Staytonfire.org
OF CONTENTS
These patient care protocols will go into effect in January 2016 for EMS Responders of the Aumsville Fire District, Gates Fire District, Falck Ambulance, Idanha Detroit Rural Fire Protection District, Keizer Fire District, Lyons Fire District, Marion County Fire District #1, Mill City Fire District, Salem Fire Department, Santiam Ambulance, Stayton Fire District, Sublimity Fire District and Turner Fire District.
These protocols, we believe, are the best of their type. Where evidence has been available, the Regional Protocol Committee has drafted protocols that will assist us in providing excellent patient care. Where evidence is lacking, we have relied on best practices, expert advice and consensus to guide the development of the protocol or procedure. These protocols are reviewed on a regular basis, updated when necessary to reflect the advances in the art and science pertaining to the care of the acutely ill, and injured.
Remember that these protocols are guidelines. EMS is performed in a stressful environment with time-critical decisions and no specific patient care matrix can be developed that will cover every type of injury, illness, and complicating circumstances that EMTs will encounter while providing on-scene care. It is our expectation that providers will use these protocols in conjunction with their training and experience to do what is best for each patient. From time to time, it is expected that circumstances will arise that are not covered within these protocols. In such instances, providers should function within their scope of practice and use all available resources (including On-Line Medical Control) to provide the best possible patient care.
Thanks to everyone who has provided assistance in protocol development and review. Anything that is complex and includes detail is prone to errors. Please review these protocols carefully and route any potential errors, unclear directions or suggestions for improvement to your agency's EMS Office.
Finally, we thank every one of you for your dedication and commitment every day to providing
the best possible pre-hospital medical care to the citizens of our respective communities•.
Salem Fire Department
Falck Ambulance
Marion County Fire District #1 Aumsville Fire District
Gates Fire District
Idanha Detroit Fire District
Lyons Fire District & Ambulance
Mill City Fire District
Santiam Ambulance
Stayton Fire District
Sublimity Fire District
Turner Fire District & Ambulance
Last Revision and Signed January 2016
SCOPE OF PRACTICE (OAR 847-035-0030)
EMERGENCY MEDICAL RESPONDER
An Emergency Medical Responder may:
A. Conduct primary and secondary patient examinations; B. Take and record vital signs; C. Utilize noninvasive diagnostic devices in accordance with manufacturer's
D. Open and maintain an airway by positioning the patient's head; E. Provide external cardiopulmonary resuscitation and obstructed airway care for infants,
children, and adults;
F. Provide care for musculoskeletal injuries; G. Assist with prehospital childbirth;
H. Complete a clear and accurate prehospital emergency care report form on all patient
contacts and provide a copy of that report to the senior emergency medical services provider with the transporting ambulance;
I. Administer medical oxygen; J. Maintain an open airway through the use of:
1. A nasopharyngeal airway device; 2. A noncuffed oropharyngeal airway device; 3. A pharyngeal suctioning device;
K. Operate a bag mask ventilation device with reservoir; L. Provide care for suspected medical emergencies, including administering liquid oral
glucose for hypoglycemia;
M. Prepare and administer aspirin by mouth for suspected myocardial infarction (MI) in
patients with no known history of allergy to aspirin or recent gastrointestinal bleed;
N. Prepare and administer epinephrine by automatic injection device for anaphylaxis; O. Prepare and administer naloxone via intranasal device or auto-injector for suspected
opioid overdose; and
P. Perform cardiac defibrillation with an automatic or semi-automatic defibrillator, only
when the Emergency Medical Responder: 1. Has successfully completed an Authority-approved course of instruction in the use
of the automatic or semi-automatic defibrillator; and
2. Complies with the periodic requalification requirements for automatic or semi-
automatic defibrillator as established by the Authority.
Marion & Polk County Regional Treatment Protocols
Medical Control 1 of 7
SCOPE OF PRACTICE (OAR 847-035)
EMERGENCY MEDICAL TECHNICIAN
An Emergency Medical Technician (EMT) may:
A. Perform all procedures that an Emergency Medical Responder may perform; B. Ventilate with a non-invasive positive pressure delivery device; C. Insert a cuffed pharyngeal airway device in the practice of airway maintenance. A cuffed
pharyngeal airway device is: 1. A single lumen airway device designed for blind insertion into the esophagus
providing airway protection where the cuffed tube prevents gastric contents from entering the pharyngeal space; or
2. A multi-lumen airway device designed to function either as the single lumen device
when placed in the esophagus, or by insertion into the trachea where the distal cuff
creates an endotracheal seal around the ventilatory tube preventing aspiration of gastric contents.
D. Perform tracheobronchial tube suctioning on the endotracheal intubated patient; E. Provide care for suspected shock; F. Provide care for suspected medical emergencies, including:
1. Obtain a capillary blood specimen for blood glucose monitoring; 2. Prepare and administer epinephrine by subcutaneous injection, intramuscular
injection, or automatic injection device for anaphylaxis;
3. Administer activated charcoal for poisonings; and 4. Prepare and administer nebulized Albuterol sulfate treatments for known asthmatic
and chronic obstructive pulmonary disease (COPD) patients suffering from suspected bronchospasm.
G. Perform cardiac defibrillation with an automatic or semi-automatic defibrillator; H. Transport stable patients with saline locks, heparin locks, foley catheters, or in-dwelling
vascular devices;
I. Assist the on-scene Advanced EMT, EMT-Intermediate, or Paramedic by:
1. Assembling and priming IV fluid administration sets; and 2. Opening, assembling and uncapping preloaded medication syringes and vials;
J. Perform other emergency tasks as requested if under the direct visual supervision of a
physician and then only under the order of that physician;
K. Complete a clear and accurate prehospital emergency care report form on all patient
L. Assist a patient with administration of sublingual nitroglycerine tablets or spray and with
metered dose inhalers that have been previously prescribed by that patient's personal physician and that are in the possession of the patient at the time the EMT is summoned to assist that patient;
M. In the event of a release of organophosphate agents, the EMT who has completed
Authority-approved training may prepare and administer atropine sulfate and pralidoxime chloride by autoinjector, using protocols approved by the Authority and adopted by the supervising physician; and
N. In the event of a declared Mass Casualty Incident (MCI) as defined in the local Mass
Casualty Incident plan, monitor patients who have isotonic intravenous fluids flowing
Marion & Polk County Regional Treatment Protocols
Medical Control 2 of 7
SCOPE OF PRACTICE (OAR 847-035)
ADVANCED EMERGENCY MEDICAL TECHNICIAN (AEMT)
An Advanced Emergency Medical Technician (AEMT) may:
A. Perform all procedures that an EMT may perform; B. Initiate and maintain peripheral intravenous (I.V.) lines; C. Initiate saline or similar locks; D. Obtain peripheral venous blood specimens; E. Initiate and maintain an intraosseous infusion in the pediatric patient; F. Perform tracheobronchial suctioning of an already intubated patient; and G. Prepare and administer the following medications under specific written protocols
authorized by the supervising physician or direct orders from a licensed physician: 1. Physiologic isotonic crystalloid solution;
2. Anaphylaxis: epinephrine; 3. Antihypoglycemics:
a. Hypertonic glucose; b. Glucagon;
H. Vasodilators: nitroglycerine; I. Nebulized bronchodilators:
1. Albuterol; 2. Ipratropium bromide;
J. Analgesics for acute pain: nitrous oxide.
Marion & Polk County Regional Treatment Protocols
Medical Control 3 of 7
SCOPE OF PRACTICE (OAR 847-035)
EMT-INTERMEDIATE
An EMT-Intermediate may:
A. Perform all procedures that an Advanced EMT may perform; B. Initiate and maintain an intraosseous infusion; C. Prepare and administer the following medications under specific written protocols
authorized by the supervising physician, or direct orders from a licensed physician: 1. Vasoconstrictors:
a. Epinephrine; b. Vasopressin;
2. Antiarrhythmics:
a. Atropine sulfate;
b. Lidocaine; c. Amiodarone;
3. Analgesics for acute pain:
a. Morphine; b. Nalbuphine Hydrochloride; c. Ketorolac tromethamine; d. Fentanyl;
4. Antihistamine: Diphenhydramine; 5. Diuretic: Furosemide; 6. Intraosseous infusion anesthetic: Lidocaine; 7. Anti-Emetic: Ondansetron;
D. Prepare and administer immunizations in the event of an outbreak or epidemic as
declared by the Governor of the state of Oregon, the State Public Health Officer or a county health officer, as part of an emergency immunization program, under the agency's supervising physician's standing order;
E. Prepare and administer immunizations for seasonal and pandemic influenza vaccinations
according to the CDC Advisory Committee on Immunization Practices (ACIP), and/or the Oregon State Public Health Officer's recommended immunization guidelines as directed by the agency's supervising physician's standing order;
F. Distribute medications at the direction of the Oregon State Public Health Officer as a
component of a mass distribution effort;
G. Prepare and administer routine or emergency immunizations and tuberculosis skin
testing, as part of an EMS Agency's occupational health program, to the EMT-Intermediate's EMS agency personnel, under the supervising physician's standing order;
H. Insert an orogastric tube; I. Maintain during transport any intravenous medication infusions or other procedures
which were initiated in a medical facility, if clear and understandable written and verbal instructions for such maintenance have been provided by the physician, nurse practitioner or physician assistant at the sending medical facility;
J. Perform electrocardiographic rhythm interpretation; and K. Perform cardiac defibrillation with a manual defibrillator.
Marion & Polk County Regional Treatment Protocols
Medical Control 4 of 7
SCOPE OF PRACTICE (OAR 847-035)
PARAMEDIC
A Paramedic may:
A. Perform all procedures that an EMT-Intermediate may perform; B. Initiate the following airway management techniques:
1. Endotracheal intubation; 2. Cricothyrotomy; and 3. Transtracheal jet insufflation which may be used when no other mechanism is
available for establishing an airway;
C. Initiate a nasogastric tube; D. Provide advanced life support in the resuscitation of patients in cardiac arrest; E. Perform emergency cardioversion in the compromised patient;
F. Attempt external transcutaneous pacing of bradycardia that is causing hemodynamic
G. Perform electrocardiographic interpretation; H. Initiate needle thoracostomy for tension pneumothorax in a prehospital setting; I. Obtain peripheral arterial blood specimens under specific written protocols authorized
by the supervising physician;
J. Access indwelling catheters and implanted central IV ports for fluid and medication
K. Initiate placement of a urinary catheter for trauma patients in a prehospital setting who
have received diuretics and where the transport time is greater than thirty minutes; and
L. Prepare and initiate or administer any medications or blood products under specific
written protocols authorized by the supervising physician, or direct orders from a licensed physician.
Marion & Polk County Regional Treatment Protocols
Medical Control 5 of 7
MEDICAL CONTROL – Medications & Procedures
The following drugs and procedures are considered
CATEGORY A, and will be used at the EMT's
discretion in accordance with these EMS Treatment Protocols.
Drugs – Category A:
Activated Charcoal (aspirin or acetaminophen < 2 hrs post ingestion)
Atropine Sulfate
Calcium Gluconate (cardiac arrest & hyperkalemia)
Dexamethasone (Decadron)
Dextrose 50%, IV
Diltiazem (Cardizem)
Diphenhydramine
Glucose, Oral
Hydrocobalamin (Cyanokit®)
IV solutions
Ipratroprium
Magnesium Sulfate (cardiac arrest, eclampsia, adult asthma)
Nitroglycerin
Proparacaine
Sodium Bicarbonate
Sodium Thiosulfate
Succinylcholine
Marion & Polk County Regional Treatment Protocols
Medical Control 6 of 7
MEDICAL CONTROL – Medications & Procedures
Procedures – Category A
Chemical patient restraint
End-tidal CO2 monitoring
Use of a cuffed pharyngeal airway (King, IGEL or LMA)
Endotracheal intubation
Intraosseous infusion
Cricothyrotomy (surgical, needle)
Paralytic Intubation
Physical patient restraint
Self-Care instructions
Cardioversion
Taser barb removal other than face, neck or groin.
Chest Decompression
Transcutaneous pacing
The following drugs and procedures are considered
CATEGORY B, and require On-line Medical
Control authorization. Confirmation of dosage or procedure will be obtained directly from a Physician
on duty at OLMC.
Drugs – Category B:
Glucagon (beta blocker OD)
Magnesium Sulfate (pediatric asthma)
Sodium Bicarbonate (hyperkalemia, tri-cyclic antidepressant overdose, crush injury)
High-dose Albuterol (hyperkalemia)
Procedures – Category B:
Automatic Implantable Cardio-Defibrillator (AICD) deactivation
Marion & Polk County Regional Treatment Protocols
Medical Control 7 of 7
TABLE OF CONTENTS
SCOPE OF PRACTICE
MEDICAL CONTROL OF MEDICATIONS AND PROCEDURES
TREATMENT
ABDOMINAL PAIN . 1
ALTERED MENTAL STATUS . 2
ANAPHYLAXIS . 3-4
CARDIAC ARREST – AED / CPR . 7
CARDIAC ARREST – ASYSTOLE . 8
CARDIAC ARREST – PEA . 9
CARDIAC ARREST – V-FIB / PULSELESS VT . 10-12
CARDIAC CHEST PAIN / ACS . 13-14
CARDIAC DYSRHYTHMIAS – BRADYCARDIA . 15-16
CARDIAC DYSRHYTHMIAS – PVCS . 17
CARDIAC DYSRHYTHMIAS – TACHYCARDIA . 18-20
CHILDBIRTH / OB-GYN EMERGENCIES . 21-22
CRUSH INJURY . 23
EYE EMERGENCIES . 24
HYPERKALEMIA . 25
HYPERTHERMIA . 26
MUSCULOSKELETAL INJURIES – EXTREMITY/PELVIC TRAUMA . 28
MUSCULOSKELETAL INJURIES – SPINAL INJURY…………………………………….…29-32
NAUSEA AND VOMITING . 33
NEONATAL RESUCITATION . 34-35
POISONING AND OVERDOSE . 36-37
RESPIRATORY DISTRESS . 38-39
Marion & Polk County Regional Treatment Protocols
Table of Contents - Page 1 of 5
TABLE OF CONTENTS
PROCEDURES
AICD DEACTIVATION . 1
AIRWAY MANAGEMENT – GENERAL APPROACH . 2
AIRWAY MANAGEMENT – COMBITUBE® . 3
AIRWAY MANAGEMENT – NEEDLE/SURGICAL CRICOTHYROTOMY . 4-5
AIRWAY MANAGEMENT – PERTRACH® . 6
AIRWAY MANAGEMENT – QUICKTRACH® . 7
AIRWAY MANAGEMENT – END-TIDAL CO2 MONITORING . 8
AIRWAY MANAGEMENT – ENDOTRACHEAL INTUBATION . 9
AIRWAY MANAGEMENT – ENDOTRACHEAL INTUBATION WITH RSI . 10-11
AIRWAY MANAGEMENT – KING® AIRWAY . 12-13
CARBON MONOXIDE EXPOSURE . 14-15
CHEST DECOMPRESSION – NEEDLE . 16
CONTINUOUS POSITIVE AIRWAY PRESSURE . 17
INDUCED HYPOTHERMIA………………………………………………………………….18 INTRAOSSEOUS INFUSION . 19-21
INTRAVENOUS ACCESS / INFUSION . 22
IT CLAMP . 23-24
LEFT VENTRICULAR ASSIST DEVICE . 25-27
NASOGASTRIC TUBE PLACEMENT . 28
PACING – CARDIAC . 29
PELVIC SLING . 30-31
PICC LINE ACCESS . 32-33
RESTRAINT OF PATIENTS . 34
TAZER® BARB REMOVAL . 36
Marion & Polk County Regional Treatment Protocols
Table of Contents - Page 2 of 5
TABLE OF CONTENTS
MEDICATIONS
ACETAMINOPHEN (TYLENOL) . 1
ACTIVATED CHARCOAL . 2
ADENOSINE (ADENOCARD®) . 3
ALBUTEROL (VENTOLIN®) . 4
AMIODARONE (CORDARONE®) . 5
ATROPINE SULFATE . 7
CALCIUM GLUCONATE . 8
DEXAMETHASONE (DECADRON®)…. ………. ………………………………………. 10
DEXTROSE 50% . 11
DIPHENHYDRAMINE (BENADRYL®) . 13
DOPAMINE (INTROPIN®) . 14
EPINEPHRINE (ADRENALIN®) . 15
ETOMIDATE (AMIDATE®) . 16
FENTANYL (SUBLIMAZE®) . 17
GLUCOSE – ORAL (GLUTOSE 15®) . 19
HEMCON (CHITOSAN) . 21
HYDROXOCOBALAMIN (CYANOKIT®) . 22
IPRATROPIUM BROMIDE (ATROVENT®) . 23
LIDOCAINE (XYLOCAINE®) . 25
MAGNESIUM SULFATE . 26
MIDAZOLAM (VERSED®) . 27
MORPHINE SULFATE . 28
NALOXONE (NARCAN®) . 29
Marion & Polk County Regional Treatment Protocols
Table of Contents - Page 3 of 5
TABLE OF CONTENTS
NITROGLYCERIN . 30
ONDANSETRON (ZOFRAN®) . 31
PRALIDOXIME (PROTOPAM® / 2PAM®). 33
PROPARACAINE (ALCAINE®) . 34
SODIUM BICARBONATE . 36
SODIUM THIOSULFATE 25%. 37
SUCCINYLCHOLINE (ANECTINE®) . 38
VASOPRESSIN . 39
OPERATIONS
GENERAL PATIENT CARE GUIDELINES…………………………………………………….1
ADVANCED DIRECTIVES / DNR ORDERS . 2-3
CRIME SCENE RESPONSE. 4
DEATH IN THE FIELD . 5-6
DOCUMENTATION. 7
GRIEVING PEOPLE . 8-9
HAZARDOUS MATERIALS . 10-12
HOSPICE PATIENT RESPONSE……. ……………………………………………………….13
MEDICAL CONTROL OF SCENE. 14-15
ON-LINE MEDICAL CONTROL . 16-17
REFUSAL AND INFORMED CONSENT . 18-20
REHABILITATION . 21-22
REPORTING OF SUSPECTED CHILD ABUSE . 23
REPORTING OF SUSPECTED ELDER ABUSE . 24
TRAUMA SYSTEM . 25-27
Marion & Polk County Regional Treatment Protocols
Table of Contents - Page 4 of 5
TABLE OF CONTENTS
MULTIPLE PATIENT SCENE / MCI / MULTIPLE TOXIC EXPOSURE
DECONTAMINATION . 1-2
HYDROGEN CYANIDE . 3-5
HYDROGEN FLUORIDE . 6-8
ORGANOPHOSPHATES . 9-11
Marion & Polk County Regional Treatment Protocols
Table of Contents - Page 5 of 5
TREATMENT
Marion & Polk County Regional Treatment Protocols
ABDOMINAL PAIN
TREATMENT:
A. Start Oxygen per General Airway Management protocol. B. Monitor vital signs frequently. C. Place patient in a position of comfort. D. Establish venous access. E. If systolic blood pressure is less than 90 mmHg systolic follow Shock Protocol and initiate rapid
transport. If traumatic injury is suspected, enter patient into Trauma System. If patient has a suspected abdominal aortic aneurysm, titrate IV/IO to maintain systolic blood pressure of 90 mmHg. Consider 2nd IV/IO.
F. Attach cardiac monitor.
G. For pain control, give Fentanyl or Morphine Sulfate. (Fentanyl is preferred; use Morphine Sulfate
if Fentanyl is unavailable or contraindications exist.) Fentanyl dose 50 mcg IV/IO/IM/IN. Repeat with 25-50 mcg every 3-5 minutes as needed to a maximum of 400 mcg as long as BP remains more than 100 mmHg systolic and there are no other contraindications. Contact OLMC if more than 400 mcg is needed for pain control. Morphine Sulfate may be used in place of Fentanyl. 2-5 mg IV/IO/IM to max of 20mg. Fentanyl and Morphine shall not be used in conjunction with each other.
