Knowledgestream.ru

Hippocampal microRNA-132 mediates
stress-inducible cognitive deficits through
its acetylcholinesterase target
G. Shaltiel, M. Hanan, Y. Wolf,
S. Barbash, E. Kovalev, S. Shoham &
Brain Structure and Function
ISSN 1863-2653
Brain Struct Funct
DOI 10.1007/s00429-011-0376-z
Your article is published under the Creative
Commons Attribution Non-Commercial
license which allows users to read, copy,
distribute and make derivative works for
noncommercial purposes from the material,
as long as the author of the original work is
cited. All commercial rights are exclusively
held by Springer Science + Business Media.
You may self-archive this article on your own
website, an institutional repository or funder's
repository and make it publicly available
Brain Struct Funct
Hippocampal microRNA-132 mediates stress-inducible cognitivedeficits through its acetylcholinesterase target
G. Shaltiel • M. Hanan • Y. Wolf • S. Barbash •E. Kovalev • S. Shoham • H. Soreq
Received: 7 November 2011 / Accepted: 29 December 2011Ó The Author(s) 2012. This article is published with open access at Springerlink.com
Diverse stress stimuli induce long-lasting cog-
of the soluble ‘‘readthrough'' AChE-R variant without the
nitive deficits, but the underlying molecular mechanisms
30-untranslated region binding site for miR-132. TgR mice
are still incompletely understood. Here, we report three
different stress models demonstrating that stress-inducible
enhanced c-fos labeling and correspondingly intensified
increases in microRNA-132 (miR-132) and consequent
reaction to the cholinergic agonist pilocarpine. They fur-
decreases in its acetylcholinesterase (AChE) target are
ther showed excessive hippocampal expression of miR-
causally involved. In a mild model of predator scent-
132, accompanied by reduced host AChE-S mRNA and the
induced anxiety, we demonstrate long-lasting hippocampal
GTPase activator p250GAP target of miR-132. At the
elevation of miR-132, accompanied by and associated
behavioral level, TgR mice showed abnormal nocturnal
with reduced AChE activity. Using lentiviral-mediated
locomotion patterns and serial maze mal-performance in
suppression of ‘‘synaptic'' AChE-S mRNA, we quantified
spite of their reduced AChE-S levels. Our findings attribute
footshock stress-inducible changes in miR-132 and AChE
stress-inducible cognitive impairments to cholinergic-
and its corresponding cognitive damages. Stressed mice
mediated induction of miR-132 and consequently sup-
showed long-lasting impairments in the Morris water maze.
pressed ACHE-S, opening venues for intercepting these
In contrast, pre-stress injected AChE-suppressing lentivi-
rus, but not a control virus, reduced hippocampal levels ofboth miR-132 and AChE and maintained similar cognitive
Acetylcholinesterase � P250GAP �
performance to that of naı¨ve, non-stressed mice. To dis-
Cholinergic � Cognition � MicroRNA-132 �
sociate between miR-132 and synaptic AChE-S as poten-
Psychological stress
tial causes for stress-inducible cognitive deficits, we furtherused engineered TgR mice with enforced over-expression
Shaltiel G and Hanan M contributed equally.
Acute and chronic stress can both induce anxiety (McEwenand Gianaros ) and either impair learning and memory
Electronic supplementary material
The online version of this
performance (Diamond et al. Nijholt et al. or
article contains supplementarymaterial, which is available to authorized users.
enhance it (Blank et al. Manipulating cholinergicneurotransmission in the hippocampus, which receives
G. Shaltiel � M. Hanan � Y. Wolf � S. Barbash � H. Soreq (&)
extensive cholinergic innervations, changes the cognitive
The Edmond and Lily Safra Center of Brain Sciences,
control over executive function and error monitoring
The Silberman Institute of Life Sciences,The Hebrew University of Jerusalem,
(Carter et al. suggesting cholinergic involvement in
Jerusalem 91904, Israel
stress responses. Recent studies associate acute and chronic
stress reactions with specific microRNA (miR)-mediatedsilencing of affected transcripts (Meerson et al.
E. Kovalev � S. ShohamHerzog Memorial Hospital, Jerusalem 91351, Israel
MiRs are short (*22 nucleotides), non-coding RNAs that
Brain Struct Funct
regulate various molecular pathways (Bartel Krol
malfunctioning, then maintaining miR-132 excess even
et al. by post-transcriptional gene silencing (Fili-
under AChE-S suppression would cause cognitive deficits.
powicz et al. ; Rana ). Each miR may target
To investigate this triple hypothesis, we employed three
several mRNAs, often in specific locations on their
diverse stress paradigms: the acute predator scent test,
30-untranslated region (30-UTR) and can modulate entire
acute unpredictable footshocks accompanied by hippo-
pathways in a rheostat-like manner (Chen et al.
campal AChE knockdown, and engineered mice with an
[reviewed in (Soreq and Wolf However, the sig-
inherited anxiogenic-like phenotype (Salas et al. due
naling pathway(s) controlling the levels of neuronal miRs,
to enforced excess of AChE-R, but with a depleted miR-
the corresponding miR-target interactions involved in their
132 binding site which ensures continuous suppression of
diverse functions and the consequent cognitive effects
the host AChE-S, but not the transgenic AChE-R proteins
remain incompletely understood.
(Shaked et al. ). In the two latter models, we also
MiR-132 is prominently neuronal-enriched, highly
evaluated cognitive deficits evolving from the stress
induced by the cAMP-response element binding protein
(CREB). In cortical neurons, miR-132 enhances neuronalmorphogenesis and neurite outgrowth by decreasing thelevels of the Rho family member GTPase-activating pro-
tein, p250GAP (Vo et al. ). Mice engineered to over-express miR-132 in forebrain neurons correspondingly
show marked increase in dendrite spine density, accom-panied by deficits in a novel object recognition test
Mice were kept in an animal room at a constant tempera-
(Wayman et al. However, which neurotransmission
ture (22 ± 1°C) and a 12-h light/dark cycle with free
signal(s) are responsible for miR-132 changes and which of
access to food and water. They were tested in the same
its target(s) mediate these effects remained unanswered.
room under dim illumination (30 lux) by the same testers
Another target of miR-132 is the acetylcholine hydro-
between 10 and 16 h. All experimental procedures were
lyzing enzyme acetylcholinesterase (AChE) (Shaked et al.
approved by The Hebrew University's Committee for
; Meshorer and Soreq ). In acute psychological
Animal Experimentation (NS-08-11485-4).
stress, activated cholinergic neurons display rapid, yettransient increases in a normally rare soluble AChE-R
splice variant (Kaufer et al. that replaces the ‘‘syn-aptic'' AChE variant (Kaufer et al. Meshorer et al.
Intraperitoneal (i.p) injection of 0.1 ml of 25 mg/kg pilo-
; Meshorer and Soreq ). In macrophages, AChE
carpine hydrochloride (Sigma, Rehovot, Israel), or saline
suppression by miR-132 accentuates cholinergic signaling
followed 30 min habituation to a holding cage.
(Shaked et al. In transfected neurons, tetracycline-controlled antisense suppression of AChE leads to
Stereotactic surgeries
enhanced dendrite extension and causes hyper-locomotionin engineered mice (Sklan et al. ). Taken together, we
Group housed C57Bl/6J 9 weeks old male mice underwent
surmised that miR-132-mediated suppression of hippo-
stereotactic surgery, after which they were singly-housed
campal AChE levels may enhance cholinergic signaling,
throughout all subsequent testing. Control mice were singly
contributing to the neurite extension, cognitive and loco-
housed as well. Mice were anesthetized by i.p. injections of
motion impairments caused by stressful experiences.
ketamine (50 mg/kg) (Forth Dodge, IA, USA) and domitor
To challenge these predictions, we sought appropriate
(0.5 mg/kg) (Orion Pharma, Espoo, Finland) mix, and then
mouse models and experimental strategies. Our rationale
mounted in a stereotactic apparatus for intra-hippocampal
was as follows: (1) If indeed stress-inducible changes in
injections. Coordinates of the injection sites (in mm) rel-
hippocampal miR-132 and AChE cause long-lasting effects,
ative to bregma were AP: -2.0, L: 1.8, DV: -1.5. Bilateral
miR-132 increases and AChE activity decreases should be
injections of 0.5 ul lentiviral suspensions were conducted
observed in different stress models and even several days
using a 10-l Glenco syringe (Houston, TX, USA). After
after the initiation of psychological stress. (2) If the changes
each injection, the needle was left in situ for 5 min before
in miR-132 and ‘‘synaptic'' AChE-S are inter-related, then
being retracted to allow complete diffusion.
