CD20 monoclonal antibody, in the treatment
mia. Comparison of flow cytometric immunophe-
J Clin Oncol. 1989;7:168-172.
of hairy cell leukaemia. Br J Haematol. 2001;115:609-611.
notyping with clonal analysis using consensus
8. Grever M, Kopecky K, Foucar MK, et al. Random-
primer polymerase chain reaction for the heavy
21. Lauria F, Lenoci M, Annino L, et al. Efficacy of
ized comparison of pentostatin versus interferon
chain gene. Am J Clin Pathol. 2003;119:213-217.
anti-CD20 monoclonal antibodies (Mabthera) in
alfa-2a in previously untreated patients with hairy
patients with progressed hairy cell leukemia.
32. Voog E, Morschhauser F, Solal-Celigny P. Neu-
cell leukemia: an intergroup study. J Clin Oncol.
tropenia in patients treated with rituximab. N Engl
22. Juliusson G, Lenkei R, Liliemark J. Flow cytom-
J Med. 2003;348:2691-2694.
9. Flinn IW, Kopecky KJ, Foucar MK, et al. Long-
etry of blood and bone marrow cells from patients
33. Cervetti G, Galimberti S, Andreazzoli F, et al. Rit-
term follow-up of remission duration, mortality,
with hairy cell leukemia: phenotype of hairy cells
uximab as treatment for minimal residual disease
and second malignancies in hairy cell leukemia
and lymphocyte subsets after treatment with
in hairy cell leukaemia. Eur J Haematol. 2004;73:
patients treated with pentostatin. Blood. 2000;96:
2-chlorodeoxyadenosine. Blood. 1994;83:3672-
34. Forconi F, Sahota SS, Raspadori D, et al. Hairy
10. Goodman GR, Beutler E, Saven A. Cladribine in
23. Ginaldi L, De Martinis M, Matutes E, Farahat N,
cell leukemia: at the crossroad of somatic muta-
the treatment of hairy-cell leukaemia. Best Pract
Morilla R, Catovsky D. Levels of expression of
tion and isotype switch. Blood. 2004;104:3312-
Res Clin Haematol. 2003;16:101-116.
CD19 and CD20 in chronic B cell leukaemias.
11. Saven A, Burian C, Koziol JA, Piro LD. Long-term
J Clin Pathol. 1998;51:364-369.
35. Vanhentenrijk V, Tierens A, Wlodarska I, Verhoef
follow-up of patients with hairy cell leukemia after
24. Byrd JC, Peterson BL, Morrison VA, et al. Ran-
G, Wolf-Peeters CD. V(H) gene analysis of hairy
cladribine treatment. Blood. 1998;92:1918-1926.
domized phase 2 study of fludarabine with con-
cell leukemia reveals a homogeneous mutation
12. Goodman GR, Burian C, Koziol JA, Saven A. Ex-
current versus sequential treatment with ritux-
status and suggests its marginal zone B-cell ori-
tended follow-up of patients with hairy cell leuke-
imab in symptomatic, untreated patients with
gin. Leukemia. 2004;18:1729-1732.
mia after treatment with cladribine. J Clin Oncol.
B-cell chronic lymphocytic leukemia: results from
36. Thorselius M, Walsh SH, Thunberg U, Hagberg
Cancer and Leukemia Group B 9712 (CALGB
H, Sundstrom C, Rosenquist R. Heterogeneous
13. Chadha P, Rademaker AW, Mendiratta P, et al.
9712). Blood. 2003;101:6-14.
somatic hypermutation status confounds the cell
Treatment of hairy cell leukemia with 2-chlorode-
25. Schulz H, Klein SK, Rehwald U, et al. Phase 2
of origin in hairy cell leukemia. Leuk Res. 2005;
oxyadenosine (2-CdA): long-term follow-up of the
study of a combined immunochemotherapy using
Northwestern University experience. Blood. 2005;
rituximab and fludarabine in patients with chronic
37. Basso K, Liso A, Tiacci E, et al. Gene expression
lymphocytic leukemia. Blood. 2002;100:3115-
profiling of hairy cell leukemia reveals a pheno-
14. Tallman MS, Hakimian D, Rademaker AW, et al.
type related to memory B cells with altered ex-
Relapse of hairy cell leukemia after 2-chlorode-
26. Kanungo A, Medeiros LJ, Abruzzo LV, Lin P. Lym-
pression of chemokine and adhesion receptors. J
oxyadenosine: long-term follow-up of the North-
phoid neoplasms associated with concurrent t(14;
Exp Med. 2004;199:59-68.
Source: https://www.hairycellleukemia.org/wp-content/uploads/2013/12/Ravandi-Eradication-of-minimal-residual-disease.pdf
bei Erwachsenen Anleitung zur medikamentösen Tumorschmerztherapie (Überarbeitet von Dr. Beekhchand Permar) Pflegeinterventionen in der Schmerztherapie Herausgeber: Arbeitsgruppe Schmerztherapie im Onkologischen Zentrum Westpfalz für die Arbeitsgruppe: Prof. Dr. Ch. Madler, Anästhesiologie Prof. Dr. H. Link, Medizinische Klinik
Items not to be prescribed at T&G CCG expense (DNP list) or only in limited circumstances (Grey list) Please note that in addition to the items listed here there should be no prescribing of any medicine listed in Part XVIIIA (Black List) of the Drug Tariff or in the case of appliances any not listed in Part IX. of the Drug Tariff. There are a number of medications that T&G CCG believes should not be used (DNP List) or should only be used in limited circumstances (Grey List). Grey listed items wil only be funded for patients who meet the specified criteria. The reasons for medicines being included on the lists are as given in the tables below and in line with the GMMMG and national guidance.