PEDIATRIC PATIENTS:
A. Consider non-accidental trauma. B. Closely monitor vital sign as blood pressure may drop quickly. C. For pain control, give Fentanyl or Morphine Sulfate. (Fentanyl is preferred; use Morphine Sulfate
if Fentanyl is unavailable or contraindications exist.) Fentanyl dose 1 mcg/kg IV/IO/IM/IN. Repeat with 0.5-1.0 mcg/kg every 3-5 minutes as needed to a maximum of 4 mcg/kg. Do not exceed adult dosing. Morphine Sulfate may be used in place of Fentanyl. 0.1 mg/kg IV/IO/IM to max of 20mg. Fentanyl and Morphine shall not be used in conjunction with each other.
NOTES & PRECAUTIONS:
A. Abdominal pain may be the first sign of catastrophic internal bleeding (ruptured aneurysm, liver,
spleen, ectopic pregnancy, perforated viscous, etc).
B. Since the bleeding is not apparent you must think of volume depletion and monitor the patient
closely for signs of shock.
KEY CONSIDERATIONS:
* Inferior MI
* Ectopic pregnancy
* Abdominal aortic aneurysm
* Perforated viscous
* Emesis type and amount
* Bowel movements
* Urinary output
* Ruptured spleen or liver * GI bleed
Marion & Polk County Regional Treatment Protocols
Treatment - Page 1 of 42
ALTERED MENTAL STATUS
TREATMENT:
A. Start Oxygen per General Airway Management protocol.
B. Monitor vital signs and oxygen saturation.
Agitated or Violent Patient
1. Ensure responder safety 2. Establish patient rapport, engage family member support if appropriate.
1. See Restraint of Patient Procedure
Hypoglycemia
1. Determine blood glucose level. If less than 60 mg/dl:
a. If patient can protect their own airway give oral glucose or D50.
b. If patient is unable to protect their own airway, establish venous access and give 25 grams
(50cc) of D50/
D10 IV drip
2. Repeat blood glucose level after 10 minutes and repeat treatment if it remains low. 3. If no IV/IO can be established, give Glucagon 1 mg IM.
Opiate Overdose
1. If opiate intoxication is suspected, administer Naloxone 0.5 mg IV/IO/IN/IM. Dose may be
repeated every two minutes up to 2 mg titrating to respiratory rate. If no improvement and opiate intoxication is still suspected, repeat Naloxone 2 mg every 3-5 minutes up to a maximum of 8 mg total.
C. Attach cardiac monitor. Treat dysrhythmias per appropriate protocol. Consider 12 Lead ECG. D. If other specific cause of altered mental status is known (i.e. seizure, stroke, poisoning) follow appropriate
E. If patient is combative consider sedation with Haldol and/or Versed per Restraint of Patient protocol.
PEDIATRIC PATIENTS:
A. Consider etiology and treat per appropriate protocol (Shock, Toxic Exposure, Seizure, Poisoning and
Overdose, etc.).
B. If suspected hypoglycemia, determine capillary blood glucose level.
Patients less than 10 kg (birth to 1 year) with CBG less than 40 mg/dl:
1. Give oral glucose if patient can protect their own airway. 2. If patient is unable to protect their own airway, establish venous access and give 0.5 grams/kg
(2 cc/kg) of
Dextrose 25%. May repeat once.
Patients 10 kg – 35kg (1 year to adolescence) with CBG less than 60 mg/dl:
1. Give oral glucose if patient can protect their own airway. 2. If patient is unable to protect their own airway, establish venous access and give 0.5 grams/kg
(1 cc/kg) of Dextrose 50%. May repeat once.
C. If no IV/IO access and patient is unable to protect their own airway, administer Glucagon 0.02 mg/kg IM
to a maximum of 1 mg.
D. If suspected opiate overdose, administer Naloxone 0.1 mg/kg IV/IOIM/IN to a maximum of 2 mg. If no
improvement and opiate intoxication is still suspected, may repeat every 3-5 minutes up to 2 mg/ dose to a maximum of 8 mg.
NOTES & PRECAUTIONS:
A. If patient is disoriented, think of medical causes. B. If patient is suicidal do not leave alone.
C. All patients in restraints must be monitored closely including pulse oximeter.
HISTORY & PHYSICAL FINDINGS:
* Hot/cold emergencies
* Recent emotional crisis
* Medic Alert tags
* Previous Psych disorder
* ETOH or drug ingestion
* Suicidal ideas
* Medication reactions
* Recent head trauma
* Possible poisoning
Marion & Polk County Regional Treatment Protocols
Treatment - Page 2 of 42
ANAPHYLAXIS
TREATMENT:
A. Start Oxygen per General Airway Management protocol. B. Monitor vital signs, ECG and oxygen saturation. C. Establish venous access.
D. If signs of significant respiratory distress are present administer 1:1,000 Epinephrine 0.3 mg SQ or IM.
The
IM administration route is preferred. May repeat once.
E. If patient exhibits signs of progressive anaphylaxis and/or significant respiratory distress:
1. With systolic blood pressure greater than 90 mmHg, administer 1:1,000 Epinephrine 0.3 mg (0.3
2. With systolic blood pressure less than 90 mmHg administer:
a) 1:1,000 Epinephrine 0.3 mg (0.3 cc) SQ or IM
OR b) 1:10,000 Epinephrine 0.3 mg (3 cc) IV/IO
c) Treat with fluid challenge per Shock Protocol
3. If no improvement noted repeat epinephrine every 5 minutes. 4. Adult EPI Pen may be used per manufacturer's recommendations.
F. If patient continues to exhibit signs of respiratory distress, administer Dexamethasone 10 mg IV/IO slowly
over 1-2 minutes.
G. Consider Diphenhydramine 1 mg/kg to a maximum of 50 mg IM or IV/IO. H. Consider Albuterol 2.5 mg via nebulizer.
I. If unable to secure a protected airway or unable to ventilate with BVM after epinephrine has been
administered cricothyrotomy may be required.
PEDIATRIC PATIENTS:
A. If signs of significant respiratory distress are present administer 1:1,000 epinephrine 0.01 mg/kg SQ to a
maximum dose of 0.3 mg (0.3 cc). May repeat once in 20 minutes.
B. If patient exhibits signs of progressive anaphylaxis and/or significant respiratory distress:
1. With normal perfusion, administer 1:1,000 Epinephrine 0.01 mg/kg SQ to a maximum dose of 0.3
2. With diminished perfusion administer:
a) 1:1,000 Epinephrine 0.01 mg/kg SQ/IM to a maximum of 0.3 mg (0.3 cc)
OR
b) 1:10,000 Epinephrine 0.01mg/kg IV/IO to a maximum of 0.1 mg (1.0 cc) c) Treat with fluid challenge per Shock Protocol.
3. If no improvement noted repeat Epinephrine every 5 minutes.
4. Pediatric EPI Pen may be used per manufacturer's recommendations.
C. If patient continues to exhibit signs of respiratory distress, administer Dexamethasone 0.6 mg/kg IV/IO
slowly over 1-2 minutes. Do not exceed adult dose.
D. Consider Diphenhydramine 1 mg/kg to a maximum of 50 mg IM or IV/IO. E. Consider Albuterol 2.5 mg via nebulizer.
F. If unable to secure patients airway or unable to ventilate with BVM after epinephrine has been
administered cricothyrotomy may be required.
NOTES & PRECAUTIONS:
A. Allergic reactions, even systemic in nature, are not necessarily anaphylaxis. Treatment may not be
indicated if only hives and itching are present.
B. Epinephrine increases cardiac work load and may cause angina or AMI in some individuals. C. Common side effects of Epinephrine include anxiety, tremor, palpitations, tachycardia and headache
particularly with IV administration.
D. Epinephrine should not be given unless signs of cardiovascular collapse or respiratory distress are present.
KEY CONSIDERATIONS:
* Toxic exposure
* Recent exposure to allergen
* Dyspnea or hives
* Abdominal cramps
* Known allergens
* Chest or throat tightness
* Swelling, numbness
Marion & Polk County Regional Treatment Protocols
Treatment - Page 3 of 42
TREATMENT:
A. Start Oxygen per General Airway Management protocol. B. Monitor vital signs, ECG and oxygen saturation. C. Establish venous access. If systolic BP less than 90 mmHg follow Shock Protocol. D. Remove jewelry and clothing that is smoldering or which is non-adherent to the patient. E. Cool burned areas (less than 10 minutes for large burns) then cover with sterile dressing. Discontinue
cooling if patient begins to shiver. Attempt to leave unbroken blisters intact.
F. Apply Carbon Monoxide (e.g. Rad-57) monitor if available.
G. For pain control, administer Fentanyl or Morphine. Fentanyl dose 50 mcg IV/IO/IM/IN. Repeat with 25-
50 mcg every 3-5 minutes as needed to a maximum of 400 mcg as long as BP remains greater than 100 mmHg systolic. Contact OLMC if more than 400 mcg are needed for pain control. Morphine Sulfate may
be used in place of Fentanyl. 2-5 mg IV/IO/IM to max of 20mg. Fentanyl and Morphine shall not be used in conjunction with each other.
H. If the patient has the following, transport to the Burn Center:
1. Total burn that is 15% or more of body surface area. 2. Full thickness burn greater than 5% of body surface area.
3. Burns with inhalation injuries. 4. Electrical burns 5. Facial burns or burns to hands, feet, genitalia or circumferential burns. 6. Burns in high risk patients (pediatrics, elderly, significant cardiac or respiratory problems) 7. Trauma system patients with burns meeting the above criteria.
I. If chemical burn:
1. Identify chemical if possible. 2. Consider Haz-Mat response. 3. Protect yourself from contamination. (See Decontamination protocol) 4. Flush contaminated areas with copious amounts of water. 5. If chemical is dry, carefully brush off prior to flushing.
J. If electrical burn:
1. Apply sterile dressings to entry and exit wounds. 2. Treat any dysrhythmias per appropriate Cardiac Dysrhythmia protocol.
K. If Cyanide Toxicity is suspected based on findings (soot in mouth, nose or oropharynx) and patient is
comatose, in cardiac or respiratory arrest, or has persistent hypotension despite fluid resuscitation:
1. Administer Hydroxocobalamin (Cyanokit®) 5 g IV/IO as an infusion and monitor for clinical
response. Contact OLMC for advice regarding a second 5 g dose.
2. If Hydroxocobalamin (Cyanokit®) is not available, then administer Sodium Thiosulfate 50 mL of
25% solution over 10-20 minutes. Do NOT administer Hydroxocobalamin (Cyanokit®) and Sodium Thiosulfate to the same patient.
3. Treat other presenting symptoms per appropriate protocol. 4. Initiate emergent transport to appropriate facility.
Marion & Polk County Regional Treatment Protocols
Treatment - Page 4 of 42
PEDIATRIC PATIENTS:
A. For pain control, administer Fentanyl – 1.0 mcg/kg IV/IO/ IM/IN. Repeat with 0.5-1 mcg/kg every 3-5
minutes as needed to a maximum of 4 mcg/kg. Do not exceed adult dosing. Morphine Sulfate may be used in place of Fentanyl. 0.1 mg/kg IV/IO/IM to max of 20mg. Fentanyl and Morphine shall not be used in conjunction with each other.
B. Consider possibility of non-accidental cause in children.
NOTES & PRECAUTIONS:
A. Remove rings or other constricting items immediately. B. Be prepared to use RSI early to control airway if necessary.
C. For firefighters, consider the potential for other traumatic injury or MI.
KEY CONSIDERATIONS:
* Enclosed space
* Possibility of inhaled toxins
* Past medical history
* CO/Cyanide poisoning
* Evidence of respiratory burns
* Extent of burns
* Explosion or trauma injuries
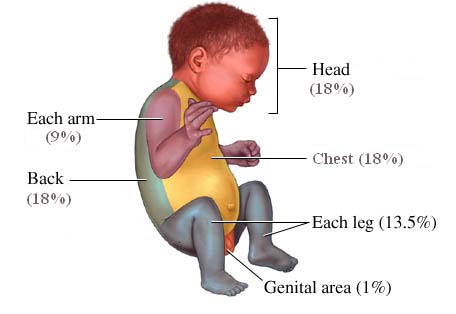
RULE OF NINES:
Marion & Polk County Regional Treatment Protocols
Treatment - Page 5 of 42
CARDIAC ARREST – AED/CPR
CPR GUIDELINES
Adolescent and older
1 yr. to adolescent
Under 1 year of age
Head tilt-chin lift. Jaw thrust if suspected cervical trauma.
Breathing: initial
2 breaths at 1 second per breath
10 to 12 breaths/min
12 to 20 breaths/min
One breath every 10 compressions
(approximately 8 to 10 breaths/min)
airway Foreign Body –
Abdominal thrusts (use chest thrusts in pregnant and
obese patients or if abdominal thrusts are not effective)
Lower half of sternum between nipples
Just below nipple line
(lower half of sternum)
Heel of one hand, other
Heel of one hand, as for
2-3 fingers or 2 thumb-
encircling hands
At least 2 inches
At least 1/3 AP diameter of chest.
2 inches Child, 1 ½ inches Infant
Compression rate
At least 100 per minute
30:2 (one or two
30:2 (single rescuer)
ventilation ratio
15:2 (two rescuers)
AED GUIDELINES
AED Defibrillation
Use adult pads, do not
Use pediatric system for
Use pediatric system if
children 1-8 yrs (less than
55 lbs) if available
NEONATAL GUIDELINES
Assisted ventilation should be delivered at a rate of 40-60 breaths/minute The ratio of compressions to ventilations should be 3:1, with 90 compressions and 30 breaths to achieve approximately 120 events per minute.
HIGH PERFORMANCE CPR
Consider two cycles of compressions without ventilations.
There will be no ventilation pauses. No pulse checks after shock. Wave form capnography can greatly reduce need for pulse checks and can accurately identify ROSC. Do NOT stop compressions during intubation attempts, other airway maneuvers, other procedures. Use the hover method during compressions to reduce time off the chest. During CPR – if possible – patient should not be moved/transported except if ROSC is obtained. Transport indicated if:
Unsafe or hostile scene
Public setting
Outdoor setting and inclement weather
Cardiac tamponade
Paramedic discretion
ALL PEDIATRIC CARDIAC ARRESTS ARE TO BE TREATED AS "LOAD AND GO" AND
TRANSPORTED IMMEDIATELY.
Marion & Polk County Regional Treatment Protocols
Treatment - Page 6 of 42
CARDIAC ARREST – Asystole
TREATMENT:
If down time is estimated at greater than 5 minutes, perform CPR for 2 minutes
If down time is less than 5 minutes, perform CPR until defibrillator is attached
Establish venous access
Secure protected airway with minimal interruptions to CPR.
Vasopressin 40 units IV/IO or 1mg of 1:10,000 Epinephrine IV/IO
1:10,000 Epinephrine 1 mg IV/IO, repeat every 3-5 minutes.
PEDIATRIC PATIENTS:
A. Begin CPR, aggressive airway management and intubation. B. Administer 1:10,000 Epinephrine 0.01 mg/kg IV/IO, repeat every 3-5 minutes.
NOTES & PRECAUTIONS:
A. If unwitnessed arrest, unknown downtime, and no obvious signs of death, proceed with resuscitation
and get further information from family/bystanders.
B. Contact OLMC for advice on continuing resuscitation. C. If history of traumatic event, consider Death in the Field protocol.
D. Use waveform Capnography to confirm and monitor ET placement and CPR effectiveness.
KEY CONSIDERATIONS:
Consider and treat other possible causes:
Acidosis - Sodium Bicarbonate 1 mEq/kg IV/IO.
Cardiac Tamponade – Initiate rapid transport. Hyperkalemia – Treat per Hyperkalemia protocol.
Hypothermia – Treat per Hypothermia protocol. Hypovolemia – Treat with fluids per Shock protocol.
Hypoxia – Oxygenate and ventilate. Pulmonary embolus – Initiate rapid transport. Tension pneumothorax – Needle decompression.
Tri-cyclic antidepressant overdose – Sodium Bicarbonate 1 mEq/kg IV/IO
Marion & Polk County Regional Treatment Protocols
Treatment - Page 7 of 42
CARDIAC ARREST – PEA
TREATMENT:
If down time is estimated at greater than 5 minutes, perform CPR for 2 minutes
If down time is less than 5 minutes, perform CPR until defibrillator is attached
Establish venous access
Secure protected airway with minimal interruptions to CPR.
Vasopressin 40 units IV/IO or 1:10,000 Epinephrine 1 mg IV/IO
1:10,000 Epinephrine 1 mg IV/IO, repeat every 3-5 minutes.
If end-tidal CO2 is more than or equal to 20 with an organized rhythm, initiate fluids per
Shock protocol and consider Dopamine administration 10 mcg/kg/min.
PEDIATRIC PATIENTS:
A. Begin CPR, aggressive airway management and intubation. B. Establish venous access. C. Administer 1:10,000 Epinephrine 0.01 mg/kg IV/IO, repeat every 3-5 minutes
D. Consider and treat other possible causes.
NOTES & PRECAUTIONS:
OLMC must be contacted prior to discontinuing resuscitation efforts.
Use waveform Capnography to confirm and monitor ET placement and CPR effectiveness.
KEY CONSIDERATIONS:
Consider and treat other possible causes:
Acidosis - Sodium Bicarbonate 1 mEq/kg IV/IO.
Cardiac Tamponade – Initiate rapid transport. Hyperkalemia – Treat per Hyperkalemia protocol.
Hypothermia – Treat per Hypothermia protocol. Hypovolemia – Treat with fluids per Shock protocol.
Hypoxia – Oxygenate and ventilate. Pulmonary embolus – Initiate rapid transport. Tension pneumothorax – Needle decompression.
Tri-cyclic antidepressant overdose – Sodium Bicarbonate 1 mEq/kg IV/IO
Marion & Polk County Regional Treatment Protocols
Treatment - Page 8 of 42
CARDIAC ARREST – V-Fib / Pulseless VT
TREATMENT:
Flow of algorithm presumes that the initial rhythm is continuing. If the rhythm changes, begin the appropriate
algorithm. Interruptions to CPR should be avoided. When necessary they should be less than 10 seconds.
Follow manufacturer's recommendations for defibrillation settings.
HEART MONITOR ADULT DEFIBRILLATION SETTINGS
Medtronics Lifepak – 200j, 300j, 360j then repeat at 360j as needed. Philips MRX – 150j all shocks.
Zoll E-Series – 120j, 150j, 200j then repeat at 200j as needed.