preventing one would avoid the other, and if these changesare the cause for stress-induced cognitive deficits, then
preventing them both from happening would avoid thesedeficits. (3) Alternatively, if miR-132 elevation by itself is
The water maze consisted of a round tank, 1.6 m in
sufficient to cause the stress phenotype and cognitive
diameter, filled with water. Mice were trained to find the
Brain Struct Funct
location of a hidden platform (16 cm in diameter), sub-
paraformaldehyde containing 4% sucrose (pH 7.4) or
merged 1 cm below the water surface, using extra maze
removed and kept frozen in -70°C.
visual cues. The training part consisted of 4 trials per day,with a 1-h brake between trials, for 3 days. The escape
Predator scent exposure and footshock acute stress
latency, i.e., the time required by the mouse to find theplatform and climb on it, was recorded for up to 60 s. Each
Group housed C57Bl/6J 9 weeks old male mice were
mouse was allowed to remain on the platform for 30 s and
placed on well-soiled cat litter for 10 min (in use by the cat
was then moved from the maze to its home cage. If the
for 2 days, sifted for stools) (Cohen et al. or were
mouse did not find the platform within 60 s, it was placed
placed in a footshock delivery system (Campden Instru-
gently on the platform for 30 s, and then returned to its
ments, UK) where they received seven inescapable electric
home cage. On the fourth day of the experiment, the
footshocks (0.3 mA; 2 ms) at unequal intervals over a total
platform was removed and a probe trial was conducted:
period of 120 min.
mice were placed in the maze for 60 s, in which thenumber of crosses over the four quadrants of the maze was
Elevated plus maze
recorded. Increased swimming in the quadrant where theplatform was originally placed was considered as an indi-
Anxiety-related behaviors were tested in a Plexiglas plus-
cation of spatial acquisition. One day after the end of
shaped maze containing two dark and enclosed arms
behavioral tests mice were anesthetized by Isoflurane
(30 9 5 cm with a 5 9 5 cm center area and 40 cm high
inhalation and decapitated. Brains were removed and two
walls) and two 30 9 5 cm open and lit arms, all elevated
half sagital sections were immediately frozen in liquid
50 cm above ground. Individual mice were placed in the
nitrogen. One section was sliced in cryostat for Karnov-
center of the maze, tracked for 5 min with a video
sky–Roots staining experiments (Sternfeld et al. and
camera, and then returned to their home cage. The plus
the other was taken for mRNA measurements. In brief,
maze was wiped clean between trials with a 10% alcohol
AChE activity staining was conducted using the Karnov-
sky–Roots staining method (Karnovsky and Roots ;Kaufer et al. 40-um frozen sections were incubatedat room temperature in soft shaking for 5 min. Staining
Lentiviral procedures
solution contained 2 mM acetylthiocholine iodide (Sigma,Israel), dissolved in PBS, 5 mM sodium citrate, 3 mM
The packaged virus was collected at 24 and 48 h post-
cupric sulfate, 0.5 mM potassium ferricyanide, 0.1 mM
transfection and concentrated using ultracentrifugation
butyrylcholinesterase specific inhibitor, ISO-OMPA.
(70,000g, 2 h, 15°C). Dilutions of concentrated virus werefollowed by infection of HEK-293T cells with diluted
Two unit serial maze
virus. The resulting titer (*1 9 109 infectious particlesper ml) was assessed for shRNA expressing viruses using
3- to 6-months-old female 30 UTR-null AChE and FVB/N
puromycin selection and for GFP expressing viruses by cell
mice were group housed. Only females were taken for this
fluorescence counting. Screening of two putative AChE
experiment due to their higher stress responsiveness com-
shRNAs, sh799 and sh800, designed to down-regulate
pared with males (Goel and Bale ). Modular units
mouse/rat AChE levels was first conducted in neuronal-like
placed in a series constitute this maze (Quartermain et al.
PC12 cells and compared to a control shRNA (shCON) that
). To complete one ‘‘run'', a 22 h water-deprived
does not target any mammalian gene. Both shRNAs
mouse must choose between turning right or left to receive,
reduced the AChE-S variant and suppressed AChE total
at the end of the maze, a reward in the form of 40 ll of 5%
activity in these cells, but only Sh799 down-regulated
sucrose and one ‘‘run'' is considered complete. Then the
overall AChE mRNA levels (Online Resources Fig. 1).
mouse must shuttle back to the other end of the maze whereit gets the same reward. There were 5 runs per session; one
Viral infection of primary cortical cell culture
session per day. Quantitative measures of performanceinclude number of left/right choice errors and ‘‘retrace
Cerebral cortex was separated from the brain of 15th
errors''—episodes in which a mouse moves in the wrong
day embryos of FVB/N mice, minced, and cells plated
direction (toward the end without a reward) (Farchi et al.
on poly-L-ornithine coated 12-well plates in Neurobasal
). Mice were euthanized 80–90 min after the behav-
medium, 2% B27 supplement, 20% Glutamax and 20%
ioral experiment by an intra-peritoneal injection of
Penicillin/streptomycin (Invitrogen, Grand Island, NY,
200 mg/kg sodium pentobarbital (‘‘Pental''). Brains were
USA). 72 h post-plating, 250 ul of non-concentrated
either fixed by transcardial perfusion with ice-cold 4%
medium containing viral particles was added to each well.
Brain Struct Funct
to bregma. Images of each hippocampal sub-region wereacquired and analyzed using AnalySIS software (SIS,
RNA was extracted (RNeasy lipid tissue kit, Qiagen,
Germany). Karnovsky staining of histochemical AChE
Valencia, CA, USA), DNase treated and its integrity con-
activity was as in (Sternfeld et al. In situ hybrid-
firmed by gel electrophoresis.
ization was essential as described (Berson et al. ).
For SYBR green real-time PCR, cDNA synthesis (Pro-
mega, Madison, WI, USA) involved 1 lg RNA samples in
AChE activity measurement
20 ll reactions. Duplicate real-time reverse transcriptase(RT) PCR tests involved ABI prism 7900HT, SYBR green
AChE activity was measured by Ellman's assay (Ellman
master mix (Applied biosystems, Foster City, CA, USA)
et al. adjusted to multiwell plates and normalized
and ROX, and a passive reference dye for signal normali-
according to the protein content of the cell lysate (DC
zation across the plate. Primer sequences are listed in
protein assay, Bio-Rad, Hercules, CA, USA).
Table . 18S rRNA or GAPDH was used as referencetranscripts. Annealing temperature was 60°C for all prim-
Statistical analysis
ers. Serial dilution of samples served to evaluate primersefficiency and the appropriate cDNA concentration that
The following statistical analyses were done using
yields linear changes. Melting curve analysis and ampli-
STATISTICA9 and GraphPad Prism 5 software: Student's
cons sequencing verified the identity of end products.
t test, one-way ANOVA and two-way ANOVA with Tukey
MicroRNA quantification using The TaqMan MicroRNA
post hoc comparisons and Pearson's correlation test.
Assays (Applied Biosystems, Foster City, CA, USA) wasconducted in two-step RT-PCR kit according to the man-ufacturer's instructions. For miR-132 (Applied Biosystems,
Foster City, CA, USA) measurements, a reference tran-script of SNO-135 (Applied Biosystems) was used.
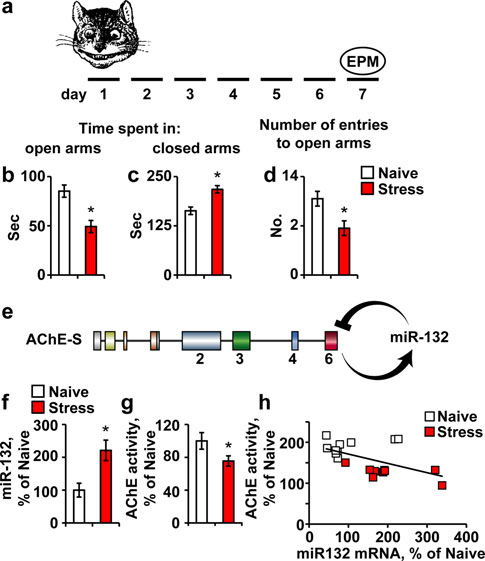
Elevated predator stress-inducible miR-132 associateswith suppression of AChE-S
Histochemistry and immunohistochemistry
Our first experimental model served to explore the inter-
The AChE-R C-terminal peptide (ARP) was labeled by a
relationship between a long-lasting stress phenotype,
rabbit polyclonal antibody (Berson et al. The com-
induced by predator scent, increases in hippocampal miR-
mon domain shared by both AChE variants was labeled
132 and decreases in AChE-S. Seven days following a
with goat anti-human AChE (Santa Cruz, CA, USA, anti-
predator scent stress (Fig. C57BI/6J mice subjected to
body N19). c-fos was stained using rabbit anti c-fos
an elevated plus maze (EPM) showed sustained anxiety.
(Sigma, Rehovot, Israel). In co-localization studies, we
Compatible with previous reports on predator smell-stres-
used fluorescein (FITC)-labeled donkey anti-rabbit to
sed mice (Cohen et al. they spent less time in the
visualize AChE-R and streptavidin-Cy3 to visualize gen-
maze open arms and more time in its closed arms and
eral AChE. Two to three coronal brain sections were
attempted fewer entries to the open arms compared to naı¨ve
sampled at an estimated distance of 2.8–3.3 mm posterior
mice (Fig. b–d). Given that miR-132 targets the AChE
Table 1 Primer sequences for
Forward primer 50–30
Reverse primer 50–30
SYBR green real-time PCR
Brain Struct Funct
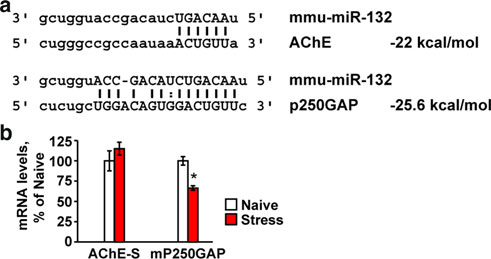
Fig. 2 Neuronal AChE and the GTPase activator p250GAP are bothtargets of miR-132. a The ‘seed' region in miR-132 is complementaryto the targeted 30-UTR in the AChE-S and P250GAP transcripts[created using Targetscan and Pictar websites (Online ResourcesTable 1)]. b Hippocampal mP250GAP mRNA, but not mAChE-Slevels are reduced in stressed compared to naı¨ve mice (Student'st tests: *p 0.05), likely reflecting different balances between therates of transcription and miR-132-mediated destruction of these twotranscripts
relationships with its neuronal targets, we quantified themajor AChE-S mRNA and the p250GAP transcript, withpredictably tighter hybridization and higher energy inter-
Fig. 1 Predator scent stress leads to inter-related miR-132 elevation
action with miR-132 (Fig. Intriguingly, p250GAP but
and AChE and p250GAP suppression. a Mice were exposed to the
not AChE mRNA levels were suppressed in the predator
predator scent test and 7 days later were tested in the elevated plusmaze (EPM). b–d Compared to naı¨ve, stressed mice spent less time in
scent-stressed hippocampus (Student's t test: p 0.05,
the open EPM arms (b; Student's t test: *p 0.05), more time in the
Fig. b), suggesting less robust stress-inducible transcrip-
closed EPM arms (c; *p 0.05) and attempted less entries to the
tion and/or more efficient destruction of p250GAP mRNA
open arms (d; *p 0.05). e AChE-S activation leads to miR-132
under stress.
up-regulation, which in return down-regulates the AChE-S transcript.
f–h 7 days post predator scent exposure, hippocampal miR-132 isup-regulated by *220% (f; Student's t test, t18 = -3.22, *p 0.01),
AChE-S knockdown prevents miR-132 up-regulation
AChE activity is reduced (g; Student's t test, t23 = 2.00, *p 0.05)
and memory impairments
and hippocampal miR-132 levels show significant negative correla-tion to AChE activity (h; Pearson's test: r = -0.55; p 0.01)
Predicting that the stress-inducible changes in hippocampalmiR-132/AChE-S impair the cholinoceptive capacities of
mRNA transcript (Fig. we quantified hippocampal
hippocampal neurons (Gray et al. we next set out to
miR-132 levels, which we found to be higher by *220%
establish an in vivo experimental model where these
compared to naı¨ve mice (Student's t test: p 0.01;
changes could be avoided. To achieve this goal, we intro-
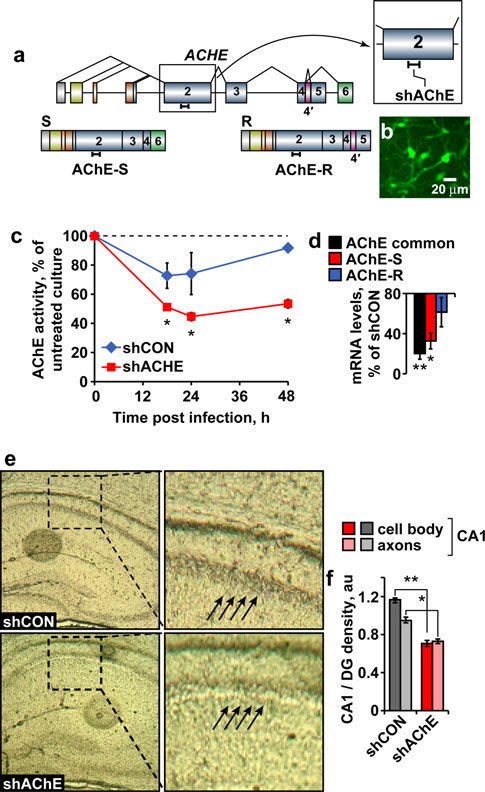
Fig. accompanied by 25% reduction in AChE activity
duced AChE knockdown. For this purpose, we used a pre-
(t test: p 0.05; Fig. A significant negative correla-
calibrated shACHE agent targeted to the exon2 common to
tion between hippocampal miR-132 levels and AChE
the AChE-S and AChE-R transcripts (Fig. and which
activity suggested causal relationship (Pearson's test:
efficiently infected cultured primary neurons (Fig. b),
r = -0.55; p 0.01; Fig. h), compatible with the
reduced AChE activity by over 50% within 12–24 h post-
capacity of miR-132 to suppress AChE activity in cultured
infection (Fig. c) and suppressed AChE-S and AChE-R
primary neurons (Online Resources Fig. 2).
mRNA levels as well as the common domain shared by
Our previous findings showed stress-inducible increases
these two transcripts (Fig. Bilateral injection of this
in neuronal AChE gene expression (Meshorer et al. ;
agent to the hippocampus CA1 region of this AChE-tar-
Meshorer and Soreq ). Given that mRNA levels reflect
geted lentiviral shRNA (shAChE) was then followed by a
a steady state between its synthesis and degradation, we
10-days healing period. At this time point, long after the
predicted that the observed decline in AChE activity
transient stress-inducible increases in hippocampal AChE
could potentially mask a balance between increases in the
have ceased, histochemical staining showed reduced AChE
‘‘synaptic'' AChE-S mRNA and miR-132-induced decrea-
activity in the hippocampus of lentiviral injected mice,
ses in its levels. To find more information on miR-132
with predictably larger reduction of AChE activity in
Brain Struct Funct
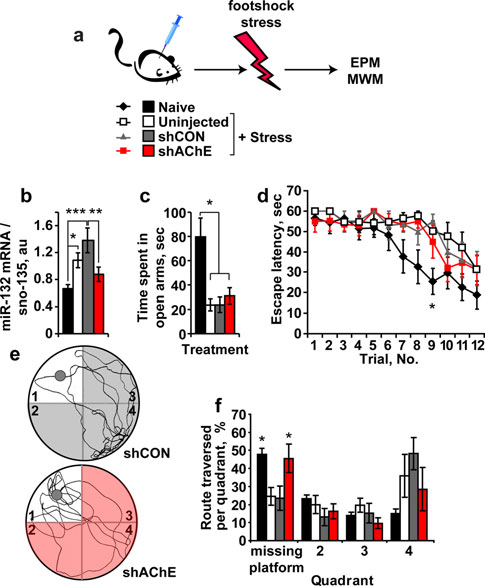
AChE suppression prevents stress-inducible cognitivedamages
Lentivirus-injected mice were subjected, 10 days post-injection, to unpredictable footshock stress compared tomice injected with a control virus (shCON) (Fig. Naı¨ve and non-injected mice exposed to the footshockstress were tested in comparison. Similar to predator scent,footshock stress alone led to 63% increase in hippocampalmiR-132, and infection with control lentiviruses extendedthe effect to a twofold increase in miR-132 levels. Theeffects on miR-132 of both stress and the chronic state ofinfection were completely prevented following shAChEsuppression (one-way ANOVA: p 0.003; Fig. sug-gesting that miR-132 elevation under stress is induced bythe reduced cholinergic neurotransmission which is pre-vented by AChE suppression. In rotorod tests, injectedmice showed normal performance reflecting unimpairedmotor coordination (Online Resources. Fig. 3). Neverthe-less, all of the stressed mice, regardless of their treatmentspent less time in the open arms of the maze compared tonaı¨ve mice (one-way ANOVA: p 0.001; Fig. dem-onstrating that preventing miR-132 increases failed toavoid the stress-inducible anxiety.