If down time is estimated at greater than 5 minutes, perform CPR for 2 minutes
If down time is less than 5 minutes, perform CPR until defibrillator is attached
Check monitor for rhythm – If V-Fib or pulseless VT
CPR until ready to defibrillate
Defibrillate x 1
Immediately continue CPR following defibrillation
Establish IV/IO access (do not stop CPR)
Check rhythm after two minutes of CPR
If V-Fib or pulseless VT persists continue CPR
Vasopressin 40 units IV/IO or 1:10,000 Epinephrine 1 mg IV/IO
Defibrillate x 1
Immediately continue CPR following defibrillation
Check rhythm after two minutes of CPR
If V-Fib or pulseless VT persists continue CPR
Amiodarone 300 mg IV/IO or Lidocaine 1.5 mg/kg IV/IO
Defibrillate x 1
Immediately continue CPR following defibrillation
Check rhythm after two minutes of CPR
If V-Fib or pulseless VT persists continue CPR
1:10,000 Epinephrine 1 mg IV/IO
Defibrillate x 1
Immediately continue CPR following defibrillation
Check rhythm after two minutes of CPR
If V-Fib or pulseless VT persists continue CPR
Amiodarone 150 mg IV/IO or Lidocaine 1.5 mg/kg IV/IO
Defibrillate x 1
Immediately continue CPR following defibrillation
Check rhythm after two minutes of CPR
Marion & Polk County Regional Treatment Protocols
Treatment - Page 9 of 42
CARDIAC ARREST – V-Fib / Pulseless VT
If V-Fib or pulseless VT persists continue CPR
1:10,000 Epinephrine 1 mg IV/IO
Defibrillate x 1
Immediately continue CPR following defibrillation
Check rhythm after two minutes of CPR
If V-Fib or pulseless VT persists continue CPR
Lidocaine 1.5 mg/kg IV/IO
Defibrillate x 1
Immediately continue CPR following defibrillation
Check rhythm after two minutes of CPR
If V-Fib or pulseless VT persists continue CPR
1:10,000 Epinephrine 1 mg IV/IO
Defibrillate x 1
Immediately continue CPR following defibrillation
Check rhythm after two minutes of CPR
If V-Fib or pulseless VT persists continue CPR
Lidocaine 1.5 mg/kg IV/IO
Defibrillate x 1
Immediately continue CPR following defibrillation
Check rhythm after two minutes of CPR
If V-Fib or pulseless VT persists continue CPR
1:10,000 Epinephrine 1 mg IV/IO
Defibrillate x 1
Immediately continue CPR following defibrillation
Check rhythm after two minutes of CPR
If V-Fib or pulseless VT persists continue CPR
Magnesium Sulfate 2 grams IV/IO
Defibrillate x 1
Immediately continue CPR following defibrillation
Check rhythm after two minutes of CPR
Note: The sequence of Amiodarone and Lidocaine may be reversed (i.e. Lido then Amio)
Marion & Polk County Regional Treatment Protocols
Treatment - Page 10 of 42
CARDIAC ARREST – V-Fib / Pulseless VT
PEDIATRIC PATIENTS:
Follow adult algorithm flow. Use the following dosing:
Defibrillation:
1. First shock 2j/kg. 2. Second shock, 4j/kg .
3. Subsequent shocks, more than or equal to 4 j/kg, up to maximum of 10 j/kg or
adult dose per shock as needed.
a) 1:10,000 – 0.01 mg/kg IV/IO
2. Amiodarone – 5 mg/kg IV/IO. May repeat twice, up to total dose of 15 mg/kg.
Max single dose is 300mg.
3. Lidocaine – Follow adult dosing. 4. Magnesium Sulfate – 25-50 mg/kg IV/IO (max 2 grams) over 10-20 min. NOTE: Vasopressin is not used in Pediatric algorithm.
NOTES & PRECAUTIONS:
A. Airway should be addressed with minimal interruption to CPR. Ventilation rate should be 8-10
breaths per minute with advanced airway. In place.
B. If the initial rhythm is Torsades de Pointes, give Magnesium Sulfate 2 grams IV/IO. C. After successful resuscitation:
a. With no antidysrhythmic – Give Lidocaine bolus 1.5 mg/kg and re-bolus with Lidocaine
0.75 mg/kg every ten minutes.
b. If Amiodarone was the last antidysrhythmic given – Re-dose after 30 minutes with
Amiodarone 150 mg over 10 minutes.
c. If Lidocaine or Magnesium was the last antidysrhythmic given – Give Lidocaine 0.75 mg/kg
every 10 minutes.
D. Be cautious with the administration of Lidocaine or Amiodarone if any of the following are present:
a. Systolic BP is less than 90 mmHg
b. Heart rate is less than 50 beats per minute c. Periods of sinus arrest d. Any AV block
E. Sodium Bicarbonate is not recommended for the routine cardiac arrest sequence, but should be
used early in cardiac arrest of known cyclic antidepressant overdose or in patients with
hyperkalemia. It may also be considered after prolonged arrest. If used, administer 1 mEq/kg. Subsequent doses per OLMC. Repeated doses at 0.5 mEq/kg every10 minutes.
F. Use waveform Capnography to confirm and monitor ET placement and CPR effectiveness.
Marion & Polk County Regional Treatment Protocols
Treatment - Page 11 of 42
CARDIAC CHEST PAIN / ACS
TREATMENT:
A. Start Oxygen per General Airway Management protocol.
B. Monitor vital signs, ECG and oxygen saturation.
C. If an acute ischemic event is suspected, obtain 12-lead ECG if available. This may be done
concurrently with other treatment and should not delay treatment or transport.
D. Establish vascular access. This should be done prior to nitroglycerin administration in
patients who have not taken nitroglycerin previously or who have a potential for hemodynamic instability.
E. Administer Aspirin 324 mg orally unless contraindicated or if patient has taken more than 1
gram in the last 24 hours.
F. If blood pressure is over 100 mmHg systolic, administer Nitroglycerin 0.4 mg sublingual.
Repeat every 5 minutes until chest pain is relieved as long as systolic BP remains over 100 mmHg.
G. For pain control, give Fentanyl or Morphine Sulfate. Fentanyl dose 50 mcg IV/IO/IM/IN.
Repeat with 25-50 mcg every 3-5 minutes as needed to a maximum of 400 mcg as long as BP remains over 100 mmHg systolic and no other contraindications. Contact OLMC if more than 400 mcg is needed for pain control. Morphine Sulfate may be used in place of Fentanyl: 2-5 mg IV/IO/IM to a max of 20mg. Fentanyl and Morphine shall not be used in conjunction with each other.
H. Treat any dysrhythmias per appropriate Cardiac Dysrhythmia protocol.
I. To relieve anxiety, consider Midazolam 1-2mg IV/IO.
PEDIATRIC PATIENTS:
A. Consider pleuritic causes or trauma.
B. Contact OLMC for advice.
NOTES & PRECAUTIONS:
A. DO NOT DELAY ADMINISTRATION OF ASPIRIN TO OBTAIN 12-LEAD ECG.
B. Nitroglycerin administration to patients with an acute inferior myocardial infarction should
be performed with close monitoring of vital signs and rhythm. Nitroglycerin in these patients may result in symptomatic hypotension and/or shock, which should be treated with fluids and positioning.
C. Do not administer nitroglycerin without OLMC if patient has taken ED medications within
the last 48 hours.
D. Do not administer aspirin in patients who have a true allergy to aspirin, who have a history
of an active bleeding disorder, GI bleed or ulcer, or who have a suspected aortic dissection.
KEY CONSIDERATIONS:
Pain evaluation (OPQRST)
Nausea and vomiting
Shortness of breath
Fever or recent illness
Other medical history
Medications and allergies
Peripheral edema
** Note for STEMI: time of diagnosis, time of notification to hospital, time of arrival at hospital
Marion & Polk County Regional Treatment Protocols
Treatment - Page 12 of 42
CARDIAC CHEST PAIN / ACS
FIELD-IDENTIFIED ST-ELEVATION MI (STEMI)
Indication: 12-lead ECG with:
Automatic ECG interpretation of "Acute MI Suspected" Paramedic interpretation of probable STEMI
Rapid transport to destination hospital ED with interventional capability. Early notification of destination and advise receiving hospital of "STEMI patient".
If available, transmit 12-lead ECG to destination hospital. Non-diagnostic ECGs with potential "imitators" of ACS or ECGs that are clinically
concerning should also be transmitted without STEMI activation. If transmission is
unavailable, describe ECG to receiving hospital or contact OLMC. These may include:
* Bundle Branch Block
* Left ventricular hypertrophy
* SVT with aberrancy
* Paced rhythms
* Pericarditis
* Benign early repolarization
* Digitalis effect
*** IF MONITOR SAYS STEMI OR IF PATIENT MEETS CRITERIA,
activate STEMI protocol. ***
STEMI FIELD NOTIFICATION/ACTIVATION ALGORITHIM
ST elevation/STEMI
identified on 12 lead EKG
12 Lead reveals:
More than 1 mm of ST elevation in 2 or more non-
precordial contiguous leads
More 2 mm of ST elevation in 2 or more precordial
contiguous leads
No activation
Presence or suspicion of:
Repeat EKG as
Actively working
Maintain supportive
Bundle branch block
Active cardiac arrest Patient refusal for PCI
Marion & Polk County Regional Treatment Protocols
Treatment - Page 13 of 42
CARDIAC DYSRHYTHMIAS – Bradycardia
HEART RATE LESS THAN 60 BPM AND INADEQUATE FOR CLINICAL CONDITION
Start Oxygen per General Airway Management protocol.
Monitor vital signs, ECG and oxygen saturation.
Establish venous access.
Are signs or symptoms of poor perfusion caused by the bradycardia present?
(Altered mental status, chest pain, hypotension or other signs of shock)
Yes – Pt Unstable
Prepare for pacing per Transcutaneous Pacing
patient. Obtain 12-lead
protocol. Use without delay for high-degree
heart blocks (2nd degree Type II, Third degree)
Consider Atropine 0.5 mg IV/IO while
awaiting pacer. May repeat every 3-5 minutes to a maximum of 3 mg.
Consider Dopamine 5 mcg/kg/min if pacing is
ineffective. Increase dose by 5mcg/kg/min to
max of 20 mcg/kg/min as needed.
If capture is achieved and patient is uncomfortable, consider Midazolam
2.0-5.0 mg IV/IO or 5 mg IM. May repeat IV/IO dose once.
If capture is not achieved, try repositioning pads. Goal of therapy is to improve perfusion and maintain a BP over 90 mmHg
NOTES & PRECAUTIONS:
A. Bradycardia may be protective in the setting of cardiac ischemia and should only be treated if
associated with serious signs and symptoms of hypoperfusion.
B. Most pediatric bradycardia is due to hypoxia. Oxygenate and ventilate aggressively.
C. Hyperkalemia may cause bradycardia. If the patient has a wide complex bradycardia with a
history of renal failure, muscular dystrophy, paraplegia, crush injury or serious burn more than 48 hours prior, consider treatment per Hyperkalemia protocol.
KEY CONSIDERATIONS:
Pain evaluation (OPQRST)
Nausea and vomiting
Fever or recent illness
Marion & Polk County Regional Treatment Protocols
Treatment - Page 14 of 42
CARDIAC DYSRHYTHMIAS – Bradycardia
PEDIATRIC PATIENTS:
BRADYCARDIA WITH A PULSE CAUSING CARDIORESPIRATORY COMPROMISE
Start Oxygen per General Airway Management protocol.
Monitor vital signs, ECG and oxygen saturation.
Is bradycardia still causing cardiorespiratory compromise?
No – pt. stable
Yes – pt. unstable
Continue to support
Start CPR if despite oxygenation and
ventilation patient's heart rate is less than
Monitor patient.
60 bpm with poor perfusion.
Consider OLMC
Persistent symptomatic bradycardia?
Give 1:10,000 Epinephrine 0.01 mg/kg IV/IO. Repeat epinephrine
every 3-5 minutes.
If increased vagal tone or AV block, consider Atropine 0.02 mg/kg
IV/IO. Minimum single dose 0.1 mg, maximum single dose 0.5 mg. May repeat once.
Consider pacing per Transcutaneous Pacing protocol.
If capture is achieved and patient is uncomfortable, consider
Midazolam 0.1mg/kg IV/IO to a maximum of 2.5 mg, or 0.2 mg/kg IM to a maximum of 5 mg.
If capture is not achieved, try repositioning pads. Goal of therapy is to improve perfusion.
Marion & Polk County Regional Treatment Protocols
Treatment - Page 15 of 42
CARDIAC DYSRHYTHMIAS – PVCs
TREATMENT:
A. Start Oxygen per General Airway Management protocol. B. Monitor vital signs, ECG and oxygen saturation. C. Establish venous access. D. Administer Lidocaine:
1. 1.5 mg/kg IV/IO over 1-2 minutes. 2. If no change, give 0.75 mg/kg every 10 minutes up to a maximum of 3 mg/kg. 3. When PVCs are suppressed, continue with Lidocaine 0.75 mg/kg every 10
4. All doses after the initial bolus must be reduced to one-quarter (0.375 mg/kg) of
the initial bolus in patients with congestive heart failure, shock, hepatic disease, or in patients over 70 years of age.
NOTES & PRECAUTIONS:
A. PVCs should be treated only in the setting of an acute ischemic event (i.e. chest pain,
couplets, R on T, runs of VT)
B. Lidocaine should not be used without OLMC direction if the following are present:
1. Systolic BP is less than 90 mmHg. 2. Heart rate is less than 50 beats per minute. 3. Periods of sinus arrest are present. 4. Second or Third degree heart block is present.
KEY CONSIDERATIONS:
Medical history
Shortness of breath
Does pulse match ECG?
Marion & Polk County Regional Treatment Protocols
Treatment - Page 16 of 42
CARDIAC DYSRHYTHMIAS – Tachycardia
Start Oxygen per General Airway Management protocol.
Monitor vital signs, ECG and oxygen saturation. Establish venous access.
Are signs or symptoms of poor perfusion caused by the dysrhythmia present?
(Altered mental status, chest pain, hypotension or other signs of shock)
Rate related symptoms uncommon if HR less than 150 bpm. Consider other causes.
No – Pt Stable. Obtain 12-lead ECG
Yes – Pt Unstable
cardioversion-Start
at 100 J. Establish
IV/IO access if not
If pt is conscious
consider sedation with Midazolam
IV/IO/IM/IN, may
Atrial flutter
Multifocal atrial
cardioversions x 3
Cardizem: 0.25 mg/kg
(repeat at 0.35 mg/kg if no response after 15 min)
If patient converts to a sinus
rhythm from a wide complex
Afib w/aberrancy
tachycardia, give Lidocaine 1.5
mg/kg IV/IO bolus. Repeat at 0.75 mg/kg every 10 minutes.
Magnesium Sulfate 4 grams
If patient does not convert
IV/IO over 10 minutes
Obtain post treatment 12-lead ECG
Contact OLMC for advice Consider contributing factors and other treatments
The sequence of Amiodarone and Lidocaine may be reversed (i.e. Lido then Amio)
Marion & Polk County Regional Treatment Protocols
Treatment - Page 17 of 42
CARDIAC DYSRHYTHMIAS – Tachycardia
PEDIATRIC PATIENTS:
Start Oxygen per General Airway Management protocol.
Monitor vital signs, ECG and oxygen saturation.
Are signs or symptoms of poor perfusion caused by the dysrhythmia present?
No – Pt Stable. Obtain 12-lead ECG
Yes – Pt Unstable
QRS (< 0.12 sec)
HR>220 child < 2
sec) HR > 150
HR>180 child 2-10 Probable SVT
Establish IV/IO
Ice water to face
If pt is conscious
children <6 y.o.
consider sedation
Valsalva in older
0.1 mg/kg IV/IO, or
0.2 mg/kg IM. Do
not exceed adult
Atrial flutter
cardioversion for
Multifocal atrial
If no response
Consider Cardizem;
cardioversion at 2
contact OLMC for pts
under 15 yrs old
If patient converts to a sinus
rhythm from a wide complex
Afib w/aberrancy
tachycardia, give Lidocaine 1.5
mg/kg IV/IO bolus. Repeat at 0.75 mg/kg every 10 minutes.
If Torsades, Magnesium Sulfate 25-50 mg/kg
If patient does not convert
IV/IO over 10 min
Obtain post treatment 12-lead ECG
Contact OLMC for advice
If patient is not symptomatic with a narrow regular QRS (less than 0.12 sec) and has a HR less than 220 (child less than
2) or HR less than 180 (child 2-10) consider Sinus Tachycardia and treat possible causes (see Notes & Precautions
below). The sequence of Amiodarone and Lidocaine may be reversed (i.e. Lido then Amio).
Marion & Polk County Regional Treatment Protocols
Treatment - Page 18 of 42
CARDIAC DYSRHYTHMIAS – Tachycardia
HEART MONITOR ADULT SYNCHRONOUS CARDIOVERSION SETTINGS
Medtronics Lifepak – 100j, 200j, 300j, 360j Philips MRX – 100j, 120J, 150J, 150J Zoll E-Series – 70j, 120j, 150j, 200j
NOTES & PRECAUTIONS:
A. If the patient is asymptomatic, tachycardia may not require treatment in the field. Continue to
monitor the patient for changes during transport.
B. Other possible causes of tachycardia include:
a. Acidosis b. Hypovolemia
c. Hyperthermia/fever d. Hypoxia e. Hypo/Hyperkalemia
f. Infection g. Pulmonary embolus
h. Tamponade i. Toxic exposure j. Tension pneumothorax
C. All Lidocaine doses after the initial bolus must be reduced to 0.375 mg/kg (1/4 of the initial
dose) in patients with CHF, shock, hepatic disease, or in patients greater than 70 y/o.
D. If pulseless arrest develops, follow Cardiac Arrest protocol. E. Use waveform capnography.
KEY CONSIDERATIONS:
* Medical history
* Shortness of breath
* Angina or chest pain
* Speed of onset
Marion & Polk County Regional Treatment Protocols
Treatment - Page 19 of 42
CHILDBIRTH/OB GYN EMERGENCIES
TREATMENT:
A. Start Oxygen per General Airway Management protocol. B. Monitor vital signs, ECG and oxygen saturation.
C. Establish venous access on mother as time allows.
D. Normal Childbirth
1. Guide/control but do not hurry or retard delivery.
2. Check for cord around neck and remove if present.
3. Suction only if obvious signs of obstruction to spontaneous breathing or requiring
Positive Pressure Ventilation.
4. In infants not requiring resuscitation: wait 1 minute from delivery, then clamp cord at 4
and 6 inches and cut between clamps. Dry infant keeping them level with mother's heart
until cord is cut.
5. If child does not need treatment, place on mother's chest and cover to maintain
6. Assess APGAR at one and five minutes after birth.
7. If infant needs resuscitation, follow Neonatal Resuscitation protocol. 8. Massage uterus to encourage contraction and prevent bleeding. Do not delay transport
to deliver the placenta.
9. If placenta delivers on scene, place in bio bag and transport it with patient.
E. Arm or Leg Presentation
1. Place mother in knee-chest position and transport immediately to nearest hospital.
2. Call OLMC early.
F. Breech Presentation (Buttocks first)
1. If delivery is imminent, prepare the mother as usual and allow the buttocks and trunk to
deliver spontaneously.
2. Support the body while the head delivers.
3. If the head does not deliver within three minutes suffocation can occur. Place a gloved
hand into the vagina to keep the vaginal wall away from the baby's face. Inserting a
finger into the infant's mouth and gently tilting the head toward the chest can sometimes
4. If child does not deliver, transport mother in knee-chest position.
G. Prolapsed Cord
1. Place mother in knee-chest position or with hips elevated on pillows.
2. With a gloved hand gently attempt to push the baby back up the vagina several inches.
Do not attempt to push the cord up.
3. Transport immediately to the nearest hospital.
H. Toxemia of Pregnancy (Eclampsia)
1. Treat seizures per Seizure protocol.
2. Administer Magnesium Sulfate (normal dose is 4 grams IV/IO infused over 10 minutes).
I. Abrupto Placenta/Placenta Previa
1. Treat per Shock Protocol if necessary.
2. Transport immediately to the nearest hospital. 3. Contact OLMC early.
Marion & Polk County Regional Treatment Protocols
Treatment - Page 20 of 42
CHILDBIRTH/OB GYN EMERGENCIES
NOTES & PRECAUTIONS:
A. If thick meconium is present, and resuscitation is needed, follow Suctioning procedure.
Contact OLMC early for advice.
B. Always consider the possibility of ectopic pregnancy in a woman of child bearing age (13
– 55) with abdominal pain or vaginal bleeding.