In the Morris water maze test, both shAChE-injected
stressed mice and naı¨ve mice learned to reach the hiddenplatform faster than either non-injected or shCON-injectedstressed mice (post hoc test: p 0.05 for both, Fig. d),which exhibited spatial learning deficits. By the third dayof learning trials, all four groups displayed the same escapelatencies; however, in the probe test, shAChE-injectedmice showed control-like spatial memory, unlike the defi-
Fig. 3 Lentiviral mediated AChE-suppression in hippocampal neu-
cits shown by the stressed non-injected and shCON-injec-
rons in vitro and in vivo. a The sh-799 agent is complementary toexon 2 in the endogenous mouse gene that is included in the two
ted groups. Both naı¨ve and shAChE-injected stressed mice
alternative transcripts. b Transduction efficiency of our GFP coding
crossed more frequently over the missing platform quad-
vector. Mouse primary cortical culture were transduced with vector
rant, deviating from either non-injected or shCON-injected
coding for GFP (MOI = 1). Expression reveals high transduction
mice, at least in one time point; demonstrating better
efficiency, with more than 90% of cells being transduced. c Primaryneuronal culture total AChE activity is suppressed 18, 24 and 48 h
learning capacity to reach the hidden platform and
following viral infection with shAChE-S versus shCON. Two-way
reflecting better spatial memory (two-way ANOVA:
ANOVA: treatment (F1,10 = 18.97, *p 0.01), time (F2,10 = 1.39,
p 0.05; Fig. e, f).
p = NS) and interaction (F2,10 = 0.46, p = NS). d AChE splice
shAChE-treated mice showed better cognitive perfor-
variants mRNA levels in mouse cortical primary culture followingtreatment with sh799 versus shCON: overall AChE transcript was
mance that could potentially be due to the enforced
downregulated by 80% [n = 4 (sh799 and shCON); **p 0.02].
decreases in AChE-S, the prevented increases in miR-132,
AChE-S was down-regulated by 68% [n = 3 (sh799 and shCON);
corresponding changes in other transcripts (e.g., p250GAP)
Mann–Whitney U test: *p 0.03], while the considerably lower
or to all of these reasons combined. To test for probable
AChE-R mRNA levels remained unchanged [n = 4 (sh799), n = 3(shCON); p = NS]. e Histochemical staining of AChE activity in
correlations between the measured molecular elements and
shAChE or shCON treated hippocampal sections. f Reduced AChE
the modified cognitive performance of stressed mice, we
activity in the shAChE versus shCON CA1 neuronal cell bodies and
plotted the route traversed by each mouse in the first
axons (relative to dentate gyrus; Student's t test; t13 = 18.5,
quadrant as a function of the levels of relevant RNA
**p 0.001 and t13 = 5.24, *p 0.002, respectively)
transcripts. Impressively, miR-132 levels in both control
neuronal cell bodies compared to axons in the injected area
and shRNA-injected mice showed a significant correlation
(t test: p 0.001 and p 0.002, respectively; Fig. e, f)
to the cognitive malfunction reflected in the route traversed
compared to hippocampi injected with a control virus.
in the missing platform quadrant during the probe test
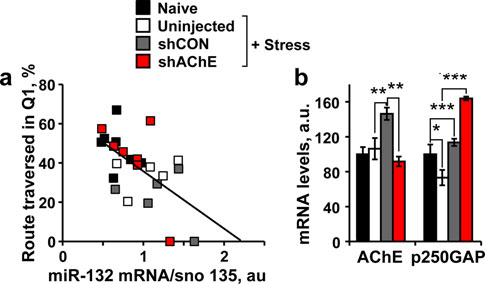
Brain Struct Funct
Fig. 5 Hippocampal miR-132 levels show inverse association withthe post-stress cognitive performance. a Significant negative corre-lation between hippocampal miR-132 levels and the route traversed inthe missing platform quadrant (Q1) in the probe test (Pearson's test:r = -0.58; p 0.01). b Suppressed hippocampal AChE-S (One-wayANOVA: F3,24 = 7.36, p 0.001; Post hoc: naı¨ve versus shCON**p 0.001; shCON versus shAChE **p 0.01) and inverseelevation
p 0.001; Post hoc: naı¨ve versus stress *p 0.05, versus shAChE***p 0.0001; stress versus shAChE ***p 0.0001) mRNA levelsin naı¨ve, stressed, shCON stressed versus shAChE stressed mice.
Note that neither AChE mRNA nor p250GAP mRNA levels showedassociation with the transverse route, attributing the cognitive declineto miR-132 itself
Fig. 4 AChE knockdown prevents the cognitive decline following
virus-injected mice but not stressed and shRNA injected
footshock stress. a Mice subjected to CA1 hippocampal injection oflentivirus-mediated knockdown of AChE (shAChE) or irrelevant
mice showed AChE mRNA up-regulation (one-way
control virus (shCON) and un-injected stressed and naı¨ve mice were
ANOVA: p 0.001, Fig. b). Furthermore, as observed in
exposed 3 weeks later to 7 unpredicted and inescapable footshock
the predator scent test, p250GAP was down-regulated
stress followed by EPM and Morris water maze (MWM) tests.
following footshock stress, and intercepting AChE and
b Hippocampal miR-132 mRNA levels increase by 63 and 100% instressed and shCON stressed mice, but remained unchanged in
miR-132 up-regulation resulted in enlarged p250GAP
shAChE stressed mice. ANOVA test: F3,28 = 6.14, p 0.003. Post
increases (one-way ANOVA: p 0.001; Fig. How-
hoc LSD test: naı¨ve versus stressed *p 0.05, versus shCON-
ever, neither AChE-S mRNA nor p250GAP levels were
infected and stressed ***p 0.001; shCON-infected and stressed
associated to the cognitive performance.
versus shAChE-infected and stressed mice **p 0.01. c Naı¨ve micespend more time in the EPM open arms than all stressed groups (one-way ANOVA: F3,28 = 8.49, p 0.001). d Naı¨ve and shAChE-
Chronic hippocampal miR-132 increases associate
infected stressed mice learn to reach the MWM hidden platform faster
with AChE-S and p250GAP decreases
than stressed or shCON stressed mice. Two-way ANOVA: trialnumber (F11,323 = 12.33, p 0.001), treatment (F3,323 = 10.11,p 0.001), interaction (F33,323 = 0.75, p = NS). Bonferroni post
To further challenge the causal association between miR-
hoc comparison—trial no. 9: naı¨ve versus stressed *p 0.05, versus
132 and stress-inducible cognitive malfunctioning, we
shCON-infected and stressed *p 0.05, versus shAChE-infected and
sought an experimental model where miR-132 would be
stressed p = NS. e Representative illustrations of mice swimming
elevated and AChE-S would be diminished. The AChE-R
tracks in the MWM probe test (gray circles: place of the missingplatform in quadrant #1): Bottom shAChE stressed mouse. Top
over-expressing TgR mice present such a system. In TgR
shCON stressed mouse. f Naı¨ve and shAChE stressed groups display
mice, enforced expression of the human hAChE-R tran-
more crosses over the previously situated-platform quadrant. Two-
script leads to excessive ACh hydrolysis which in turn
way ANOVA: quadrant (F3,100 = 9.95, p 0.0001), treatment
elevates brain miR-132 levels (Shaked et al. The
(F3,100 = 0.002, p = NS), interaction (F9,100 = 3.38, p 0.012).
Bonferroni post hoc comparison for quadrant #1 (where the platform
elevated miR-132 may target nascent host AChE-S mRNA
was previously situated): Naı¨ve versus stressed *p 0.05, versus
but not the transgenic 30UTR-null AChE-R transcripts
shCON-infected and stressed *p 0.05, versus shAChE-infected and
(Fig. a). Correspondingly, TgR mice show an anxiogenic-
stressed p = NS; shCON-infected and stressed versus shAChE-
like phenotype accompanied by hyper-reactivity to nicotine
infected and stressed *p 0.05
(Salas et al. ). Hippocampal miR-132 levels increased
(Pearson's test: r = -0.58; p 0.01; Fig. Normal
by *2.7-fold in TgR mice compared to non-transgenic
AChE mRNA levels were already retrieved at this time
FVB/Ns (Student's t test: p 0.001; Fig. This pre-
following footshock stress; however, stressed and control
dictably led to suppression of both host AChE-S and yet
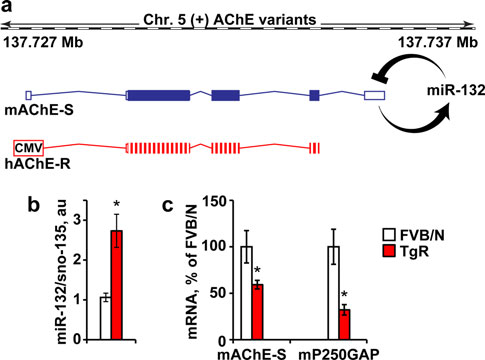
Brain Struct Funct
Fig. 6 Hippocampal miR-132 elevation and suppressed AChE-S andp250GAP in TgR mice. a The mouse AChE gene in chromosome 5encodes the miR-132-targeted synaptic mAChE-S transcript, whereasthe CMV-regulated transgenic 30 UTR-null human AChE-R is miR-132 refractory. b Hippocampal miR-132 mRNA levels are increasedby 2.7-fold in TgR compared to non-transgenic FVB/N mice(Student's t test, t19 = 3.72, *p 0.001). c Hippocampal AChE-Sand p250GAP mRNA levels, are both reduced in TgR versus FVB/Nmice (Student's t tests: *p 0.05)
more so, of p250GAP levels (Student's t test: p 0.05;Fig. Therefore, if AChE-S suppression could by itselfprevent stress-inducible cognitive decline, TgR mice shouldshow no such decline; but if miR-132 increases are thecause, then these mice should present a stress phenotype.
Chronic AChE-S suppression associateswith intensified cholinergic hyper-excitation
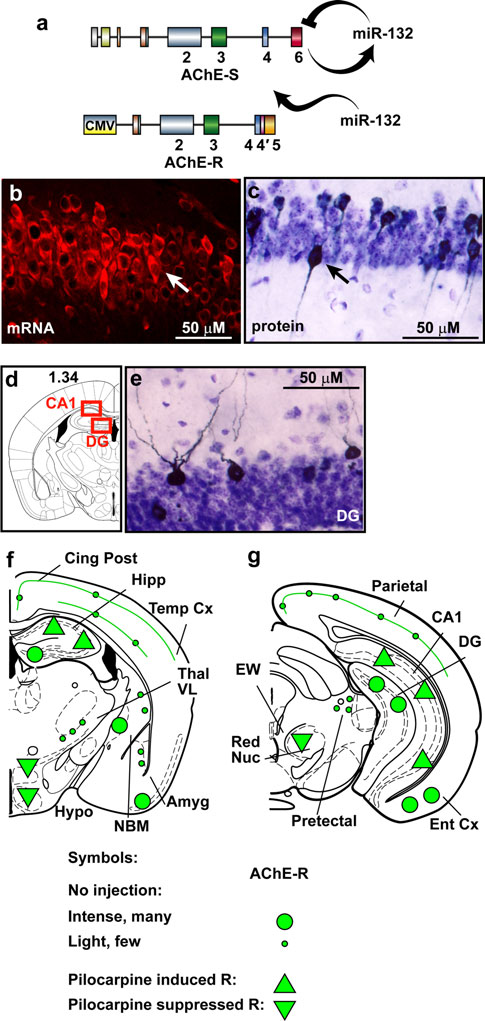
The AChE-R transgene avoids miR-132 surveillance owingto the lack of its native 30-UTR (Fig. Therefore, the TgRbrain presents continuous excess of AChE-R which cannotbe suppressed, mimicking prolonged conditions of stress(Meshorer and Soreq ). Of note, choline acetyl trans-ferase (ChAT)-expressing cholinergic neurons appeared inthe engineered brains in normal numbers and size, with anormal symmetric distribution between right and left hemi-
Fig. 7 miR-132-refractory AChE excess induces cholinergic hyper-
spheres. In general, the engineered AChE-R protein accu-
reactivity. a Mouse AChE-S, but not transgenic AChE-R with a
mulated in cholinoceptive brain regions that tend to express
truncated 30-UTR, is recognized by miR-132. b CA1 TgR neurons
the primary synaptic AChE-S variant [e.g., the CA1 and
display higher than background expression of human AChE-RmRNA. c–d CA1 and dentate gyrus (DG) TgR neurons express
dentate gyrus (DG) in the hippocampus and the entorhi-
higher than background human AChE-R protein. f–g Schemes of TgR
nal cortex]. Typically, the neuronal cytoplasm, nucleus
coronal brain sections showing mouse and transgenic-human AChE-R
and dendrite(s), but not axons, were stained (Fig. b–e).
labeling patterns in hippocampal regions (e.g., dentate gyrus, DG,
Furthermore, TgR mice presented an extreme sensitivity to
CA1), subcortical regions [e.g. red nucleus (RN), lateral hypothala-mus (LH), amygdala (Amyg)] and in the entrohinal cortex (Ent Cx).
cholinergic stimulators. Thus, intraperitoneal exposure to
Large and small green circles denote intense or faint transgenic
25 mg/kg of the muscarinic agonist pilocarpine (Dickson
protein expression, respectively. Green triangles show intracellular
and Alonso, induced massive up-regulation of
increases (up) or decreases (down) in host and transgenic protein
AChE-R in multiple cholinergic brain regions (Fig. g).
expression under pilocarpine treatment, respectively
Specifically, the hippocampus and entorhinal cortex showed
Brain Struct Funct
numerous AChE-expressing cells with further intensified
AChE protein increases in the hippocampal CA1 region of
labeling under pilocarpine treatment. Under control condi-
the transgenic mice, revealing an hyper-excitatory sensitiv-
tions, several regions showed intense AChE-R labeling; but,
ity (Frankland et al. ) with c-fos significantly correlated
pilocarpine treatment caused massive AChE-R elevation
to AChE-R labeling (Pearson's test: saline r = 0.86,
throughout the hippocampus (Fig. Yet more specifi-
p 0.05; pilocarpine r = 0.77, p 0.05; Fig. c).
cally, both TgR and strain-matched FVB/N control miceshowed intensified labeling under pilocarpine administration
TgR mice show motion and cognitive malfunctioning
compared to saline-injected mice of the Ca2?-responsiveCREB-dependent c-fos mRNA, associated with contextual
Nocturnal activity monitoring of TgR mice versus non-
fear conditioning (two-way ANOVA: p 0.05; Fig. a).
transgenic controls revealed interchangeable hyper- and
The c-fos labeling pattern largely overlapped the observed
hypo-locomotion activity of the transgenics during thedark, but not the light phase of the day compared to con-trols (Fig. ANOVA repeated measures: p 0.001).
Given parallel, yet distinct changes in the nocturnal activityof mice over-expressing the synaptic AChE-S variant(Cohen et al. the altered nocturnal activity of theTgR mice likely reflects changes in cholinergic signaling.
At the molecular level, hippocampal host AChE-S proteinamounts were 55% reduced in TgR mice compared toFVB/N controls (t test: p 0.01; Fig. To evaluatelearning capacities and adaptive behavior, we subjected
Fig. 9 TgR mice show nocturnal hyper locomotion, suppressedACHE-S and impaired memory. a Nocturnal locomotion patterns ofTgR versus FVB/N mice. One-way ANOVA: Light F1,22 = 0.4,p = NS; Dark F1,22 = 79.02, p 0.001. b Reduced hippocampal
Fig. 8 Pilocarpine induces robust hyperactivation of transgene and
mAChE-S protein levels in TgR mice compared to FVB/N mice
c-fos expression in the TgR brain. a Pilocarpine induces CA1 c-fos
(Student's t test: t13 = 2.71, *p 0.01). c A serial maze task requires
and AChE-R protein increases in TgR mice. b Pilocarpine-induced
a water-deprived mouse to find a sweetened water reward at each end
c-fos activation in the CA1 TgR hippocampus: two-way ANOVA
of the maze. The mouse must shuffle five times between the two ends
shows a significant effect of treatment (F1,19 = 226, p 0.0001) with
of the maze to obtain five rewards. d TgR mice display lower serial
a significant interaction between strain and treatment (F2,19 = 5.88,
maze performance than FVB/Ns. Right/left errors: F1,33 = 9.97;
p 0.05). c AChE-R labeling associates with c-fos activation.
Retrace errors: F1,33 = 9.37, *p 0.005. ANOVA with repeated
Pearson's test: for saline r = 0.86, p 0.05 and for pilocarpine
measures reveals a significant transgene effect in the two measures of
Brain Struct Funct
these mice to the serial choice maze and measured theanimals' ability to avoid right or left turning and/or retraceerrors (See Fig. c for a scheme of the maze). The cogni-tive test was conducted during the light phase of the daywhere no changes in total locomotion activity wereobserved between the two tested groups. TgR mice dis-played significantly more right/left choice errors andretrace errors compared to control FVB/N mice (two-wayANOVA: p 0.005; Fig. d). Close examination of vid-eotaped maze behavior revealed two major types of error:‘‘trapping behavior''—running in a repeated path severaltimes without correction, and lack of spatial orientationwith respect to reward location, manifested in repeatedvisits at the end of the maze where the last reward wasgiven and hence no reward was to be expected. Our tests ofsensory-motor function did not reveal deficits (OnlineResources: Methods) and thus excluded the possibility thattrapping behavior was due to the secondary effects of thetransgenic intervention.