C. If there is an imminent delivery, go directly to ED. Patients over 20 weeks may still be
transported to a hospital who is on divert if the hospital's Labor & Delivery department will take the patient directly. Contact the destination hospital.
D. Blood loss of up to 500 ccs is normal in vaginal deliveries.
KEY CONSIDERATIONS:
Due date/prenatal care
Last menstrual period
Previous childbirth history
Single or multiple birth
Fetal heart tones
Ruptured membranes
Vaginal bleeding
Edema or hypertension
APGAR SCORE:
Body pink, blue extremities
Slow (less than 100 bpm)
Marion & Polk County Regional Treatment Protocols
Treatment - Page 21 of 42
CRUSH INJURY / ENTRAPMENT
TREATMENT:
A. Start Oxygen per General Airway Management protocol. B. Monitor vital signs, ECG and oxygen saturation.
C. Establish venous access.
D. Protect patient from environment (rain, snow, direct sun, etc). If applicable, begin warming
methods to prevent hypothermia (warm blankets, heated air with blower, warm IV fluids).
E. Plan extrication activities to allow for periodic patient assessment. Plan for occasional
extrication equipment "shut down" to assess vital signs.
F. Carefully track vitals, IV/IO fluids, and medications during extrication. G. Evaluate degree of entrapment and viability of extremities. (absent pulse, blanched skin,
capillary refill, diminished sensation, extremely cold to the touch) If one or more
extremities are trapped and circulation is compromised or absent consider the placement of
constricting bands to inhibit rapid venous return to the central circulatory system of potassium, lactic acid, and myoglobin upon extrication. Contact OLMC for direction.
H. If extrication of a limb will be prolonged, direct mechanical crush injuries are present
(tissue is crushed), and patient's condition is deteriorating, strongly consider calling OLMC to arrange on-scene amputation.
I. Carefully assess collateral injuries that may have occurred during event.
J. If patient is trapped in a heavy dust environment, consider methods to provide filtered
oxygen to the patient. If patient is in respiratory distress, consider dust impaction injuries and prepare to administer nebulized Albuterol per OLMC direction.
K. During extrication of a severely trapped patient who is at risk for crush syndrome,
administer 1000-2000 cc NS via IV/IO bolus, then maintain at 500 cc/hr.
L. Contact OLMC for consideration of Sodium Bicarbonate to buffer acid release from anaerobi
M. For pain control, administer Fentanyl or Morphine. Fentanyl dose 50 mcg IV/IO/IM/IN.
Repeat with 25-50 mcg every 3-5 minutes as needed to a maximum of 400 mcg as long as BP remains greater than 100 mmHg systolic. Contact OLMC if more than 400 mcg are
needed for pain control. Morphine Sulfate may be used in place of Fentanyl. 2-5 mg IV/IO/IM
to max of 20mg. Fentanyl and Morphine shall not be used in conjunction with each other.
NOTES & PRECAUTIONS:
A. Do not allow any personnel into extrication area (inner circle) without proper protective
equipment and thorough briefing to include evacuation signal.
B. Notify the receiving Trauma Center through OLMC early in the extrication process to
facilitate receiving advanced medical resources if needed.
C. Technical Rescue Team Leader should coordinate all extrication activities, especially the
release of patient, with Medical Branch Director.
D. Keep patient well-hydrated and warm during extrication efforts.
KEY CONSIDERATIONS:
Previous medical history
Current medications
Length and degree of entrapment
Use of technical rescue
Length of extrication
Alternate treatment plans
Marion & Polk County Regional Treatment Protocols
Treatment - Page 22 of 42
EYE EMERGENCIES
TREATMENT:
A. Start Oxygen per General Airway Management protocol.
B. Monitor vital signs. C. Establish venous access as needed. D. Treat specific injuries as follows:
1. Chemical Burns
a. Identify the chemical if possible. b. Remove corrective lenses. c. Give Proparacaine as outlined below. d. Irrigate with Normal Saline or other clean fluid for at least 30 minutes, from the center
of the eye towards the eyelid.
e. Do not attempt to neutralize acids or bases. f. Prevent tissues of the inner eyelid and globe from adhering to each other.
2. Rupture/Penetration of Globe
a. Protect the affected eye and its contents with a hard shield or "Dixie-cup" device and
cover the other eye.
b. Contact OLMC for sedation orders.
c. Consider Ondansetron per Nausea and Vomiting protocol to decrease patient risk of
3. Foreign body on outer eye
a. Remove corrective lenses. Do not wipe eye. b. Give Proparacaine as outlined below. c. Irrigate with Normal Saline or other clean fluid for at least 30 minutes, or until foreign
body is removed.
d. Use sterile Q-tip or gauze to invert lid to look for or remove foreign bodies. e. Cover eye with a soft patch.
PROPARACAINE ADMINISTRATION:
One drop in the affected eye for initial anesthetic effect. If effect is not felt within one minute, three
additional drops may be given at one-minute intervals. If no anesthetic effect is felt after the fourth
drop, consult OLMC. For transports longer than 15 minutes, if eye pain returns, 1-4 additional drops
may be given as previously done to continue anesthetic effect. Contact OLMC for transports greater
than 30 minutes.
NOTES & PRECAUTIONS:
A. Any blunt trauma to the eye can cause blood in the anterior chamber. Patients should be
transported sitting at least at a 30 degree angle unless contraindicated. This prevents an acute rise in intraocular pressure. Document new onset of blurring, double vision, blind spots or
perceived flashes of light.
B. Patients with serious eye injuries can be surgical candidates and may require transport to a
specialized facility. Contact OLMC as soon as possible to determine destination facility.
C. Determine when the patient has last had food or drink and minimize oral intake during
D. Patients with corneal abrasions may exhibit symptoms of a foreign body in the eye. Cover both
Marion & Polk County Regional Treatment Protocols
Treatment - Page 23 of 42
TREATMENT:
A. Start Oxygen per General Airway Management protocol. B. Monitor vital signs, ECG and oxygen saturation. C. Establish venous access. D. If hyperkalemia is suspected based on history and physical findings:
1. Administer10% Calcium Gluconate 10 cc slow IV/IO over 5 – 10 minutes in a
2. If no change in rhythm following Calcium Gluconate administration and transport
time is prolonged consider alternate therapy per OLMC contact:
a) Glucose and regular insulin if available b) High dose Albuterol (10 mg by nebulizer)
c) Sodium Bicarbonate 50 mEq IV/IO.
PEDIATRIC PATIENTS:
A. If hyperkalemia is suspected based on history and physical findings:
1. Administer10% Calcium Gluconate 0.6 mL/kg slow IV/IO over 5 – 10 minutes in a
2. If no change in rhythm following Calcium Gluconate administration and transport
time is prolonged consider alternate therapy per OLMC contact:
a) Glucose and regular insulin if available b) High dose Albuterol (10 mg by nebulizer) c) Sodium Bicarbonate 1 mEq/kg IV/IO.
NOTES & PRECAUTIONS:
A. Treatment is going to be based on patient history. Renal failure may elevate blood
potassium levels (hyperkalemia) causing bradycardia, hypotension, weakness, weak pulse and shallow respirations. Other patients who are predisposed to hyperkalemia are those who have muscular dystrophy, paraplegia/quadriplegia, crush injury, or patients who have sustained serious burns more than 48 hours ago. A 12-lead ECG may be helpful.
B. ECG changes that may be present with hyperkalemia include:
1. Peaked T waves 2. Lowered P wave amplitude or no P waves 3. Prolonged P-R interval (greater than 0.20 seconds) 4. Second degree AV blocks 5. Widened QRS complex
C. DO NOT mix Sodium Bicarbonate solutions with Calcium preparations. Slowly flush
remaining Calcium Gluconate from the catheter prior to administering Sodium
KEY CONSIDERATIONS:
Previous medical history
Medications and allergies
Marion & Polk County Regional Treatment Protocols
Treatment - Page 24 of 42
TREATMENT:
A. Start Oxygen per General Airway Management protocol. B. Monitor vital signs, ECG and oxygen saturation. C. Establish venous access as needed. D. Remove clothing and begin cooling measures that maximize evaporation. (Spray bottle
with tepid water, cool wipes, fans.)
E. If blood pressure is less than 90 mmHg systolic, treat per Shock Protocol.
NOTES & PRECAUTIONS:
A. Heat stroke is a medical emergency. Differentiate from heat cramps or heat exhaustion.
Be aware that heat exhaustion can progress to heat stroke.
B. Wet sheets over a patient without good airflow will increase temperature and should be
C. Do not let cooling measures in the field delay transport.
D. Suspect hyperthermia in patients with altered mental status or seizures on a hot, humid
E. Consider sepsis and/or contagious disease. Examine patient for rashes or blotches on
the skin or nuchal rigidity.
F. Malignant Hyperthermia
a. Signs/Symptoms- Increased ETCO2, trunk or body rigidity, acidosis, tachycardia,
trismus or Masseter spasm, tachypnea, increased temperature (late sign)
b. Treatment- Hyperventilate w/100% O2 more than 10 L/min, cool patients with
core temperatures over 39 C utilizing active and/or passive cooling measures.
c. Dysrhythmias usually respond to treatment of acidosis and hyperkalemia.
Contact OLMC for consideration of; Sodium Bicarbonate and/or Glucose and/or Calcium Gluconate.
KEY CONSIDERATIONS:
History of onset
Is patient sweating?
Patient's temperature
Recent infection/illness
Medications and allergies
Marion & Polk County Regional Treatment Protocols
Treatment - Page 25 of 42
HYPOTHERMIA
Start Oxygen per General Airway Management protocol.
Assess ABC's. Allow up to 30-45 seconds to confirm respiratory
arrest, pulseless cardiac arrest or bradycardia that is profound enough
Gently remove wet clothes and protect patient from further
environmental exposure.
Patient perfusing
VF/PULSELESS VT/ASYSTOLE
Monitor ECG and pulse oximetry.
Handle patient gently to avoid VF
Treat per Cardiac Arrest Guidelines Contact OLMC for medication
Warm patient as required with:
ORGANIZED RHYTHM
Heated blankets
Warm environment
Contact OLMC for direction regarding CPR
and medication administration.
Warm (109°F) IV/IO fluids
FROZEN TISSUE/LIFELESS
Consider declaring death in the field. If in
Patients with severe hypothermia
doubt, consult OLMC for directions.
(Temp less than 86F) may need internal rewarming. Contact OLMC early for direction.
NOTES & PRECAUTIONS:
A. At-risks groups for hypothermia include trauma victims, alcohol and drug abuse patients,
homeless persons, elderly, low income families, infants and small children, and entrapped patients.
B. Hypothermia may be preceded by other disorders (alcohol, trauma, OD) look for and treat any
underlying conditions while treating the hypothermia.
C. The hypothermic heart may be unresponsive to cardiovascular drugs, pacer stimulation or
KEY CONSIDERATIONS:
Cool, rainy weather
Prolonged exposure
Marion & Polk County Regional Treatment Protocols
Treatment - Page 26 of 42
TREATMENT:
A. Start Oxygen per General Airway Management protocol.
B. Monitor vital signs, ECG and oxygen saturation. C. Establish venous access if indicated. D. Control external bleeding with direct pressure, elevation, hemostatic dressings, and/or tourniquet.
Fracture, Sprain or Dislocation
1. Check for pulses, sensation and movement distal to the injury site before and after
2. Splint fractures/dislocations in the position found. If PMS is compromised distal to fracture
consider applying axial traction to bring extremity into normal anatomical position. If
patient complains of increase in pain or resistance is felt, stop and immobilize. If PMS is compromised distal to dislocation, contact OLMC.
3. If fracture/dislocation is open, place a moist sterile dressing over wound and cover with a
4. Elevate and/or place cold packs over fracture site if time/injuries allow.
5. Apply traction splint to femur shaft fractures.
Pelvic Fractures
1. Utilize pelvic sling and secure patient to a backboard to minimize movement and blood loss.
Amputation
1. Cover stump or partial amputation with moist sterile dressing.
2. Splint partial amputations in anatomical position to avoid torsion and angulation. 3. Control bleeding by direct pressure, indirect pressure and/or elevation and/or tourniquet. 4. Wrap amputated part in a sterile dressing, and place in a plastic bag to keep dry. Place bag
in ice water if available.
5. If transport time is prolonged (extended extrication, etc.) consider sending the amputated
part ahead to be prepared for re-implantation. Consider calling for a Field Surgeon for entrapped patients that may need field amputation to extricate.
E. For pain control, give Fentanyl or Morphine Sulfate. Fentanyl dose 50 mcg IV/IO/IM/IN. Repeat with
25-50 mcg every 3-5 minutes as needed to a maximum of 400 mcg as long as BP remains above 100 mmHg systolic and no other contraindications. Contact OLMC if more than 400 mcg is needed for pain control. Morphine Sulfate may be used in place of Fentanyl. 2-5 mg IV/IO/IM to max of 20mg. Fentanyl and Morphine shall not used in conjunction with each other.
PEDIATRIC PATIENTS:
A. Fentanyl dose for children is 1.0 mcg/kg IV/IO/IM/IN. May repeat with 0.5–1 mcg/kg every 3-5
minutes as needed to a maximum of 4 mcg/kg. Do not exceed adult dosing. Morphine Sulfate may be used in place of Fentanyl. 0.1 mg/kg IV to max of 20mg. Fentanyl and Morphine shall not
used in conjunction with each other.
B. Consider non-accidental trauma as a cause of injury.
NOTES & PRECAUTIONS:
A. Repeat vital signs often.
KEY CONSIDERATIONS:
Mechanism of injury
Previous medical history
Medications and allergies
Quality of distal pulses
Capillary refill
Marion & Polk County Regional Treatment Protocols
Treatment - Page 27 of 42
MUSCULOSKELETAL – Spinal Injury
TREATMENT:
A. Provide initial cervical spine immobilization using manual in-line stabilization. B. Start Oxygen per General Airway Management protocol. C. Immobilize patient using a long spine board if the patient has a mechanism with the
potential for causing spinal injury and who has ANY of the following clinical criteria:
1. Altered mental status. 2. Evidence of intoxication. 3. Distracting injury (extremity fracture, communication barrier, situational or
emotional distress, drowning, etc.).
4. Neurological deficit of an extremity (numbness, tingling, paralysis). 5. Spinal pain or tenderness.
D. Monitor vital signs and establish venous access as needed. E. Complete a secondary exam to include serial neurological status after immobilization.
PEDIATRIC PATIENTS:
A. Children may require extra padding under the upper torso to maintain neutral cervical
B. Movement of the pediatric patient on the backboard can be minimized by using dense,
soft support material on both sides of the patient prior to securing the straps on the backboard. Consider using a short-spine device (OSS, KED) to immobilize the patient prior to placing on the backboard.
C. Children are very susceptible to respiratory compromise from straps that are too tight.
Continually monitor child's respiratory status and loosen straps as needed.
NOTES & PRECAUTIONS:
A. If any immobilization techniques cause an increase in pain or neurological deficits,
immobilize patient in the position found or position of greatest comfort.
B. Carefully assess the patient's respiratory status during transport. Loosen straps as
needed to avoid respiratory compromise.
C. Comorbid age factors (less than 12 or over 60) may impact the EMT's ability to assess
the patient's perception and communication of pain. A conservative approach to immobilizing these patients is recommended.
D. Patients in the third trimester of pregnancy who are immobilized should have the right
side of the backboard elevated six inches.
E. If sports injury, immobilize patient per NATA guidelines.
KEY CONSIDERATIONS:
Mechanism of injury
Loss of consciousness
Medications and allergies
Neurological deficits
PMS before and after immobilization
Pain or tenderness
Marion & Polk County Regional Treatment Protocols
Treatment - Page 28 of 42
MUSCULOSKELETAL – Spinal Injury
SIGNIFICANT MECHANISM OF INJURY:
Meets state trauma system entry criteria under mechanism: fall from height (more
than 3 ft. or 5 stairs), axial load (e.g. diving, football leading with helmet),
motorized vehicle crash > 50 mph, non-motorized vehicle crash > 20 mph, ejection,
co-occupant death, auto vs. pedestrian
– Language barrier
– No paresthesias
Or tenderness?
Or deformity?
No pain with ROM of neck?
Marion & Polk County Regional Treatment Protocols
Treatment - Page 29 of 42
MUSCULOSKELETAL – Spinal Injury
NON-SIGNIFICANT MECHANISM OF INJURY:
Includes ground-level fall, penetrating trauma, fist fights, other low velocity impacts
– Language barrier
– No paresthesias
Or tenderness?
Or deformity?
Marion & Polk County Regional Treatment Protocols
Treatment - Page 30 of 42
MUSCULOSKELETAL – Spinal Injury
GUIDING PRINCIPLES OF SMR:
Clear, complete documentation is required for patients evaluated for and cleared from
Any hard device used to move patients to cot (scoop stretcher, backboard, slider board,
etc) will be removed after the patient is placed on the stretcher. The only exceptions are
those patients who had a severe mechanism of injury and were noted to have a neurologic
deficit or who were unresponsive during the assessment.
Patients who can self-extricate or who are ambulatory on scene will have a collar placed
and will be helped to cot under their own power. No standing takedowns.
Children in car seats with a built-in 5 point harness (not a belt-positioning booster) may
remain strapped in the car seat as long as patient care is not hindered.
An improvised soft collar should only be used if a rigid collar worsens the patient's
condition (e.g. airway obstruction, increased pain) or if a rigid collar simply will not fit. If the
patient meets criteria for a rigid collar (i.e. Full or modified SMR) and one cannot be placed, an EMT will maintain manual C-spine stabilization en route to the hospital.
Marion & Polk County Regional Treatment Protocols
Treatment - Page 31 of 42
NAUSEA AND VOMITING
TREATMENT:
A. Start Oxygen per General Airway Management protocol.
B. Monitor vital signs, ECG and oxygen saturation.
C. Start IV/IO if needed; if shock syndrome is present follow Shock protocol.
D. Consider fluid challenge in patients exhibiting signs of dehydration.
E. Consider offering patient an isopropyl alcohol swab and allowing the patient to self-
administer the swab by inhalation. Emphasize slow deep inhalation. May be repeated up to 2 times (total of 3 administrations) but should not delay the administration of Ondansetron.
F. Give 8 mg Ondansetron orally dissolving tablets (Zofran® ODT) , 4 mg Ondansetron
slow IV/IO push over 2 minutes, or 4 mg Ondansetron IM
1. If nausea and/or vomiting are inadequately controlled after 10 minutes, may
repeat Ondansetron once.
G. If patient continues to vomit administer fluid challenge and consider other causes.
PEDIATRIC PATIENTS:
A. Ondansetron use in patients under 2 years of age requires OLMC consultation except
for children in spinal immobilization or children receiving chemotherapy.
B. For children 2-12 years of age, administer one (1) 4mg Ondansetron orally dissolving
tablet (Zofran® ODT) or administer Ondansetron 0.1mg/kg via slow IV/IO push over 2 minutes up to a total maximum IV/IO dose of 4mg. Consider IM at same dose if unable to start IV and ODT tablet is contraindicated.
NOTES & PRECAUTIONS:
A. Do not administer Ondansetron (Zofran®) to patients with a hypersensitivity to the
drug or other 5-HT3 type serotonin receptor agonists (e.g., dolasetron [Anzemet®] and granisetron [Kytril]
B. Do not administer alkaline medications or preparations in the same IV/IO as
Ondansetron as it may cause precipitation.
C. Monitor EKG for ectopy and QT prolongation following administration.
KEY CONSIDERATIONS:
Vomiting blood or bile
Complaint of nausea
Medications and allergies
Abdominal pain or trauma
Orthostatic vital signs
Marion & Polk County Regional Treatment Protocols
Treatment - Page 32 of 42
NEONATAL RESUSCITATION
BIRTH to 3 months
Is this a term gestation?
Is the amniotic fluid clear?
Warm and dry infant
Is the infant breathing or crying?
Consider plastic wrapping infant
Does the infant have good
Clear airway if needed
Assess APGAR Follow Normal Childbirth protocol
Warm and dry infant
Position and clear airway as
needed. If thick meconium is
present and infant is depressed, suction airway.
Dry, stimulate and reposition
Evaluate respirations, heart rate, color
HR > 100, pink
Observe Assess APGAR
Follow Normal Childbirth
Continue to assess and
Provide positive pressure ventilation
Effective ventilation
HR > 100 & Pink
Provide positive pressure ventilations
Begin chest compressions
Establish IO access; consider IV if time and personnel permit.
Administer 1:10,000 Epinephrine 0.01 mg/kg IO/IV. Repeat every 3-5 minutes. Consider fluid challenge with Normal Saline 10 cc/kg. Use warm IV fluids if available. In prolonged resuscitation, consider Sodium Bicarbonate 1 mEq/kg IO/IV over 2 minutes. Check CBG and treat per Altered Mental Status protocol.
Marion & Polk County Regional Treatment Protocols
Treatment - Page 33 of 42
NEONATAL RESUSCITATION
NOTES & PRECAUTIONS:
A. Do not use Atropine in neonatal resuscitation. B. If meconium is lightly stained and infant is vigorous (strong respiratory effort, good
muscle tone, heart rate over 100 bpm) endotracheal suctioning should not be performed.
C. An infant may need resuscitation if intra-partum risk factors for asphyxia are present
(prolapsed cord, painful bleeding, prolonged rupture of membranes, maternal fever, multiple births, abnormal presentation, maternal hypo-hypertension or seizure)
KEY CONSIDERATIONS:
Fetal presentation
Maternal health/risk factors
Maternal medications
Previous birth difficulties
APGAR SCORE:
Body pink, blue extremities
Slow (less than 100 bpm)
Marion & Polk County Regional Treatment Protocols
Treatment - Page 34 of 42
POISONING AND OVERDOSE
USE PROPER PRECAUTIONS. DECONTAMINATE PT PRIOR TO TREAMENT/TRANSPORT
TREATMENT:
A. Start Oxygen per General Airway Management protocol. B. Monitor vital signs, ECG and oxygen saturation. C. Establish venous access. If systolic BP is less than 90 mmHg follow Shock Protocol. D. If unknown poison or overdose and patient has a decreased level of consciousness treat per
Altered Mental Status protocol. Contact Poison Control for advice.
E. Treat specific poisons/overdoses as outlined below:
Aspirin or Acetaminophen:
1. If it is less than two hours since ingestion, administer 50 grams of Activated Charcoal
2. Consider antiemetic.
Beta blockers:
Contact OLMC for consideration of Glucagon, 1 mg IV/IO.
Calcium channel blocker:
Contact OLMC for consideration of Calcium Gluconate 10cc of 10% over 5-10 min.
Carbon Monoxide:
1. Remove patient from area if not already done. 2. Apply CO monitor if available. Treat findings per Carbon Monoxide Exposure
procedure. If patient is pregnant, comatose, has cardiac ischemia or focal neurological symptoms, contact OLMC.
Tricyclic antidepressant:
1. If patient exhibits arrhythmias or a widening QRS complex administer Sodium
Bicarbonate 1 mEq/kg IV/IO.
2. Treat hypotension per Shock protocol.
Organophosphates:
1. Prepare to handle copious secretions. 2. Administer Atropine 1–2 mg until symptoms improve. Contact OLMC for frequency of
dosing. See Haz-Mat Protocol for more specifics of treatment.
F. Contact Poison Control as needed. (local 503-494-8968, landline only:1-800-222-1222) G. Contact OLMC for advice on Activated Charcoal for other ingested poisons if not previously
advised to by Poison Control Center.
PEDIATRIC PATIENTS:
A. Atropine dose for children if indicated is 0.05 mg/kg. Minimum single dose 0.1 mg, maximum
single dose 1 mg. Contact OLMC for dosing.
B. Aspirin or Acetaminophen: 1.0 g/kg Activated Charcoal orally.
C. Beta-blocker: Contact OLMC or Poison Control for Glucagon use in patients under 2 years of
NOTES & PRECAUTIONS:
A. If the patient exhibits extrapyramidal symptoms/dystonias with a history of phenothiazine use,
consider diphenhydramine.
B. For large organophosphate poisonings, refer to Haz Mat protocol.
C. Do not neutralize acids or alkalis.
D. Consider Haz Mat Team activation.
KEY CONSIDERATIONS:
Route of poisoning
Amount of ingestion
Multiple patients
Psychiatric history
Marion & Polk County Regional Treatment Protocols
Treatment - Page 35 of 42
POISONING AND OVERDOSE
TOXIDROME TABLE
Toxidrome Examples Clinical
Features
Antidotes
Sympathomimetic Cocaine
Midazolam (OLMC)
Diaphoresis Hypertension Hyperthermia Dilated pupils
Depressed mental
Constricted pupils
Cholinergic (anti-
Pralidoximine (2-
Organophosphates
Sedative-Hypnotic Barbiturates
Depressed mental
Hypotension Hypothermia
Cardiotoxic drugs Beta-blockers
Calcium channel blockers Conduction issues
Physostigmine (ED)
Tricyclic antidepressants
Altered mental status
Sodium Bicarbonate
Type 1A – quinidine,
Type 1C – flecanide,
*Muscarinic **Nicotinic
***Central
Diarrhea ,Urination, Miosis,
Mydrasis, Tachycardia,
Confusion, Convulsions,
Bradycardia, Bronchospasm,
Weakness, Hypertension,
Bronchorrhea, Emesis,
Hyperglycemia, Fasciculations
Lacrimation, Salivation, Sweating
Marion & Polk County Regional Treatment Protocols
Treatment - Page 36 of 42
RESPIRATORY DISTRESS
TREATMENT:
A. Administer oxygen, obtain vital signs, SpO2, ETCO2 and ECG.
B. Assess lung sounds. C. Start IV if needed. D. Follow appropriate Airway Management or Cardiac Dysrhythmia protocol if indicated.
E. Treat patients clinical impression as follows:
Upper Airway
1. Croup & Epiglottitis – Transport in position of comfort, monitor airway 2. Anaphylaxis – Treat per Anaphylaxis and Allergic Reaction protocol. 3. Foreign Body – Obstructed airway procedures. Remove object using direct laryngoscopy
if complete obstruction.
4. Complete Obstruction – If you cannot effectively BVM ventilate the patient and the
patient is deteriorating, consider cricothyrotomy.
Pulmonary Edema
1. Sit patient upright. 2. If BP is less than 100 mmHg systolic, consider cardiogenic shock and treat with
dopamine drip per Shock protocol.
3. If BP is more than 100 mmHg systolic:
a. Nitroglycerin 0.4 mg SL every 3-5 minutes as long as pt's BP remains above 100
mmHg systolic. (Do not administer Nitroglycerin without OLMC approval if pt has
taken Viagra®, Levitra® or other similar drugs in the last 24 hours, or Cialis® within the last 48 hours).
b. If the patient remains in severe respiratory distress (e.g. unable to speak more than
1-2 words, SPO2 less than 90%, resp rate over 40) start CPAP if available.
1. Albuterol, 2.5 mg and one unit dose of Ipratropium 0.5 mg by nebulizer. Repeat as
needed, max Ipratropium doses are 2.
2. If patient has moderate to severe respiratory distress based on Severity Assessment
Guide, give Dexamethasone 10 mg IV/IO slowly over 1-2 minutes.
3. Administer CPAP if available. 4. If continuous nebulizer treatment is needed contact OLMC for advice.
Asthma
1. Albuterol, 2.5 mg and one unit dose of Ipratropium 0.5 mg by nebulizer. Repeat as
needed, max Ipratropium doses are 2.
2. If patient has Moderate to Severe asthma based on Severity Assessment Guide, give
Dexamethasone 10mg IV/IO slowly over 1-2 minutes.
3. If patient is deteriorating and over 40 years old, give epinephrine 0.3 mg IM or SQ. For
patients 40 years or older with known cardiac history, administer epinephrine 0.15 mg IM or SQ.
4. If transport time is long and asthma is severe, administer Magnesium Sulfate (usual dose
is 4 grams over 10 minutes).
5. If continuous nebulizer treatment is needed during transport (which may be necessary in
some pediatric patients) contact OLMC for advice.
6. Consider use of CPAP if available.
Marion & Polk County Regional Treatment Protocols
Treatment - Page 37 of 42
RESPIRATORY DISTRESS
PEDIATRIC PATIENTS:
A. Upper Airway
1. In patients 6 months to 6 years of age with audible stridor at rest, give 5cc epinephrine
1:1,000 via nebulizer.
2. Treat anaphylaxis and foreign body obstruction per adult guidelines.
3. The usual cause of respiratory arrest in children with croup, epiglottitis or laryngeal
edema is exhaustion, not complete obstruction. If the child with suspected upper
airway compromise deteriorates, you may still be able to ventilate with a BVM. Only attempt intubation if you cannot effectively ventilate with BVM.
4. If complete obstruction is present and you cannot effectively BVM ventilate the patient
and the patient is deteriorating, consider needle cricothyrotomy.
B. Asthma
1. Give Albuterol and Ipratropium per adult guidelines. 2. If patient is deteriorating give 1:1,000 epinephrine 0.01 mg/kg IM or SQ (max dose 0.3
mg). Contact OLMC for additional doses.
3. If patient has Moderate to Severe asthma based on Severity Assessment Guide and is
not improving with treatment, consider Dexamethasone 0.6 mg/kg IV/IO max 10mg, slowly over 1-2 minutes.
4. Contact OLMC for consideration of Magnesium Sulfate 25-50 mg/kg IV/IO over 10 min.
Max dose 2 grams.
NOTES & PRECAUTIONS:
A. In addition to specific interventions for respiratory distress, aggressive airway management,
including early intubation, is appropriate for the patient who does not respond to treatment or
is rapidly deteriorating.
B. The best indicator for the cause of respiratory distress is past history. If a person has had
COPD or CHF in the past, it is likely the person has the same condition again.
C. In cases of tachypnea it is essential to consider all causes such as pulmonary embolus, hypoxia,
cardiac causes, infection and trauma. Hyperventilation may be a response to an underlying medical problem and should only be considered after these other causes have been excluded.
Do not treat hyperventilation by rebreathing CO2. Reassurance and oxygen via mask are appropriate.
KEY CONSIDERATIONS:
Recent illness/infection
Fever, chills or productive cough
Medications and allergies
Distended neck veins
Peripheral edema
Medical history including asthma, CHF, COPD, pneumonia
ASTHMA SEVERITY ASSESSMENT GUIDE
MODERATE
Short of breath
Able to speak
Heart rate
Respiratory
Elevated Elevated
Lung sounds
End expiratory wheezes
Full expiratory wheezes
Wheezes both phases or absent
Accessory muscle
use
Alertness
Possibly agitated
Usually agitated
Usually agitated
Marion & Polk County Regional Treatment Protocols
Treatment - Page 38 of 42
SEIZURES
TREATMENT:
A. Start Oxygen per General Airway Management protocol.
B. Monitor vital signs, ECG, oxygen saturation and waveform Capnography. C. Establish IV access as required. D. If patient is in status seizure (continuous seizure or repetitive seizures without regaining
1. Administer Midazolam 2-5 mg IV/IO/IN. If no IV, administer Midazolam 5 mg IM.
2. If patient continues to exhibit seizure activity for more than five minutes, repeat
Midazolam 2-5 mg IV/IO/IN or 5 mg IM. Contact OLMC if further doses are needed.
3. Monitor patient's respiratory status closely after Midazolam administration.
E. Check blood glucose and treat per Altered Mental Status protocol. F. Place patient on their left side for transport. G. Transport may be unnecessary if patient becomes fully oriented, is taking their anti-seizure
medication as prescribed, has a physician, and is experiencing their usual frequency of seizure activity. If patient is not transported have the patient (or guardian) sign a Patient Refusal Form
and document the patient's mental status.
H. All first time seizure patients require medical evaluation by a physician. Contact OLMC if
patient refuses transport.
PEDIATRIC PATIENTS:
A. If patient is in status seizure (continuous seizure or repetitive seizures without regaining
1. Administer Midazolam 0.1 mg/kg IV/IO/IN to a maximum total dose of 2.5 mg. If no IV
access, administer Midazolam 0.2 mg/kg IM to a total maximum dose of 5 mg.
2. If patient continues to exhibit seizure activity for more than five minutes, repeat
Midazolam 0.1 mg/kg IV/IO/IN or 0.2 mg/kg IM. Contact OLMC if further doses are needed.
3. Monitor patient's respiratory status closely after Midazolam administration.
B. Febrile seizures are generally found between the ages of 1- 6 and are usually short in duration.
Administer Acetaminophen 15mg/kg rectally, max dose 1.0 gm.
C. First time seizures in children should be considered sepsis or meningitis until proven otherwise.
NOTES & PRECAUTIONS:
A. Seizures in patients over 50 years of age are frequently caused by arrhythmias. Treat per
appropriate protocol.
B. New onset of seizures in a pregnant patient, especially in the third trimester, may indicate
toxemia of pregnancy. Administer Magnesium Sulfate – 4 grams IV/IO over 10 minutes via Buretrol.
C. Remember to check a pulse once a seizure stops. Seizure activity may be the sign of hypoxia or
KEY CONSIDERATIONS:
Length/type of seizure
Medications and allergies
Prior seizure history
ETOH or drug use
Marion & Polk County Regional Treatment Protocols
Treatment - Page 39 of 42
TREATMENT:
A. Start Oxygen per General Airway Management protocol.
B. Monitor vital signs, ECG and oxygen saturation. C. Establish IV/IO access. D. Prepare for rapid transport.
E. Determine type of shock and treat as follows:
Hypovolemic Shock:
1. Give 500cc fluid bolus, repeat if needed if no signs of pulmonary edema. 2. For trauma or AAA, do not fluid overload. Goal is a systolic BP of 90 mmHg.
Cardiogenic Shock:
1. Follow appropriate cardiac dysrhythmia protocol. 2. Consider fluid challenge.
3. Dopamine infusion. Start at 5 mcg/kg/min and increase in 5 mcg/kg/min increments
every five minutes to a maximum of 20 mcg/kg/min, or until systolic BP is at least 90 mmHg and signs of shock are alleviated.
Distributive Shock (anaphylaxis, sepsis, neurogenic):
1. Give 500cc fluid bolus, repeat if needed if no signs of pulmonary edema. May repeat to a
total of 1,000cc. If shock persists consider dopamine as above.
2. If possible, treat underlying cause.
Obstructive Shock (cardiac tamponade, tension pneumo, aneurysm, PE):
1. If tension pneumothorax is suspected, decompress following Needle Decompression
2. Consider 500cc fluid challenge. 3. Contact OLMC for advice on other care including Dopamine infusion.
PEDIATRIC PATIENTS:
Treat as outlined above with the exception of the follow Fluid Administration guidelines:
1. Infants – 10 cc/kg. 2. Children – 20 cc/kg.
3. Maximum fluid amount in Cardiac and Obstructive shock is 20 cc/kg. 4. Maximum fluid amount Distributive Shock is 500 cc. 5. Contact OLMC early for advice.
NOTES & PRECAUTIONS:
A. Closely monitor patient's respiratory status and vital signs. Avoid fluid overload. B. Other signs and symptoms of shock include confusion, restlessness, altered mental status, moist
skin, apathy and tachycardia.
KEY CONSIDERATIONS:
Mechanism of injury
Marion & Polk County Regional Treatment Protocols
Treatment - Page 40 of 42
TREATMENT:
A. Start Oxygen per General Airway Management protocol.
B. Monitor vital signs, ECG and oxygen saturation. C. Check CBG and treat per Altered Mental Status guidelines. D. Establish IV/IO access (16–18 gauge in proximal site if possible).
E. Transport patient in supine position with less than 15 degree of head elevation if tolerated. F. Expedite transport to hospital.
G. Complete Cincinnati Pre-hospital Stroke Scale. H. Document serial neurologic examinations.
NOTES & PRECAUTIONS:
A. Do not treat hypertension or give aspirin to these patients.
B. Consent is normally required prior to the administration of thrombolytics. Have family/guardian
ride in ambulance to hospital or follow immediately.
C. Intervention with thrombolytics, if indicated, must begin within three hours of symptom onset.
KEY CONSIDERATIONS:
Bleeding disorders
Bedridden/wheelchair pt
Neurological exam
CINCINNATI PRE-HOSPITAL STROKE SCALE
FACIAL DROOP (Ask patient to smile / show teeth)
Normal: Both sides of face move equally well
Abnormal: One side of face does not move as well as the other side
ARM DRIFT (patient closes eyes and holds both arms out palms up)
Normal: Both arms move the same OR do not move at all
Abnormal: One arm does not move or one arm drifts down compared to the
SPEECH (Have patient say "It never rains in Oregon")
Normal: Correct words with no slurring
Abnormal: Words slurred, inappropriate words or is unable to speak
Marion & Polk County Regional Treatment Protocols
Treatment - Page 41 of 42
SUBMERGED PATIENT
TREATMENT:
A. Start Oxygen per General Airway Management protocol. B. Monitor vital signs, ECG and oxygen saturation. C. Establish IV/IO access as required. D. If there is any doubt as to mechanism of injury or any possibility of cervical injury,
immobilize patient and consider Trauma System entry.
E. If indicated, treat per Hypothermia protocol. F. If patient is in cardiac arrest, do not attempt resuscitation if patient has been submerged
for more than 30 minutes, with the following exceptions:
Resuscitation may be initiated if the patient is recovered within 60 minutes if: 1. Children under 6 years of age and water temperature at recovery depth of less
2. Patients who may have been trapped in an underwater air pocket. 3. Water temperature at recovery depth is less than 40 F and information suggests
that patient may have been swimming on the surface for at least 15 minutes prior to becoming submerged.
4. Paramedic discretion.
NOTES & PRECAUTIONS:
A. If patient is still in the water rescue should be performed by properly trained and
equipped personnel only.
B. Be prepared to manage vomiting. C. Even if patient initially appears fine, delayed pulmonary edema is likely to occur.
KEY CONSIDERATIONS:
Medical history
Length of submersion
Water temperature at recovery depth
Medications and allergies Events prior to submersion
Marion & Polk County Regional Treatment Protocols
Treatment - Page 42 of 42
SUBMERGED PATIENT
TREATMENT:
A. Start Oxygen per General Airway Management protocol.
B. Monitor vital signs, ECG and oxygen saturation.
C. Establish IV/IO access as required.
D. If there is any doubt as to mechanism of injury or any possibility of cervical injury,
immobilize patient and consider Trauma System entry.
E. If indicated, treat per Hypothermia protocol.
F. If patient is in cardiac arrest, do not attempt resuscitation if patient has been submerged
for more than 30 minutes, with the fol owing exceptions:
Resuscitation may be initiated if the patient is recovered within 60 minutes if:
1. Children under 6 years of age and water temperature at recovery depth of less
2. Patients who may have been trapped in an underwater air pocket.
3. Water temperature at recovery depth is less than 40 F and information suggests
that patient may have been swimming on the surface for at least 15 minutes
prior to becoming submerged.
4. Paramedic discretion.
NOTES & PRECAUTIONS:
A. If patient is stil in the water rescue should be performed by properly trained and
equipped personnel only.