Stress-inducible total alterations and inter-individualvariability in miR-132 and p250GAP
Our working hypothesis predicted that the key processeswhich are activated under stress conditions would be com-mon to different animal strains and stress paradigms.
Therefore, we compared the outcome of the three models westudied integrated together by calculating the percentagechange under stress in specific hippocampal transcripts. Thisanalysis again showed increases in miR-132 in both thepredator scent (t test: p 0.01) and the AChE-R excessmodels (p 0.01), but not in the footshock stress under
Fig. 10 Integrated analysis of the 3 stress models. Shown in fold
shAChE, which led to miR-132 decreases (p 0.05); an
changes from controls are the inter-animal variability values and meanchanges in hippocampal miR-132, AChE and p250GAP transcript
accompanying decline in p250GAP in both models
levels in the predator scent (a, Student's t test: miR-132 **p 0.01,
(p 0.01 and p 0.05 for the predator scent and AChE-R
AChE p = NS, p250GAP **p 0.01), footshock (b, Student's t test:
excess models, respectively) with miR-132 increases. In
miR-132 *p 0.05, AChE *p 0.05, p250GAP *p 0.05) and
addition, in all three models, the stress-inducible changes in
transgenic AChE (c, Student's t test: miR-132 **p 0.01, AChE*p 0.05, p250GAP *p 0.05) stress models employed in our study.
the hippocampal levels of miR-132 and its p250GAP and
d Scheme of the proposed mechanism involved
AChE targets showed inverse patterns of individual vari-ability. Thus, both predator-stressed C57BI/6J and the
p250GAP. Our analysis thus attributes the observed stress-
chronically anxious TgR FVB/N mice showed larger inter-
inducible cholinergic hyper-excitation to the feed-forward
animal hippocampal miR-132 variability compared to mat-
regulation of miR-132 and AChE transcripts, and shows that
ched controls. Inversely, they both presented smaller
avoiding p250GAP reduction and the corresponding breadth
p250GAP variability under stress than in control mice
of inter-individual variability in miR-132 associates with
(Fig. , Online Resources Fig. 4), suggesting that miR-132
stress-inducible cholinergic hyper excitation and cognitive
changes serve to mitigate p250GAP variability under stress
impairments (Fig.
in these two models. In comparison, C57BI/6J mice sub-jected to shAChE knockdown predictably showed a ten-dency to reduce AChE levels (60–80% of control levels;
p 0.05), while p250GAP levels were elevated in thismodel (p 0.5; Fig. ). These findings are compatible
Using three different mouse stress models, we found long-
with the hypothesis that the inter-individual variability in
lasting stress-inducible enhancement of both the levels
hippocampal miR-132 is inversely interlocked with that of
and the individual variability in hippocampal miR-132
Brain Struct Funct
expression. This phenomenon was accompanied by the
(Salas et al. hyper-excitation in TgR mice is likely
suppression of the levels of hippocampal AChE and the
due to the failure of miR-132 in these mice to suppress
GTPase activator p250GAP, which are both validated
AChE. This hypothesis is reinforced by the co-expression
miR-132 targets. Knockdown of AChE production greatly
of TgR AChE with the early immediate protein c-fos,
limited miR-132 increases in a footshock stress model,
which is expressed in neurons whose activity is strongly
suggesting continuous surveillance by cholinergic signaling
stimulated by synaptic input (Dragunow and Faull ;
of miR-132 levels in the hippocampal which is disrupted
Frankland et al. ). c-fos is also essential for hippo-
under psychological stress due to AChE over-production.
campus-dependent learning and memory (Fleischmann
Suppressing AChE further prevented footshock stress-
et al. and represents part of the signal transduction
inducible damages in cognition, but not anxiety, attributing
cascade underlying the molecular basis of long-term
to miR-132 a regulatory role over post-stress cognition but
potentiation (Miyamoto suggesting relevance for the
not anxiety. Corroborating this finding, engineered mice
observed cognitive impairments in TgR mice. In stressed
with chronic excess of both miR-132 and engineered AChE
wild type mice, however, activation of the AChE gluco-
showed an anxiogenic-like phenotype, impaired locomo-
corticoid-responsive element also takes place (Meshorer
tion and cognition, and cholinergic hyper-excitation when
and Soreq ). This might reduce ACh levels and con-
exposed to pilocarpine. Of note, hippocampal AChE
sequently suppress CREB-inducible miR-132 transcription,
mRNA levels remained elevated 7 days following predator
regaining homeostasis. Hippocampal miR-132 was not
scent test and 14 days following footshock stress accom-
up-regulated following stress when AChE-S was sup-
panied by chronic hippocampal lentiviral infection. In the
pressed is compatible with proposed inter-locked regula-
pre-frontal cortex, we found weeks-long elevation of AChE
tion of these two stress-inducible genes (Fig. d).
following mild stress (Meshorer et al. ). In the hip-
Cre-lox mediated deletion of miR-132 in newborn
pocampus, we noted such elevation during 1 h and 1 day
hippocampal neurons decreases dendrite length and
post-stress (Kaufer et al. , Nijholt et al. AChE
arborization in adult mice (Magill et al. ), supporting a
levels were normal in mice 14 days following footshock
long-lasting role for miR-132 in neuronal differentiation,
stress alone thus indicates the transient nature of this stress
synaptogenesis and maintenance. The GTPase activator
in the hippocampus.
p250GAP emerges as an additional target of miR-132
In predator-stressed C57BI/6J mice and in FVB/N mice
which is involved in mediating the stress-inducible cogni-
with engineered over-expression of AChE, we found ele-
tive malfunctioning. Suppressing p250GAP in cultured
vated inter-animal variability of hippocampal miR-132
cortical neurons enhances neurite sprouting, similar to the
levels which was accompanied by narrower variability of
neurological reaction following stress (Kawashima et al.
its p250GAP target, suggesting causal links between miR-
). Both neuronal activity and the GABAA inhibitor
132 and this neuronal protein. Given the in-bred features of
bicuculline induce miR-132 transcription, which further
these mouse strains, we hypothesized that the stress-
down-regulates p250GAP and might enhance neurite
inducible inter-animal variability in hippocampal miR-132
growth (Wayman et al. Also, miR-132 is up-regu-
reflects life-long differences in individual experience
lated during post-natal development, when massive neurite
which mediate these changes. Reinforcing this notion,
sprouting occurs; miR-132-targeted antisense oligonucle-
personal experience determines much of the stress reac-
otides attenuate neurite growth (Ponomarev et al.
tions in human patients with post-traumatic stress disorder
The reported link between p250GAP and the NMDA
(Feder et al. ).
receptor (Nakazawa et al. further supports a causal
Both miR-132 and AChE transcription are controlled by
role in the post-stress cognitive impairments for p250GAP
CREB (Shaked et al. which is notably involved in
suppression. Likewise, engineered anti-sense suppression
learning and memory, neural growth and by the neuronal
of AChE modulates neuronal sprouting in the mouse hip-
growth factor BDNF associated with cholinergic func-
pocampus (Sklan et al. Our findings of miR-132
tioning (Cogswell et al. ; Im et al. ; Wayman
excess in the hippocampus of mature TgR mice and in
et al. ). Correspondingly, individual differences in
footshock-stressed mice over a month after the insult, and
response to chronic stress were recently attributed to hip-
7 days post-predator scent stress are compatible with the
pocampal BDNF (Taliaz et al. ). Also, contextual fear
hypothesis that the long-term dual suppression of AChE
conditioning increases pri-miR-132 levels in the mouse
and p250GAP in the hippocampus might serve as a com-
hippocampus (Ponomarev et al. and in the hippo-
pensatory mechanism to balance the damages associated
campus of chronically-stressed rats (Meerson et al.
with excessive neuronal sprouting.
and the cholinergic agonist pilocarpine leads to a transient
AChE suppression prevents both miR-132 increases and
up-regulation of pri- and mature-miR-132 (Nudelman et al.
the accompanying cognitive malfunctioning that suggests
). The observed pilocarpine and nicotine-inducible
the stress-associated changes in neurite sprouting is a
Brain Struct Funct
pivotal cause of these damages, and opens new venues for
treating trauma patients by mitigating irreversible changesin their neuronal network. Nevertheless, our study did not
Alvarez-Saavedra M, Antoun G, Yanagiya A, Oliva-Hernandez R,
exclude the involvement of other experimentally validated
Cornejo-Palma D, Perez-Iratxeta C, Sonenberg N, Cheng HY
miR-132 targets. For example, the miR-132-targeted light-
(2011) miRNA-132 orchestrates chromatin remodeling andtranslational control of the circadian clock. Hum Mol Genet
induced transcription regulatory factor X 4 (RFX4),
20(4):731–751. doi:
abundant in the supra-chiasmatic nucleus (SCN) of the
Bartel DP (2009) MicroRNAs: target recognition and regulatory
hypothalamus, regulates biological clocks and rhythms
functions. Cell 136(2):215–233.
(Alvarez-Saavedra et al. Cheng and Obrietan
Berson A, Knobloch M, Hanan M, Diamant S, Sharoni M, Schuppli
D, Geyer BC, Ravid R, Mor TS, Nitsch RM, Soreq H (2008)
In wild type mice, miR-132 levels are lower during the
Changes in readthrough acetylcholinesterase expression modu-
dark part of the circadian cycle in the SCN (Cheng et al.
late amyloid-beta pathology. Brain 131(Pt 1):109–119
). The nocturnal locomotive fluctuations in the TgR
Blank T, Nijholt I, Eckart K, Spiess J (2002) Priming of long-term
mice may therefore reflect circadian variations in ACh lev-
potentiation in mouse hippocampus by corticotropin-releasingfactor and acute stress: implications for hippocampus-dependent
els, (Erb et al. which lead to uncontrolled miR-132
learning. J Neurosci 22(9):3788–3794
and RFX4 levels. The miR-132/-212 cluster also targets the
Carter CS, Braver TS, Barch DM, Botvinick MM, Noll D, Cohen JD
Rett syndrome-related MeCP2 co-factor of the neuronal
(1998) Anterior cingulate cortex, error detection, and the online
transcription silencer REST (Klein et al. REST
monitoring of performance. Science 280(5364):747–749
Chen CZ, Li L, Lodish HF, Bartel DP (2004) MicroRNAs modulate
binding to the regulatory huntingtin protein is impaired
hematopoietic lineage differentiation. Science 303(5654):83–86
during the progression of Huntington's disease (Packer et al.
Cheng HY, Obrietan K (2007) Revealing a role of microRNAs in the
), suggesting parallel stress-associated effects. Inver-
regulation of the biological clock. Cell Cycle 6(24):3034–3035
sely, rapid ischemic pre-conditioning, which protects the
Cheng HY, Papp JW, Varlamova O, Dziema H, Russell B, Curfman
brain from subsequent prolonged ischemia (Lusardi et al.
JP, Nakazawa T, Shimizu K, Okamura H, Impey S, Obrietan K
) is accompanied by miR-132 decreases and MeCP2
(2007) MicroRNA modulation of circadian-clock period and
increases. MiR-132 also targets the fragile-X mental retar-
entrainment. Neuron 54(5):813–829
dation protein FMRP, knockdown of which abolishes the
Cogswell JP, Ward J, Taylor IA, Waters M, Shi Y, Cannon B, Kelnar
K, Kemppainen J, Brown D, Chen C, Prinjha RK, Richardson
morphological effects of miR-132 transfection. This sug-
JC, Saunders AM, Roses AD, Richards CA (2008) Identification
gests competitive interaction between FMRP and p250GAP
of miRNA changes in Alzheimer's disease brain and CSF yields
which may balance out the miR-132-mediated effect on
putative biomarkers and insights into disease pathways. J Alz-
neuronal sprouting (Wayman et al. In addition,
heimers Dis 14(1):27–41
Cohen O, Erb C, Ginzberg D, Pollak Y, Seidman S, Shoham S,
miR-132 is predicted to target several ion channels, and
Yirmiya R, Soreq H (2002) Neuronal overexpression of ‘read-
might thus affect cell excitability; correspondingly, over-
through' acetylcholinesterase is associated with antisense-sup-
expressed pre-miR-132 potentiates glutamate, NMDA, or
pressible behavioral impairments. Mol Psychiatry 7(8):874–885
K?-mediated depolarization of cultured neurons, suggesting
Cohen H, Kaplan Z, Matar MA, Loewenthal U, Kozlovsky N, Zohar J
(2006) Anisomycin, a protein synthesis inhibitor, disrupts
global involvement in regulating neurotransmission and
traumatic memory consolidation and attenuates posttraumatic
plasticity (Wibrand et al. ; Edbauer et al. which
stress response in rats. Biol Psychiatry 60(7):767–776
may either be direct or function through p250GAP. The
Diamond DM, Park CR, Heman KL, Rose GM (1999) Exposing rats
multitude targets of neuronal-expressed miRs thus point at
to a predator impairs spatial working memory in the radial armwater maze. Hippocampus 9(5):542–552
combinatorial, rather than single miR-target relevance.
Dickson CT, Alonso A (1997) Muscarinic induction of synchronous
population activity in the entorhinal cortex. J Neurosci 17:
The authors are grateful to Drs. O. Cohen and
G. Zimmerman (Jerusalem) for their contribution to this study. Also
Dragunow M, Faull R (1989) The use of c-fos as a metabolic marker
acknowledged is support by the Israel Science Foundation Legacy
in neuronal pathway tracing. J Neurosci Methods 29(3):261–265
Heritage Biomedical Science Partnership (Grant No. 378/11), the
Edbauer D, Neilson JR, Foster KA, Wang CF, Seeburg DP, Batterton
Gatsby Foundation and the German Research Foundation Trilateral
MN, Tada T, Dolan BM, Sharp PA, Sheng M (2010) Regulation
Cooperation Program (to H.S.). G.S. was the incumbent of an Eshkol
of synaptic structure and function by FMRP-associated microR-
post-doctoral fellowship by the Israel Ministry of Science, M.H. and
NAs miR-125b and miR-132. Neuron 65(3):373–384. doi:
S.B. were both awarded pre-doctoral fellowships by the Edmond and
Lily Safra Center for Brain Sciences.
Ellman GL, Courtney KD, Andres V Jr, Feather-Stone RM (1961) A
new and rapid colorimetric determination of acetylcholinesterase
Conflict of interest
The authors declare that they have no conflict
activity. Biochem Pharmacol 7:88–95
of interest.
Erb C, Troost J, Kopf S, Schmitt U, Loffelholz K, Soreq H, Klein J
(2001) Compensatory mechanisms enhance hippocampal ace-
This article is distributed under the terms of the Cre-
tylcholine release in transgenic mice expressing human acetyl-
ative Commons Attribution Noncommercial License which permits
cholinesterase. J Neurochem 77(2):638–646
any noncommercial use, distribution, and reproduction in any medium,
Farchi N, Ofek K, Podoly E, Dong H, Xiang YY, Diamant S, Livnah
provided the original author(s) and source are credited.
O, Li J, Hochner B, Lu WY, Soreq H (2007) Peripheral site
Brain Struct Funct
acetylcholinesterase blockade induces RACK1-associated neu-
Meshorer E, Soreq H (2002) Pre-mRNA splicing modulations in
ronal remodeling. Neurodegener Dis 4(2–3):171–184
senescence. Aging Cell 1(1):10–16
Feder A, Nestler EJ, Charney DS (2009) Psychobiology and molecular
Meshorer E, Soreq H (2006) Virtues and woes of AChE alternative
genetics of resilience. Nat Rev Neurosci 10(6):446–457. doi:
splicing in stress-related neuropathologies. Trends Neurosci
Filipowicz W, Bhattacharyya SN, Sonenberg N (2008) Mechanisms of
Meshorer E, Erb C, Gazit R, Pavlovsky L, Kaufer D, Friedman A,
post-transcriptional regulation by microRNAs: are the answers in
Glick D, Ben-Arie N, Soreq H (2002) Alternative splicing and
sight? Nat Rev Genet 9(2):102–114
neuritic mRNA translocation under long-term neuronal hyper-
Fleischmann A, Hvalby O, Jensen V, Strekalova T, Zacher C, Layer
sensitivity. Science 295(5554):508–512
LE, Kvello A, Reschke M, Spanagel R, Sprengel R, Wagner EF,
Miyamoto E (2006) Molecular mechanism of neuronal plasticity:
Gass P (2003) Impaired long-term memory and NR2A-type
induction and maintenance of long-term potentiation in the
NMDA receptor-dependent synaptic plasticity in mice lacking
hippocampus. J Pharmacol Sci 100(5):433–442
c-fos in the CNS. J Neurosci 23(27):9116–9122
Nakazawa T, Watabe AM, Tezuka T, Yoshida Y, Yokoyama K,
Frankland PW, Bontempi B, Talton LE, Kaczmarek L, Silva AJ
Umemori H, Inoue A, Okabe S, Manabe T, Yamamoto T (2003)
(2004) The involvement of the anterior cingulate cortex in
p250GAP, a novel brain-enriched GTPase-activating protein for
remote contextual fear memory. Science 304(5672):881–883.