B. Be prepared to manage vomiting.
C. Even if patient initially appears fine, delayed pulmonary edema is likely to occur.
KEY CONSIDERATIONS:
Length of submersion
Water temperature at recovery depth
Medications and allergies Events prior to submersion
Marion & Polk County Regional Treatment Protocols
Treatment - Page 43 of 43
PROCEDURES
Marion & Polk County Regional Treatment Protocols
DEFINITION:
An AICD is an implanted defibrillator device that consists of: a lead system that senses cardiac
activity, logic circuitry to analyze sensed signals, a power supply for device function and
generating high voltage, and a capacitor that stores and delivers shocks. This device activates
when brady and/or tachyarrhythmias are detected within programmed parameters.
INDICATIONS:
For verified frequent and recurrent inappropriate AICD discharges, a doughnut magnet may be
utilized to deactivate "runaway" devices. Inhibition of AICD devices should be considered when
continuous ECG monitoring verifies malfunction and ACLS is readily available.
PROCEDURE:
1. Contact OLMC. 2. Monitor ECG and verify sinus rhythm AND inappropriate defibrillator discharge. 3. Locate the position of the AICD device. 4. Place doughnut magnet directly over the device. 5. After proper positioning and AICD deactivation, tape magnet securely in place and
NOTES & PRECAUTIONS:
A. It is very important to make the correct diagnosis before utilizing this protocol. Be sure
that the ECG is showing a normal sinus rhythm without ectopy AND indications of recurrent AICD discharges.
B. Some AICD devices will emit varying beeping or continuous tones when magnets are
applied, others will not. Disregard these tones.
C. If the magnet placement is successful in overriding the pulse generation of the AICD,
DO NOT REMOVE THE MAGNET. Some units will return to normal operation
after removal from the magnetic field.
D. Magnets should be stored so as not to come into contact with magnetic sensitive
materials, i.e., monitor screens, tapes, credit cards, magnetic door entry cards, and other electronic equipment.
E. A small percentage of AICDs are impervious to magnetic fields (AICD recipients who
normally work around magnetic fields have these special units). This will not be
deactivated with the doughnut magnet. In such cases, advise OLMC and transport.
F. Consider use of the AICD magnet in deactivating cardiac pacemaker malfunctions. Call
G. Identification information of the AICD type, date implanted, and location of implantation
should accompany the patient to the hospital. This information is typically found on a wallet card that the patient has.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 1 of 39
AIRWAY MANAGEMENT – General Approach
There is a spectrum of adjuncts available to the EMT/Paramedic for airway control. The goal of airway management is to ensure proper oxygenation and ventilation of the patient.
DELIVERY SYSTEMS
A. Nasal Cannula: Used when small amounts of oxygen are desired. Flow rates are generally 2-6
liters/minute. It provides between 24-40% inspired oxygen.
B. Non Re-breather Mask (NRB): Used when higher concentrations of oxygen are needed. It
provides approx 90% inspired oxygen.
C. "Blow-By" Oxygen: Typically used in infants or toddlers or those who cannot tolerate a
cannula or mask.
D. CPAP Continuous Positive Airway Pressure: Improves gas exchange and decreases the work of
breathing in patients who suffer from shortness of breath secondary to CHF/pulmonary edema,
E. Bag Valve Mask (BVM): Used when respiratory drive is compromised and patient needs
ventilatory assistance. Proper facial seal and head positioning are required to deliver
maximum inspired oxygen and effectively ventilate the patient. Capnography and chest rise and fall should be monitored to ensure proper ventilation.
MAINTENANCE DEVICES
A. Nasopharyngeal Airway (NPA)
Used in patients who are unconscious or have an altered LOC and are unable to maintain their own airway and who will not accept an OPA.
B. Oropharyngeal Airway (OPA)
Used in patients who are unconscious or have an altered LOC and are unable to maintain
their own airway.
C. Supraglotic Airways (ex. King Airway, Combitube)
Supraglottic airways were created as an alternative to tracheal intubation or mask
ventilation. Used as alternative when intubation or face mask only ventilation may more difficult or unable to be performed. When placed, capnography and chest rise and fall should
be monitored to ensure proper ventilation. Only to be used with BVM.
D. Endotracheal Intubation
Visualized placement of an airway directly into the trachea. Repeated attempts can cause increased trauma to the oropharynx. Attempts when made should be limited to no more
than two (2) total on any given patient. When placed, capnography and chest rise and fall
should be monitored to ensure proper ventilation. Only to be used with BVM.
E. Needle/Surgical Cricothyrotomy/Pertrach/QuickTrach
Techniques only to be used when all other attempts to establish and maintain an airway
have been unsuccessful. (i.e. you are unable to ventilate using any other method) and a
respiratory obstruction exists.
CAPNOGRAPHY
Perform continuous end-tidal capnography following intubation and placement of advanced airways.
Record numerical values and waveforms on the EMS run report. Ventilation rate and depth should
be adjusted to reflect optimal ETCO2 values for each specific patient complaint. ETCO2 should not
be used as the sole indicator of successful tube placement.
NOTES & PRECAUTIONS:
In trauma patients, airway maintenance with cervical spine control is the primary concern. If unable to establish or maintain an airway, transport the patient to the closest hospital. This includes patients
entered into the Trauma System. Marion & Polk County Regional Treatment Protocols
Procedures – Page 2 of 39
AIRWAY MANAGEMENT – Combitube
DEFINITION:
The Combitube® is a two-tube system similar to the PTL, EOA or EGTA airways. However,
the Combitube® has combined the lumens of an endotracheal and esophageal tubes. The
device is inserted blindly, entering the esophagus (approx. 90% of the time) or the trachea
(approx. 10% of the time). Depending on which structure it enters it will function as an
esophageal or endotracheal ventilation device. The Combitube® may be used by EMT-
Paramedics, EMT-Intermediates and EMT-Basics who have received the appropriate training.
INDICATIONS:
A. Immediate intubation is not available or cannot be performed. B. Access to the patient's head is inhibited due to entrapment.
C. Direct visualization of the larynx is inhibited.
CONTRAINDICATIONS:
A. Patient less than 16 years of age. B. Patient under five (5) feet tall. C. Patient who has an intact gag reflex. D. Patient with known esophageal disease (i.e. varices, cancer.) E. Patient who has ingested a caustic substance.
PROCEDURE:
1. Pre-oxygenate. 2. Place head in neutral position. 3. Insert device using a jaw-lift maneuver to the depth indicated by the marking on the tube.
The black rings on the tube should be positioned between the patient's teeth (or gums if patient has no teeth).
4. Inflate the pharyngeal (large) cuff with 100cc of air. 5. Inflate the distal (small) cuff with 15cc of air. 6. Ventilate through longer blue connector (number 1) tube. 7. Listen for sounds in both lungs and stomach. 8. If you hear breath sounds instead of gastric sounds, continue ventilation through tube
9. If you hear gastric sounds and no lung sounds, begin ventilation through shorter (number
10. Confirm lung sounds with 5-point auscultation. 11. Ventilate with 100% oxygen. 12. Secure using ETT securing device. 13. Apply ETCO2 detection device.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 3 of 39
AIRWAY MANAGEMENT – Needle/Surgical Cricothyrotomy
INDICATIONS:
For pediatric patients 12 years and younger. This technique is to be used only when
other attempts to establish an airway have been unsuccessful (i.e., you are unable to intubate or
ventilate using BVM) and respiratory obstruction exists. Such conditions are most likely to be
found with foreign-body obstruction; facial and laryngeal trauma; inhalation, thermal, or caustic
injury to the upper airway; angioneurotic edema; upper airway bleeding; epiglottitis; and severe
croup.
PROCEDURE:
1. Assemble equipment. 14 ga or 16 ga angiocath, 3cc syringe, 3.0 ETT adapter, oxygen,
2. Place the patient in a supine position with support under the shoulders and mild
hyperextension of the neck unless C-Spine injury is suspected.
3. Palpate the neck in the midline and locate the slight depression just below the notch of
the thyroid cartilage. This is the position of the cricothyroid membrane.
4. Prepare the area with antiseptic solution. 5. Stabilize the airway between thumb and forefingers. 6. Insert the needle with catheter into the cricothyroid membrane at a 30 degree angle
caudally (toward the pts feet).
7. When the needle is through the membrane. Stop and aspirate for air to ensure tracheal
8. Advance the catheter over the needle and then remove the needle. 9. Attach the 3.0 ETT adapter to the hub of the catheter and begin ventilations with the
10. Secure the cannula with tape after confirming correct placement by auscultation for
breath sounds (5 point check). Observe for kinking of cannula.
11. Apply ETCO2 detection device to provide continuous monitoring of waveform
12. Consider sedation with Versed as with RSI if not already given.
NOTES & PRECAUTIONS:
A. Hazards in performing this procedure are primarily those of damage to nearby structures -
major vessels to either side of the midline, to the vocal cords if the puncture is made too high, or a through and through injury of the trachea if the puncture is made too deeply.
B. Palpation of the cricothyroid membrane is very difficult in the infant and young child. The
key to success is immobilization of the trachea throughout the procedure.
C. Needle cricothyrotomy is only a temporizing measure providing oxygenation not adequate
Marion & Polk County Regional Treatment Protocols
Procedures – Page 4 of 39
AIRWAY MANAGEMENT – Needle/Surgical Cricothyrotomy
INDICATIONS:
This technique is to be used only when other attempts to establish an airway have been
unsuccessful (i.e., you are unable to intubate or ventilate using BVM) and respiratory
obstruction exists. Such conditions are most likely to be found with foreign-body obstruction;
facial and laryngeal trauma; inhalation, thermal, or caustic injury to the upper airway;
angioneurotic edema; upper airway bleeding; epiglottitis; and severe croup.
PROCEDURE:
1. Assemble equipment: scalpel, 6.0 cuffed ETT, oxygen, BVM. 2. Place the patient in a supine position with support under the shoulders and mild
hyperextension of the neck unless C-Spine injury is suspected.
3. Palpate the neck in the midline and locate the slight depression just below the notch of
the thyroid cartilage. This is the position of the cricothyroid membrane.
4. Prepare the area with antiseptic solution (i.e Betadine).
5. Stabilize the airway between thumb and forefingers.
6. Make vertical incision through the skin over the cricothyroid membrane 2-3 cm in
length with sufficient depth to expose the cricothyroid membrane. Horizontally
puncture the membrane with the scalpel to facilitate access to the trachea.
7. Insert and maintain airway with a cuffed 6.0 mm Endotracheal tube. Advance cuff 2cm
past the opening and check for chest excursion and auscultate the lung fields. Inflate cuff. Reassess (visualize, palpate, auscultate, compliance).
8. Confirm tube placement by required methods. 9. Apply ETCO2 detection device to provide continuous monitoring of waveform
10. Secure the tube and ventilate with high flow Oxygen.
11. Consider sedation with Versed as with RSI if not already given.
NOTES & PRECAUTIONS:
A. Hazards in performing this procedure are primarily those of damage to nearby structures -
major vessels to either side of the midline, to the vocal cords if the puncture is made too high, or a through and through injury of the trachea if the puncture is made too deeply.
B. Perforating the thyroid gland and lacerating the vagus and phrenic nerve are also possible.
(highly unlikely if placement is midline).
Marion & Polk County Regional Treatment Protocols
Procedures – Page 5 of 39
AIRWAY MANAGEMENT – Pertrach®
INDICATIONS:
This technique is to be used only when other attempts to establish an airway have been
unsuccessful (i.e., you are unable to intubate or ventilate using BVM) and respiratory
obstruction exists. Such conditions are most likely to be found with foreign-body obstruction;
facial and laryngeal trauma; inhalation, thermal, or caustic injury to the upper airway;
angioneurotic edema; upper airway bleeding; epiglottitis; and severe croup.
PROCEDURE:
Place the patient in a supine position with support under the shoulders and mild
hyperextension of the neck. Palpate the neck in the midline and locate the slight depression
just below the notch of the thyroid cartilage. This is the position of the cricothyroid
PerTrach
1. Locate the cricothyroid membrane. 2. Palpate the cricothyroid membrane with gloved hand. 3. Pinch the skin, and make a 1-2 cm vertical incision, cutting away from the patient. 4. Firmly grasp the trachea and insert the needle. 5. Aspirate for air with a syringe. 6. Remove syringe, and thread dilator through needle. 7. Squeeze wings of needle and open out to split needle. Carefully remove needle. 8. Insert dilator into airway, place tube in functional position, (faceplate against skin.) 9. Remove dilator. 10. Inflate cuff with 1-8 ccs of air. 11. Secure the device to the neck and ventilate. 12. Apply ETCO2 detection device to provide continuous monitoring of waveform
13. Consider sedation with Versed as with RSI if not already given.
NOTES & PRECAUTIONS:
Hazards in performing this procedure are primarily those of damage to nearby structures - major
vessels to either side of the midline, to the vocal cords if the puncture is made too high, or a
through and through injury of the trachea if the puncture is made too deeply. The latter is more
commonly seen in infants and children whose tracheas may be deceptively narrow.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 6 of 39
AIRWAY MANAGEMENT – QuickTrach®
INDICATIONS:
This technique is to be used only when other attempts to establish an airway have been
unsuccessful (i.e., you are unable to intubate or ventilate using BVM) and respiratory
obstruction exists. Such conditions are most likely to be found with foreign-body obstruction;
facial and laryngeal trauma; inhalation, thermal, or caustic injury to the upper airway;
angioneurotic edema; upper airway bleeding; epiglottitis; and severe croup.
PROCEDURE:
Place the patient in a supine position with support under the shoulders and mild
hyperextension of the neck. Palpate the neck in the midline and locate the slight depression
just below the notch of the thyroid cartilage. This is the position of the cricothyroid
QuickTrach
1. Locate the cricothyroid membrane. 2. Palpate the cricothyroid membrane with gloved hand. 3. Firmly hold the device and puncture the cricothyroid membrane at a 90 degree angle. No
incision is necessary.
4. After puncturing the cricothyroid membrane, check entry of the needle into the trachea by
aspirating air through the syringe.
5. If air is aspirated the needle is in the trachea. 6. Change the angle of the needle to 60 degrees and advance the device forward into the
trachea to the level of the stopper.
7. Remove the stopper being careful not to advance the device further into the trachea with
the needle still attached.
8. Hold the needle and syringe firmly and slide only the plastic cannula along the needle into
the trachea until the flange rests on the neck.
9. Carefully remove the needle and syringe. 10. Secure the device to the neck. 11. Apply the connecting tube to the device and ventilate. 12. Apply ETCO2 detection device to provide continuous monitoring of waveform
13. Consider sedation with Versed as with RSI if not already given.
NOTES & PRECAUTIONS:
Hazards in performing this procedure are primarily those of damage to nearby structures - major
vessels to either side of the midline, to the vocal cords if the puncture is made too high, or a
through and through injury of the trachea if the puncture is made too deeply. The latter is more commonly seen in infants and children whose tracheas may be deceptively narrow.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 7 of 39
AIRWAY MANAGEMENT – ETCO2 Monitoring
INDICATIONS:
For use to measure effectiveness of ventilation by measuring the amount of carbon dioxide in
exhaled air and the ETCO2 Waveform.
PROCEDURE:
1. Manage airway according to appropriate Airway Management Procedure. 2. Apply continuous waveform Capnography, if available. Maintain ETCO2 output between
The following approximates the degree of ventilation:
Over 40 mmHg = Hypoventilation
35 – 40 mmHg = Normal ventilation
30 – 35 mmHg = Hyperventilation
Less than 30 mmHg =
Aggressive hyperventilation should be avoided in all
Less than 20 mm Hg = Check for effectiveness or ET placement
3. Patients who are posturing, or who have other clinical presentations indicative of head
trauma (blown pupil, focal motor findings) should be ventilated to maintain an ETCO2 level between 30-35 mmHg.
NOTES & PRECAUTIONS:
A. Remember, pulse oximetry does not equate ventilation. You can have a poorly
ventilated patient displaying an oxygen saturation of 100%. Excessively high PaCO2
levels can be detrimental to your patient's outcome.
B. A sudden drop in CO2 output from normal (35-40 mmHg) to 15-20 mmHg and an obvious
change in waveform is indicative of tube displacement, most likely into the hypopharynx.
Apply the DOPE mnemonic (Displacement, Obstruction, Pneumothorax, Equipment failure), re-assess tube placement immediately and take corrective action. Consider other causes of a sudden decrease which include: decompensated shock, flash pulmonary edema, cardiogenic shock and pre-cardiac arrest.
C. DO NOT rely on pulse oximetry or ETCO2 monitoring solely to determine the efficacy
Marion & Polk County Regional Treatment Protocols
Procedures – Page 8 of 39
AIRWAY MANAGEMENT – Endotracheal Intubation
A. Respiratory insufficiency or arrest. B. Airway obstruction. C. Brain injury (GCS of 8 or less). D. Unconsciousness or altered mental status with airway compromise. E. Situations that require positive pressure ventilation. F. With the exceptions of a rapidly closing airway in cases such as thermal injury or anaphylaxis, children under age 12 requiring airway management should not be intubated. BVM or a supraglottic device are acceptable alternatives.
PROCEDURE:
1. Open airway and pre-oxygenate while maintaining cricoid pressure. 2. Assemble and check all equipment, including cardiac monitor, suction, and pulse
3. Intubate in a controlled, but timely manner. 4. Verify placement with ETCO2 detection device and a careful five-point check. Watch for
chest expansion; use continual ETCO2 monitoring and pulse oximetry to confirm patient
5. Secure the tube utilizing ETT securing device. Record ET Tube depth at the teeth or
6. Ventilate and monitor patient's vital signs including SpO2.
7. Administer Midazolam 2-5 mg IV/IO may repeat does once for agitation or
8. Administer Vecuronium 0.1 mg/kg or Rocuronium 0.2 mg/kg as needed for long
transports (longer than 20 minutes) or when Midazolam is ineffective. Paralytics must never be given to an awake patient.
DOCUMENTATION:
The following items should be documented in the PCR:
A. Visualization of the cords (if applicable). B. Number of attempts. C. 5-point check and equal chest expansion. D. Any other devices/ techniques used. E. ETCO2 device used/reading.
F. Any changes during patient contact. G. Reconfirmation of placement after patient movement.
NOTES & PRECAUTIONS:
A. Recheck tube placement with every major movement of the patient, movement of
equipment, or if a significant change in vital signs occurs.
B. DO NOT rely solely on monitoring equipment to determine the efficacy of intubation.
Auscultate for lung sounds and/or re-visualize with laryngoscope.
C. Max ETT attempts is 2 total by any combination of providers. An ETT attempt is defined as
an insertion of the laryngoscope into the mouth.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 9 of 39
AIRWAY MANAGEMENT –Intubation with RSI
Patient meets criteria described previously under "Intubation," and patient has any of the
following:
A. Clenched jaw. B. Active gag reflex. C. Uncontrollable combative behavior. D. Clinical condition requiring airway protection.
PROCEDURE:
1. Open airway and pre-oxygenate while maintaining cricoid pressure. 2. Assemble airway equipment including suction and alternative airway devices and attach
required monitoring equipment (ECG, pulse oximeter). Initiate IV/IO.
3. Pre-medicate with Ketamine (2 mg/kg over 1 minute) OR Etomidate (0.3 mg/kg IV/IO
over 10 seconds).
4. Continue cricoid pressure and maintain until ET tube is in place, verified and secured. 5. Administer Succinylcholine 1.5 mg/kg IV/IO for patients 6 years and older and 2 mg/kg
for patients less than 6 years old.