Rho family GTPases, is involved in the N-methyl-d-aspartate
receptor signaling. Mol Biol Cell 14(7):2921–2934.
Goel N, Bale TL (2010) Sex differences in the serotonergic influence
on the hypothalamic-pituitary-adrenal stress axis. Endocrinology
Nijholt I, Farchi N, Kye M, Sklan EH, Shoham S, Verbeure B, Owen
D, Hochner B, Spiess J, Soreq H, Blank T (2004) Stress-induced
Gray R, Rajan AS, Radcliffe KA, Yakehiro M, Dani JA (1996)
alternative splicing of acetylcholinesterase results in enhanced
Hippocampal synaptic transmission enhanced by low concentra-
fear memory and long-term potentiation. Mol Psychiatry 9(2):
tions of nicotine. Nature 383(6602):713–716. doi:
Nudelman AS, DiRocco DP, Lambert TJ, Garelick MG, Le J,
Im H-I, Hollander JA, Bali P, Kenny PJ (2010) MeCP2 controls BDNF
Nathanson NM, Storm DR (2010) Neuronal activity rapidly
expression and cocaine intake through homeostatic interactions
induces transcription of the CREB-regulated microRNA-132, in
with microRNA-212. Nat Neurosci 13(9):1120-–127.
vivo. Hippocampus 20(4):492–498.
Packer AN, Xing Y, Harper SQ, Jones L, Davidson BL (2008) The
bifunctional microRNA miR-9/miR-9* regulates REST and
Karnovsky MJ, Roots L (1964) A ‘‘Direct-Coloring'' thiocholine
CoREST and is downregulated in Huntington's disease. The
method for cholinesterases. J Histochem Cytochem 12:219–221
Journal of neuroscience : the official journal of the Society for
Kaufer D, Friedman A, Seidman S, Soreq H (1998) Acute stress
Neuroscience 28(53):14341–14346.
facilitates long-lasting changes in cholinergic gene expression.
Ponomarev ED, Veremeyko T, Barteneva N, Krichevsky AM, Weiner
Kawashima H, Numakawa T, Kumamaru E, Adachi N, Mizuno H,
HL (2010) MicroRNA-124 promotes microglia quiescence and
Ninomiya M, Kunugi H, Hashido K (2010) Glucocorticoid
suppresses EAE by deactivating macrophages via the C/EBP-
attenuates brain-derived neurotrophic factor-dependent upregula-
[alpha]-PU.1 pathway. Nat Med advance online publication.
tion of glutamate receptors via the suppression of microRNA-132
expression. Neuroscience 165(4):1301–1311.
Quartermain D, Mower J, Rafferty MF, Herting RL, Lanthorn TH
Klein ME, Lioy DT, Ma L, Impey S, Mandel G, Goodman RH (2007)
(1994) Acute but not chronic activation of the NMDA-coupled
Homeostatic regulation of MeCP2 expression by a CREB-
glycine receptor with D-cycloserine facilitates learning and
induced microRNA. Nat Neurosci 10(12):1513–1514
retention. Eur J Pharmacol 257(1–2):7–12
Krol J, Loedige I, Filipowicz W (2010) The widespread regulation of
Rana TM (2007) Illuminating the silence: understanding the structure
microRNA biogenesis, function and decay. Nat Rev Genet
and function of small RNAs. Natl Rev Mol Cell Biol 8(1):
11(9):597–610. doi:
Lusardi TA, Farr CD, Faulkner CL, Pignataro G, Yang T, Lan J, Simon
Salas R, Main A, Gangitano DA, Zimmerman G, Ben-Ari S, Soreq H,
RP, Saugstad JA (2010) Ischemic preconditioning regulates
De Biasi M (2008) Nicotine relieves anxiogenic-like behavior in
expression of microRNAs and a predicted target, MeCP2, in
mice that overexpress the read-through variant of acetylcholin-
mouse cortex. J Cereb Blood Flow Metab 30(4):744–756. doi:
esterase but not in wild-type mice. Mol Pharmacol 74(6):1641–
Magill ST, Cambronne XA, Luikart BW, Lioy DT, Leighton BH,
Shaked I, Meerson A, Wolf Y, Avni R, Greenberg D, Gilboa-Geffen
Westbrook GL, Mandel G, Goodman RH (2010) MicroRNA-132
A, Soreq H (2009) MicroRNA-132 potentiates cholinergic anti-
regulates dendritic growth and arborization of newborn neurons
inflammatory signaling by targeting acetylcholinesterase. Immu-
in the adult hippocampus. Proc Natl Acad Sci USA 107(47):
nity 31(6):965–973
Sklan EH, Berson A, Birikh KR, Gutnick A, Shahar O, Shoham S,
McEwen BS, Gianaros PJ (2011) Stress- and allostasis-induced brain
Soreq H (2006) Acetylcholinesterase Modulates Stress-Induced
plasticity. Annu Rev Med 62:431–445. doi:
Motor Responses Through Catalytic and Noncatalytic Properties.
Biol Psychiatry 60:741–751
Meerson A, Cacheaux L, Goosens KA, Sapolsky RM, Soreq H,
Soreq H, Wolf Y (2011) NeurimmiRs: microRNAs in the neuroim-
Kaufer D (2010) Changes in brain microRNAs contribute to
mune interface. Trends Mol Med 17(10):548–555
cholinergic stress reactions. J Mol Neurosci 40(1–2):47–55
Sternfeld M, Shoham S, Klein O, Flores-Flores C, Evron T, Idelson
Meshorer E, Bryk B, Toiber D, Cohen J, Podoly E, Dori A, Soreq H
GH, Kitsberg D, Patrick JW, Soreq H (2000) Excess ‘‘read-
(2005) SC35 promotes sustainable stress-induced alternative
through'' acetylcholinesterase attenuates but the ‘‘synaptic''
splicing of neuronal acetylcholinesterase mRNA. Mol Psychiatry
variant intensifies neurodeterioration correlates. Proc Natl Acad
Sci USA 97(15):8647–8652
Brain Struct Funct
Taliaz D, Loya A, Gersner R, Haramati S, Chen A, Zangen A (2011)
S (2008) An activity-regulated microRNA controls dendritic
Resilience to chronic stress is mediated by hippocampal brain-
plasticity by down-regulating p250GAP. Proc Natl Acad Sci
derived neurotrophic factor. The Journal of neuroscience : the
USA 105(26):9093–9098
official journal of the Society for Neuroscience 31(12):
Wibrand K, Panja D, Tiron A, Ofte ML, Skaftnesmo KO, Lee CS,
Pena JT, Tuschl T, Bramham CR (2010) Differential regulation
Vo N, Klein ME, Varlamova O, Keller DM, Yamamoto T, Goodman
of mature and precursor microRNA expression by NMDA and
RH, Impey S (2005) A cAMP-response element binding protein-
metabotropic glutamate receptor activation during LTP in the
induced microRNA regulates neuronal morphogenesis. Proc Natl
adult dentate gyrus in vivo. Eur J Neurosci 31(4):636–645. doi:
Acad Sci USA 102(45):16426–16431
Wayman GA, Davare M, Ando H, Fortin D, Varlamova O, Cheng
HY, Marks D, Obrietan K, Soderling TR, Goodman RH, Impey
Source: http://knowledgestream.ru/system/uploads/lecture/file/file_en/7/Hippocampal_microRNA-132_mediates_stress-inducible_cognitive_deficits.pdf
COMMITTEE ON SOCIAL POLICY OF THE JOGORKU KENESH OF THE KYRGYZ REPUBLIC Special report on the results of monitoring and evaluation of implementation of the Law of the Kyrgyz Republic «On Preventing and Combating Trafficking in Persons» COMMITTEE ON SOCIAL POLICY OF THE JOGORKU KENESH OF THE KYRGYZ REPUBLIC
37412_SpanishCover:37412_SpanishCover 9/17/09 10:37 AM Page 1 Publicado por la American Society for Reproductive Medicine, bajo la dirección del Comité de Educación del Paciente y el Comité de Publicaciones. Ninguna parte en este documento puede ser reproducida en ninguna forma sin permiso por escrito. Este folleto no pretende de ninguna manera sustituir, dictar ni definir