6. If inadequate paralysis exists after 1 minute, check patency of IV/IO line. If IV/IO not
patent, establish patent IV/IO and repeat succinylcholine with same dose
7. If Succinylcholine is contraindicated, substitute Vecuronium 0.1 mg/kg IV/IO push as the
8. Treat bradycardia with ventilation first. If bradycardia persists, administer Atropine 0.5
mg in an adult patient. 0.02 mg/kg IV/IO for children less than 2 years old. Minimum dose is 0.1 mg. Max dose 0.5 mg
9. Intubate in a controlled but timely manner when patient becomes relaxed. 10. Verify placement with ETCO2 detection device and a careful five-point check. Watch for
chest expansion; use continual ETCO2 monitoring and pulse oximetry to confirm patient
11. Secure the tube using ETT securing device. Record ET tube depth at the teeth. 12. Ventilate and monitor patient's vital signs including ETCO2 and SpO2.
13. If intubations attempts fail, ventilate with BVM. Consider rescue airway or
cricothyrotomy to secure airway if needed.
14. Administer Midazolam 2-5 mg IV/IO if systolic BP is over 100 mmHg to max of 5 mg for
agitation or combativeness. (Pediatric dose is 0.1 mg/kg up to 2.5 mg).
15. If additional paralysis is needed in the event of long transport, administer Vecuronium
0.1 mg/kg or Rocuronium 0.2 mg/kg IV/IO. Dose may be repeated once if needed.
16. Contact OLMC if additional sedation or paralysis is needed.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 10 of 39
AIRWAY MANAGEMENT –Intubation with RSI
NOTES & PRECAUTIONS:
A. Recheck tube placement with every major movement of the patient,
movement of equipment, or if a significant change in vital signs occurs.
B. Check IV/IO patency if the first dose of Succinylcholine does not appear to be effective in
paralyzing the patient.
C. In most situations, the total number of intubation attempts shall be limited to 2. D. DO NOT rely solely on monitoring equipment to determine the efficacy of intubation.
Auscultate for lung sounds.
E. Succinylcholine does not affect the level of consciousness. F. Succinylcholine is contraindicated in the following situations:
1. Hypersensitivity to the drug.
2. Major burns and crush injuries between 48 hours and 6 months old. 3. Stroke or spinal cord injuries with profound residual deficits between 48 hours and
4. Suspected hyperkalemia
Marion & Polk County Regional Treatment Protocols
Procedures – Page 11 of 39
AIRWAY MANAGEMENT – King Airway
DEFINITION:
The KING Airway is a disposable supraglottic airway created as an alternative to tracheal intubation or mask ventilation. The KING Airway is designed for positive pressure ventilation as well as for
spontaneously breathing patients.
INDICATIONS:
Use of the King Airway is indicated if endotracheal intubation can not be performed or after 2 failed
endotracheal intubation attempts and the patient needs a secure airway.
A. Intact gag reflex B. Airway obstruction
C. Patients under 35 inches tall
D. Known or suspected caustic ingestion
E. Known esophageal disease
PROCEDURE:
1. Attach pulse oximeter and monitor oxygen saturation.
2. Ventilate with BVM to optimize oxygen saturation prior to King Airway intubation especially if
several endotracheal intubations were attempted.
3. Estimate patient's height (for sizing of King Airway) and place head in neutral position.
4. Use for both pediatric and adult patients:
a. 35-45 inches tall Size 2 (Green) b. 41-51 inches tall Size 2.5 (Orange)
c. 4 – 5 feet tall Size 3 (Yellow)
d. 5 – 6 feet tall Size 4 (Red)
e. 6 – 7 feet tall Size 5 (Purple)
5. Ensure that both balloons are completely deflated.
6. Lubricate distal end of King Airway.
7. Insert King Airway into mouth posteriorly against the hard palate and in the midline. Advance
gently until the proximal cuff is just visible under the base of the tongue. Inflate the cuffs using the supplied color-coded syringe. The King Airway may rise as it seats itself in the airway.
8. Attach BVM to the tube and ventilate patient. Evaluate breath sounds and air movement over
stomach. Monitor oxygen saturation, chest rise, and attach continuous ETCO2 monitor.
9. Unlike the Combitube, the King Airway device is not designed to ventilate the patient if placed
in the trachea. If unable to ventilate the patient after placement deflate balloons and adjust
depth of tube to optimize ventilation.
10. After successful placement, continue to monitor for adequate ventilations and possible
displacement or balloon failure.
NOTES & PRECAUTIONS:
A. It is important that the tip of the device be maintained in the patient's midline. Keeping
the tip at midline assures that the distal tip is properly placed in the hypopharynx and upper esophagus.
B. Depth of insertion is key to providing a patent airway. A shallow initial insertion will
require deflation of the cuffs to advance the tube deeper.
C. It is extremely important to open the airway and ensure that the tip of the King Airway
advances past the base of the tongue.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 12 of 39
AIRWAY MANAGEMENT – King Airway
SUCTIONING THROUGH THE KING AIRWAY:
Use of the gastric access lumen for suctioning and removal of stomach contents will be at the discretion
of the user.
1. Attach a maximum size 18 Fr suction catheter to a portable suction unit. 2. If necessary, lubricate the catheter with a water-soluble gel. 3. Insert suction catheter into the opening of the gastric access lumen and advance to the
4. Turn on suction unit and maintain continuous suction until there is no further return of
stomach contents.
5. After detaching the suction unit, the suction catheter may be left in place to prevent any
additional stomach contents from being expelled from the gastric access lumen.
6. If active suctioning is not performed, a suction catheter may be placed in the gastric
access lumen to act as a passive vent, and to prevent stomach contents from being expelled from the lumen.
A. It is important that the tip of the device be maintained at the patient's midline. Keeping
the tip at midline assures that the distal tip is properly placed in the hypopharynx and upper esophagus.
B. Depth of insertion is the key to providing a patent airway. A shallow initial insertion will
require deflation of the cuffs to advance the tube deeper.
C. It is extremely important to properly open the airway and ensure that the tip of the
King Airway advances past the base of the tongue.
D. Adequacy of ventilation and position of the King must be re-evaluated any time after a
patient has been moved (e.g., floor to stretcher; stretcher to ambulance, etc.).
Marion & Polk County Regional Treatment Protocols
Procedures – Page 13 of 39
CARBON MONOXIDE EXPOSURE
DEFINITION:
Carboxyhemoglobin monitors (e.g. Masimo Rad-57®) provide quick and accurate non-invasive
measurement of carbon monoxide levels.
INDICATIONS:
A. Measurement of SpCO levels in patients with suspected intentional or unintentional
exposure to carbon monoxide. Examples of unintentional exposures include malfunctioning or misused furnace, engine, heater, stove or grill.
B. Measurement of carbon monoxide levels in patients with smoke inhalation. C. Monitoring of carbon monoxide levels in firefighters. D. Carbon monoxide exposure should be strongly suspected when there are multiple
patients or sick pets from the same location.
PROCEDURE:
If carboxyhemoglobin monitor is available, obtain SpCO readings per manufacturer's guidelines. The SpCO is a screening tool and the patient's clinical picture should be used when making treatment and transport decisions. Symptoms of carbon monoxide exposure include headache, dizziness, nausea, syncope, seizures, and loss of consciousness. Levels should be interpreted based on the attached algorithm.
NOTES & PRECAUTIONS:
A. SpCO levels may be elevated in smokers. Levels can range from 3-20% depending on the
number of packs smoked.
B. Pulse oximeter may provide false readings in patients with elevated SpCO levels. C. If a patient has suspected cyanide poisoning, consider obtaining SpCO, if available,
before administration of Cyanokit™ since the latter will interfere with the carboxyhemoglobin monitor.
D. Transport the following patients directly to a hyperbaric capable facility if you suspect an
isolated exposure (i.e. no significant trauma or burns)
Patient with a SpCO greater than or equal to 25%.
Pregnant patient with a SpCO greater than or equal to 15%.
E. Always consider other causes of altered mental status. Continuous cardiac monitoring is
recommended and if feasible, obtain a field 12 –lead ECG with SpC0 greater than or equal to 10%.
F. In patients with significant burns as well as elevated SpCO readings, the destination
should be the Oregon Burn Center
G. Contact OLMC about transport destination for patients that cannot be readily triaged
by the algorithm. Examples include patients with cardiac ischemia or neurological compromise (focal deficits, confusion, cerebellar dysfunction) as well as multiple patient scenes.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 14 of 39
CARBON MONOXIDE EXPOSURE
• Is Pt's SpCO ≥ 25% or
Transport to
• Pregnant with SpCO ≥ 15%
Hyperbaric chamber
if available.
• Is the Pt symptomatic?
Transport to
Emergency
Department.
• SpCO ≥ 10%
•No Further treatment. •Try to determine source of exposure if non-smoker.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 15 of 39
CHEST DECOMPRESSION – Needle
DEFINITION:
The emergency decompression of a tension pneumothorax using an over-the-needle catheter.
INDICATIONS:
Some of the following signs of simple pneumothorax as well as some of the signs of tension
pneumothorax must be present before decompression is undertaken:
A. Tension pneumothorax (indications):
1. Consistent history, i.e., chest trauma, COPD, patient on positive pressure
2. Shock, low BP or rapidly decreasing BP.
3. Progressive respiratory distress. 4. Tracheal shift away from affected side (late sign). 5. Distended neck veins. 6. Asymmetrical movement on inspiration. 7. Hyperexpanded chest on affected side. 8. Drum-like percussion on affected side. 9. Increased resistance to positive pressure ventilation, especially if intubated.
B. Simple or non-tension pneumothorax is relatively common, is not immediately life
threatening, and should not be decompressed in the field.
PROCEDURE:
A. Expose the entire chest.
B. Clean chest vigorously with alcohol or Betadine.
C. Mid-clavicular Insertion: On affected side, locate the mid-clavicular line and insert a
large gauge angiocath (10-14 gauge) over the superior margin of the third rib. Hit the rib
and then slide over it.
D. Axillary Insertion: On affected side, locate the anterior axillary line and insert large
angiocath (10-14 gauge) over the superior margin of the fifth rib.
E. If the air is under tension, the barrel will pull easily and "pop" out the back of the
syringe. Remove syringe, advance catheter, and remove needle.
F. Secure from movement using tape and/or gauze placed on each side. G. Reassess lung sounds and vital signs. H. Consider administering Midazolam 2-5 mg slow IV/IO push or if no IV/IO, 5 mg IM, to
patient if it can be done concurrently with the procedure. Repeat x 1 if needed.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 16 of 39
CONTINUOUS POSITIVE AIRWAY PRESSURE
DEFINITION:
Continuous Positive Airway Pressure (CPAP) has been shown to rapidly improve vital signs, gas
exchange, and to decrease the work of breathing, the sense of dyspnea, and the need for
endotracheal intubation in patients who suffer from shortness of breath secondary to
CHF/Pulmonary edema, asthma or COPD. In patients with CHF, CPAP improves
hemodynamics by reducing preload and afterload.
INDICATIONS:
Medical patients complaining of moderate to severe respiratory distress meeting ALL the
following criteria:
A. Is awake, oriented and has the ability to maintain an open airway. B. Has signs and symptoms consistent with either CHF/pulmonary edema or COPD. C. Has a systolic blood pressure above 100 mmHg. D. Is over 12 years old and is able to fit the CPAP mask.
A. Respiratory arrest. B. Non-cooperative patient.
C. Suspected pneumothorax D. Hemodynamically unstable E. Inability to maintain mask seal F. Active vomiting
PROCEDURE:
1. EXPLAIN and COACH THE PATIENT ON THE PROCEDURE. 2. Ensure adequate oxygen supply to ventilate device. 3. Place the patient on continuous pulse oximetry. Apply ETCO2 detection device to
provide continuous monitoring of waveform capnography.
4. For the CPAP device, start with 5-10cm H2O and adjust as needed. 5. Place the CPAP mask over the patient's mouth and nose. 6. Secure the mask with the provided straps. 7. Check for air leaks. 8. Monitor and document the patient's respiratory response to the treatment. 9. Continue to coach patient to keep mask in place and readjust as needed.
10. IF RESPIRATORY STATUS DETERIORATES, REMOVE THE DEVICE AND CONSIDER
BAG VALVE MASK VENTILATION AND/OR ENDOTRACHEAL INTUBATION.
REMOVAL PROCEDURE:
CPAP therapy needs to be continuous and should not be removed unless the patient cannot
tolerate the mask or experiences continued or worsening respiratory failure.
SPECIAL NOTES:
A. Advise receiving hospital as soon as possible that a patient with CPAP is en route to
their hospital so they can be prepared for patient.
B. Reassessment of the patient's status is critical and documentation should be performed
every 5 minutes until patient is stable.
C. CPAP mask may be removed temporarily to administer nitroglycerin. D. Suctioning of secretions may be required on some patients. E. Watch for gastric distention and/or nausea.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 17 of 39
PURPOSE:
To define the procedures for inducing hypothermia following post-resuscitation from sudden
cardiac arrest, with the aim to reduce the patients body temperature to 89.6-93.2 degrees F
(32-34 degrees C)
INDICATIONS (Must meet all indications):
A. Patients with a sustained return of spontaneous circulation (ROSC) more than
5 minutes post cardiac arrest.
B. Unconscious and without purposeful response to pain or verbal stimuli.
C. Systolic BP 100 mm Hg or higher (may use pressors to maintain pressure)
CONTRAINDICATIONS:
A. Age: less than 15 years old
B. Traumatic cardiac arrest or suspected significant hemorrhage
C. Hypothermia already present- defined as 93.2 degrees F (34 degrees C)
D. Known pregnancy
E. Refractory or recurrent VF/VT, 2nd, or 3rd degree heart blocks.
F. End stage terminal illness (if known)
COOLING METHODS:
A. Exposure combined with ice packs, and /or
B. Chilled normal saline (NS); stored at a temperature of approximately 39 deg F
C. If applicable, turn off heater on ventilator or use humidifier
PROCEDURES:
A. Remove patient's clothing (undergarments may remain in place)
B. Obtain 12-lead ECG if feasible. If STEMI is identified, follow STEMI protocol.
C. Begin cooling process with ice packs applied to the groin and axilla (wet towels
may be used along with ice packs).
D. If feasible, establish large-bore IV/IO. Using high-pressure bag or other method,
rapidly infuse up to 2 liters of chilled normal saline. Do not administer medications
at the same time through the same IV line as the chilled saline.
E. If patient begins to shiver, move, or have an increased level of consciousness,
administer 2-5 mg Midazolam IV or IO. May repeat once as long as systolic BP is
100 mmHg or higher.
F. Maintain ETCO2 at 35-45 mmHg.
G. Maintain O2 Sat at 94-99%.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 18 of 39
INTRAOSSEOUS (IO) ACCESS/INFUSION
DEFINITION:
Intraosseous cannulation is an alternative technique for establishing IV access in critical adult
and pediatric patients when peripheral IV access is difficult or time-sensitive.
INDICATIONS:
A. Intraosseous infusion is indicated in emergency situations when life-saving fluids or drugs
should be administered and IV cannulation is difficult, impossible or too time-consuming to perform.
B. If a peripheral IV cannot be established after two attempts or within 60–90 seconds of
elapsed time and in:
C. Adult and pediatric patients, within the proper weight range, who present with one or
more of the following clinical conditions: 1. Cardiac arrest. 2. Hemodynamic instability (BP less than 90 mmHg and clinical signs of shock). 3. Imminent respiratory failure 4. Status epilepticus with prolonged seizure activity greater than 10 minutes, and
refractory to IM anticonvulsants.
5. Toxic conditions requiring immediate IV access for antidote.
D. IO placement may be considered prior to peripheral IV attempts in cases of
cardiopulmonary or traumatic arrest, in which it may be obvious that attempts at placing an IV would likely be unsuccessful and or too time consuming, resulting in a delay of life-saving fluids or drugs.
EZ-IO™ PROCEDURE:
1. Determine patient's weight. 2. Assemble all necessary equipment
a. Use the appropriate size IO needle based on manufacturer's recommendation.
3. Adult Site Selection
a. The preferred site is the proximal humerus, two finger widths inferior to the
coracoid process and acromion
b. An alternate site patella and tibial tuberosity. c. Palpate the landmarks at the proximal tibia insertion site should be
approximately one finger width to the medial side of the tibial tuberosity.
d. An alternate site may be used at the distal tibia, two finger widths proximal to
the medial malleolus along the midline of the tibia.
4. Pediatric Site Selection (Patients weighing 3-39 kg)
a. The preferred site is the proximal humerus, two finger widths inferior to the
coracoid process and acromion
b. An alternate site patella and tibial tuberosity. c. Palpate the landmarks at the proximal tibia insertion site should be
approximately one finger width to the medial side of the tibial tuberosity.
d. Insertion site should be one finger width below the tibial tuberosity, then medial
along the flat aspect of the tibia.
e. If the tibial tuberosity cannot be identified on the child, then the insertion site
may be two finger widths below the patella, then medial along the flat aspect of the tibia.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 19 of 39
INTRAOSSEOUS (IO) ACCESS/INFUSION
5. Pediatric Site Selection (Patients weighing 3-39 kg)
a. Palpate the landmarks at the proximal tibia (patella and tibial
b. Insertion site should be one finger width below the tibial tuberosity,
then medial along the flat aspect of the tibia.
c. If the tibial tuberosity cannot be identified on the child, then the
insertion site may be two finger widths below the patella, then medial
along the flat aspect of the tibia.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 20 of 39
INTRAOSSEOUS (IO) ACCESS/INFUSION
6. Needle Insertion
a. Prep the surface with Betadine® and allow to air dry. b. Stabilize patient's leg and begin insertion from a 90-degree angle to the plane of
the tibial plateau. Gently advance the needle set into position–do not force. Stop when you feel the "pop" on smaller patients.
c. When needle is in proper position, remove stylet (if insertion fails, leave the
needle in place and clamp the EZ-Connect; do not attempt second insertion on same leg).
d. Connect extension tubing or EZ-Connect, primed with saline, to IO hub. e. Confirm the catheter position (catheter is stable at a 90-degree angle to the
bone, able to aspirate blood, and fluids flow without evidence of extravasation).
f. Rapid bolus or "power" flush with approximately 10 mL normal saline when
using the EZ-IO AD® needle, and 5 mL normal saline when using the EZ-IO PD® needle.
g. Connect IV tubing and bag to extension tubing or EZ-Connect. h. Consider additional bolus of saline if flow rates slower than expected. i. Utilize a blood pressure cuff or pressure bag to help infuse fluids. j. Dress site, secure tubing and apply wristband.
7. Pain Management
a. If the procedure is performed on a conscious patient, immediately following
placement of the IO needle; for Adult or Pediatric patient administer 0.5 mg/kg IO of 2% Lidocaine up to max of 50 mg slowly through the IO site. Wait approximately 30–60 seconds before flushing with normal saline.
b. In the event a patient regains consciousness and complains of severe pain
secondary to the IO insertion, temporarily stop infusing the fluids, and
administer Lidocaine as in G.1 above. Wait approximately 30–60 seconds before continuing fluid administration.
c. If fluids do not flow freely, flush IO site with an additional 10 ccs normal saline.
PEDIATRIC PROCEDURE WITH MANUAL IO DEVICE:
1. Assemble equipment
a. Approved bone marrow needles, 15 or 18 gauge size b. Betadine® swabs c. Two small syringes (3-5cc) d. One large Luer-lock® syringe (35-50cc) e. Flush solution f. Sterile gauze pads and tape
2. Site Selection – Proximal tibia. Palpate the landmarks and note the entry point that is
the anteriomedial flat surface 1-3 cm below the tibial tuberosity.
3. Prep the surface with Betadine® and wipe dry with a sterile gauze pad.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 21 of 39
INTRAOSSEOUS (IO) ACCESS/INFUSION
4. Needle Insertion
a. Insert the needle at the proximal tibial site, directing the needle caudally. The
needle should penetrate the skin and subcutaneous tissue and be pushed through the cortex of the bone using rotation (avoid rocking the needle) until a "pop" or loss of resistance is felt.
b. Confirm placement of the needle by:
i. Firm fixation of the needle and free aspiration of marrow/blood.
ii. Infusion of 2-3 ccs of NS, palpating for extravasation or noting significant
resistance. If extravasation occurs, further attempts at the site should be avoided.
iii. It is not always possible to aspirate blood/marrow but the line may be
5. Tape and secure IO needle firmly in place. 6. Start Infusion 7. Although gravity drainage may suffice, pressurized infusions may be needed during
8. When infusing medications via an IO route, pressure must be applied to the fluid bag in
order to maintain flow rates. The EMT must continually monitor the rate of infusion.
CONTRAINDICATIONS:
A. Fracture of the bone selected for IO insertion. B. Previous significant orthopedic procedures (IO within 24 hours, prosthesis) C. Infection at the site of insertion. D. Excessive tissue at insertion site with the absence of landmarks.
NOTES & PRECAUTIONS:
A. Osteomyelitis, growth plate injury (in pediatric patients), and extravasation of
fluid with compression of popliteal vessels or the tibial nerve may occur.
B. Airway and breathing should be established first in accordance with other
C. Do not perform more than one attempt in each tibia. D. Any ALS medication may be administered IO.
E. Do not give Hypertonic Saline though an IO line.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 22 of 39
INTRAVENOUS (IV) ACCESS/INFUSION
A. Normal Saline is indicated for replacement of fluid volume losses such as in trauma,
burns, dehydration, or shock.
B. An IV lock may be substituted for an IV line in all situations, except where IV fluid is the
therapy of choice for volume replacement.
PROCEDURE for IV Access:
a. Establish IV access and prepare NS. b. Connect an extension set between the IV hub and the solution bag and tubing. c. All IVs will be started using regular drip sets (15 gtts/cc), unless otherwise
2. IV access with an IV lock:
a. Establish IV access. b. Connect male adapter plug (with pre-flushed short extension tubing) to IV hub. c. After placement, the line should be flushed with normal saline. d. If the IV lock system is used for the administration of medication, the line must
be flushed after each administration.
PROCEDURE for IV Medication Infusions:
1. Using a Buretrol or Soluset type device:
a. Establish IV access and prepare solution. b. Connect the Buretrol between the solution bag and the IV tubing. c. Place one hour's solution into the Buretrol and close the connection between
the Buretrol and the solution bag. Note: The number of microdrops/minute=the number of ccs/hour.
d. Begin infusing solution at the appropriate rate. e. If desired, additional solution may be placed in the Buretrol. The Buretrol
should never contain more than one hour of solution.
2. Using an infusion pump:
a. Establish IV access and prepare solution. b. Connect IV tubing to infusion pump according to manufacturer's directions. c. Begin infusing solution at the appropriate rate.
NOTES & PRECAUTIONS:
Normal Saline should be used with caution in patients with renal impairment (hyperkalemia),
cardiac and respiratory disorders (fluid overload), or extremes of age.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 23 of 39
INDICATIONS:
The ITClamp™50 device is a trauma clamp device for the temporary control of severe bleeding
in the extremities, axilla and inguinal areas.
PROCEDURE: If the patient is conscious, explain procedure.
1. Apply non-sterile gloves. 2. Bring package into close proximity to patient wound. 3. Open the sterile package by pulling forward on the outer tabs. 4. Remove the device from the package by lifting up, taking care to not close the device
until it has been applied to the wound. a. If the device has been inadvertently closed, push the side buttons inward with one
hand, and pull the device open (using the device arms, do not slide fingers beyond the safety bar).
5. Locate the wound edges. 6. Align the device parallel to the length of wound edge. Position the needles
approximately 1-2 cm from the wound edge on either side. a. For very large wounds the device can be applied to one side of the wound first and
then pulled over to the other side, or the tissue can be approximated by hand and then the iTClamp applied.
7. Press the arms of device together to close the device. Device safety seal will break with
8. Ensure the entire wound is sealed and bleeding stops, using your gauze pad to wipe the
area to verify no leaking of blood from the wound. a. A gauze or compression wrap can be placed around the device on the wound to
protect the device and increase the pressure on the wound to limit hematoma expansion. More than one device may be required for large wounds.
9. If bleeding continues:
a. If the device is in the correct position (pressure bars parallel to the wound edges),
close the device more firmly by applying further pressure to the arms of the device.
b. If bleeding continues because the wound is too large: apply a second device (or as
many devices as needed) to the open section.
c. If bleeding continues because the device is not positioned correctly, remove the
device according to instructions and reapply.
d. If desired, wound packing may be done before the iTClamp50 is placed on the
e. The iTClamp50 may be placed on a wound that has a hemostatic agent applied. The
hemostatic agent does not have to be removed.
CONTRAINDICATIONS:
The ITClamp50 is contraindicated where skin approximation cannot be obtained (for example,
large skin defects under high tension).
Marion & Polk County Regional Treatment Protocols
Procedures – Page 24 of 39
CONSIDERATIONS:
1) Assess significance of bleeding wound, capillary bleeds or very slow flowing
hemorrhages may be effectively controlled with directed pressure and standard gauze dressings.
2) If direct pressure will not be effective (either due to hemorrhage rate or lack of
resources), apply the iTClamp(s) as described above.
3) For extreme extremity injuries not amenable to iTClamp application (e.g. the skin edges
cannot be approximated) consider tourniquet application in accordance with policy and procedure.
4) Transport patient to appropriate facility for definitive care of wounds.
PRECAUTIONS:
A. Single-use, disposable device; not for reuse. Re-use of contents may cause cross
contamination, leading to patient risk and complication(s). Can be repositioned.
B. iTClamp50 is provided sterile (sterilized by EtO). Do not use if sterility seal has been
tampered with or packaging is damaged.
C. Not compatible with Magnetic Resonance Imaging (MRI) procedures. D. Dispose of the device in accordance with local guidelines for biohazard sharps. E. The devices and/or component(s) are not made from natural rubber, latex free.
REMOVAL:
A. Removal, except to reposition the device, should be done in a medical facility
prepared to manage the wound.
B. Hold the device by the gripping bars, press the device further closed to release the lock. C. While maintaining this pressure on the arms, press both release buttons with your
D. While pressing the release buttons, pull one of the gripping bars open and rotate the
needles out of the wound, one side at a time.
E. Pick up the device ONLY by the buttons to prevent accidental contact with needles F. Dispose of the device in accordance with local guidelines for biohazard sharps.
WARNINGS:
A. This device is intended for temporary use only; use beyond three hours has not been
B. Patients must be seen promptly by medical personnel for device removal and surgical
C. Only use device as directed to avoid needle stick injury. D. Do not use where delicate structures are near the skin surface, within 10 mm, such as
the orbits of the eye.
E. Will not control hemorrhage in non-compressible sites, such as the abdominal and chest
F. Ensure personal protective equipment is utilized to protect against potential splashing of
blood during application.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 25 of 39
LEFT VENTRICULAR ASSIST DEVICE
INDICATIONS:
Evaluation of patient with implanted Left Ventricular Assist Device.
DEFINITIONS: HeartMate II®
The current LVAD deployed is the HeartMate II. This device provides "pump" assistance to the
left ventricle in patients with heart failure. It is implanted in the chest of the patient with
connections from the apex of the left ventricle to the ascending aorta. The pump itself is
implanted in the chest with the controller external to the chest with a conduit connecting the
two.
Troubleshooting: HeartMate II
A. Two potential treatable device complications may present with an LVAD patient:
Battery failure Controller failure
B. This may present as a catastrophic failure to the pump with the resulting "cardiac arrest
in the patient" or a "low flow state".
PROCEDURES:
A. When the pump has stopped:
1. Check the connections between the controller and the pump and the power source. 2. Fix any loose connections to restart the pump. 3. If the pump does not restart and the patient is connected to batteries, replace the
current batteries with a new fully-charged pair of batteries.
4. If pump does not start, then change the controller.
B. Changing batteries:
Warning: At least one power lead must be connected to the power source AT ALL
TIMES. DO NOT remove both batteries at the same time or the pump will stop.
1. Obtain two (2) charged batteries from the patient's accessory bag. Charged batteries
should be marked with a white fuzzy tab at the end of the battery.
2. Remove only one battery from the clip by pressing the black tab on the battery clip to
unlock the battery.
3. Controller will start beeping and flashing green light signals when you remove the
battery. This is normal.
4. Replace the new battery by lining up arrows on the battery and clip. 5. Slide a new, fully-charged battery into the empty battery clip by aligning the black
arrows. The battery will click into the clip. Gently tug at battery to ensure connection. If battery is properly secured, the beeping and green flashing lights will stop.
6. Repeat the previous steps with the second battery.
C. Changing Controllers
1. Place the replacement controller within easy reach, along with the battery and battery
clips or PBU/Power Module cable. The spare controller is usually found in the patient's travel case.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 26 of 39
Changing Controllers (continued)
2. Make sure patient is sitting or lying down since pump will momentarily stop during this
3. Rotate the PERC lock on the replacement controller in the direction of the "unlocked
icon" until the PERC locks clicks into the fully unlocked position.
4. Repeat this same step for the original controller until the PERC Lock clicks into the
unlocked position.
5. Attach the power leads on the new, replacement Controller to the battery clips or
PBU/Power Module cable.
6. If using battery power, place fully-charged batteries into the battery clips after attaching
the power leads.
7. Press the silence alarm button on the new, replacement Controller to silence its Red
Heart Alarm for 2 minutes.
8. Disconnect the PERC Lead / Driveline from the original controller by pressing the metal
release tab on the connector socket. The pump will stop and an alarm will sound. The
alarm will continue until power is removed from the original controller. Getting the
new replacement controller connected and the pump restarted is the first
priority.
9. Connect the new replacement Controller.
a. Line up the mark on the PERC Lead connector with the mark on the metal tab of
the new Controller.
b. Fully insert the connector into the socket of the new Controller. The pump should
restart / alarms should stop.
c. Gently tug on the metal end of the lead to make sure the PERC lead is fully inserted
into the socket. DO NOT pull the lead.
10. If the pump restarts, skip to Step 12.
–– OR ––
11. If the pump does not restart and the RED Heart Alarm continues:
a. Firmly press the Silence Alarm or Test Select Button to restart the pump.
If the pump speed is set below 8,000 rpm, the pump will NOT automatically restart when power is restored. Pressing the Silence Alarm or Test Select button is required to restart the pump if the pump speed is set below 8,000 rpm.
b. Check the power source. Make sure that power is going to the Controller. c. Gently tug on the metal end of the lead to make sure the PERC lead is fully inserted
into the socket. DO NOT pull the lead.
d. If the pump still does not restart, then try to restart the pump using the System
Controller backup system:
Press and hold both the Test Select and Silence Alarm Buttons at the same time. The RED Heart Alarm will stop and you will hear a repeating cycle of 1 beep per second for 2 seconds, followed by 2 seconds of silence to indicate that the System Controller is operating on the backup system.
12. After the pump restarts, rotate the PERC lock on the new replacement Controller in
the direction of the "locked" icon until the PERC Lock clicks into the fully locked position. If unable to engage PERC lock to the locked position, gently push the driveline into the Controller to assure a proper connection. Retry to engage PERC Lock.
13. Disconnect power from the original Controller. The original Controller will stop
alarming once power is removed.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 27 of 39
PRECAUTIONS:
A. Blood pressure may be difficult to obtain on these patients. Most patients have a mean
arterial blood pressure of 70-90 with a narrow pulse pressure.
B. Palpable pulse may be weak or absent. C. The conduit from the chest to the Controller is the electrical line from the pump which
connects to the Controller which runs the pump.
D. Common presenting non-pump related complications include bleeding and infection. E. If device slows down, LOW FLOW STATE alarm will go off. F. Do NOT perform CPR. G. All ACLS drugs may be administered. H. Pump does not affect the patient's ECG. I. The patient can be defibrillated while connected to the device without any
disconnection required.
J. The patient can be paced; treat per protocol. K. One set of batteries lasts approximately 8-10 hours. L. Any emergency mode of transportation is OK. These patients are permitted to fly. M. Be sure to bring ALL of the patient's equipment with them. N. ALARMS:
1. Yellow or Red Battery Alarm:
a. Need to change batteries
2. Red Heart Flashing Alarm:
a. This may indicate a Low Flow Hazard. Check patient; the flow may be too low.
This alarm will consist of a red heart alarm indicator light and steady audio alarm if the flow rate is less than 2.5 liters per minute.
b. If the patient is hypovolemic, treat per protocol. c. If the patient is in right heart failure, treat per protocol. d. If the pump has stopped, check connections, batteries, and Controllers as
instructed in the section above.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 28 of 39
NASOGASTRIC TUBE PLACEMENT
INDICATIONS:
To alleviate gastric distention which is inhibiting effective ventilation in an intubated patient.
PROCEDURE:
1. Assemble equipment: Proper size Gastric Tubes (10, 12, 14 or 18 French), lubricant, 30
or 60 cc syringe, suction unit.
2. Measure tube length from nose to tip of earlobe and then to xiphoid process. 3. Select nostril. 4. Lubricate end of tube (6-8 inches) 5. Position head in slightly flexed position if no spinal precautions. 6. Gently insert and advance toward posterior nasopharynx and into stomach.
7. Confirm location by:
a. Aspirating gastric contents b. Placing stethoscope over epigastrium and auscultate while inserting 20-30 ccs of
air into the tube.
8. Secure tube in place with tape. 9. Mark and document tube size and depth.
NOTES & PRECAUTIONS:
A. Do not use in patients with obvious skull fracture or severe head/facial injuries with
suspected skull fracture.
B. Do not use in patients with known esophageal varices or who have had caustic
substance ingestion.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 29 of 39
PACING - Cardiac
DEFINITION:
Transcutaneous pacing is the technique of electronic cardiac pacing accomplished by using skin
electrodes to pass repetitive electrical impulses through the thorax.
INDICATIONS:
Transcutaneous pacing should be considered in bradycardia with evidence of inadequate
perfusion, (e.g. altered mental status, chest pain, hypotension, other signs of shock).
PROCEDURE:
1. Ensure ECG pads are attached and monitor displays a rhythm. 2. Attach pacing electrodes to anterior and posterior chest just to the left of the sternum
and spinal column, respectively. Alternatively pads may be placed in the standard anterior and lateral position as with defibrillation. If there is difficulty in obtaining capture, try alternative position.
3. Begin pacing at a heart rate of 80 beats per minute and 30mA current output. 4. Increase current by increments of 10mAs while observing monitor for evidence of
electrical capture. Confirm mechanical capture by checking pulses and BP.
5. If patient is comfortable at this point, continue pacing. If patient is uncomfortable,
administer Midazolam 2-5 mg slow IV/IO push or if no IV/IO, 5 mg IM.
6. If patient still complains of pain, repeat dose of Midazolam® and contact OLMC. 7. If the patient remains unconscious during pacing, assess capture by observing the
monitor and evaluating pulse and blood pressure changes. In the event of electrical capture and no pulses, follow PEA protocol.
8. If there is no response to pacing and drugs, consult with OLMC. If a change in pacing
rate is desired, contact OLMC.
PEDIATRIC PATIENTS:
Use above guidelines except:
1. Give Midazolam 0.1 mg/kg IV/IO to a MAX of 2.5mg. (May repeat once after 5 minutes.)
If more needed, call OLMC.
2. Use anterior/posterior pad placement first for patients less than 1 year. 3. Begin pacing at 100 bpm with smallest mA output.
4. Increase current in increments of 10 mAs while observing monitor for evidence of
electrical capture.
5. Confirm mechanical capture by checking pulses and BP. 6. Contact OLMC for adjustments to rate based on age and response to pacing.
NOTES & PRECAUTIONS:
Transcutaneous pacing should not be used in the following settings:
A. Asystole. B. Patients meeting Death In the Field criteria. C. Patients in traumatic cardiac arrest.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 30 of 39




PELVIC SLING
A. For pelvic instability in the presence of trauma. B. For pelvic pain without instability as a comfort measure.
PROCEDURE:
1. Place the Pelvic Sling underneath the pelvis, centered on the greater trochanters.
Assure the sling is smooth and that the patients pockets are empty (if applicable) to avoid placing pressure on the objects into the patient.
2. Move the adjustable strap so that it will allow enough tension to be made. 3. Place the strap through the buckle and pull tension until the buckle makes a popping
sound. This indicates sufficient tension has been achieved.
4. Secure the strap by the Velcro to the side of the splint.
NOTES & PRECAUTIONS:
A. Blood loss in a pelvic fracture can be significant. Monitor closely and treat per Shock
B. Consider placing prior to extrication from a vehicle if feasible. C. The Pelvic Sling is contraindicated in isolated hip fractures.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 31 of 39
PELVIC SLING
A. For pelvic instability in the presence of trauma. B. For pelvic pain without instability as a comfort measure.
PROCEDURE:
1. Place the Pelvic Sling underneath the pelvis, centered on the greater trochanters.
Assure the sling is smooth and that the patients pockets are empty (if applicable) to avoid placing pressure on the objects into the patient.
2. Move the adjustable strap so that it will allow enough tension to be made. 3. Place the strap through the buckle and pull tension until the buckle makes a popping
sound. This indicates sufficient tension has been achieved.
4. Secure the strap by the Velcro to the side of the splint.
NOTES & PRECAUTIONS:
A. Blood loss in a pelvic fracture can be significant. Monitor closely and treat per Shock
B. Consider placing prior to extrication from a vehicle if feasible. C. The Pelvic Sling is contraindicated in isolated hip fractures.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 32 of 39
PICC LINE ACCESS
PURPOSE:
A Peripherally Inserted Central Line (PICC) is a common method of maintaining long-term
venous access in select patients. PICC lines are typically inserted into the antecubital fossa and
then threaded into central circulation. PICC lines are flushed with heparin to maintain patency,
and therefore, it is imperative to aspirate 5 mL of blood from the line prior to use.
INDICATIONS:
A. PICC lines may be accessed for emergency situations when vascular access is not
obtainable and when there is a need for drug or fluid administration.
B. Patient refuses peripheral IV placement.
CONTRAINDICATIONS:
A. Inability to aspirate or infuse through the catheter. B. Catheter located in any place other than the patient's upper arm. C. Need for rapid fluid resuscitation.
PROCEDURE:
A. Use clean gloves and maintain sterility as much as possible. B. If there is a needleless type port on the distal end of the catheter (figure 1), perform the
following: 1. Scrub the port with an alcohol pad and allow it to dry for 5 seconds. 2. Attach a 10 mL syringe (without saline) to the port. 3. Unclamp if necessary (needless port may not have a clamp). 4. Attempt to aspirate at least 5 mL of blood. Blood should draw freely. If it does not,
remove the syringe and DO NOT use the catheter for access.
5. If blood aspirates freely, remove the 10 mL syringe with blood and discard. 6. Attach a 10 mL syringe with NS and gently flush the line. Never use a smaller
syringe. If line does not flush, remove the syringe and DO NOT use the catheter for access.
7. If line flushes, remove the syringe and attach the catheter to the end of the IV tubing
and begin infusion of NS. Adjust the rate to the needs of the patient within the limits of the catheter.
8. Administer medications though IV tubing port if indicated.
C. If there is a capped needle-type port on the distal end of the catheter (figure 2), perform
the following: 1. Scrub the cap with an alcohol pad and allow it to dry for 5 seconds. 2. Clamp the catheter tubing using ONLY the existing clamp on the catheter and then
remove the cap. Never allow a central line to be open to air.
3. Attach a 10 mL syringe on the catheter end. 4. Unclamp the catheter. 5. Attempt to aspirate at least 5 mL of blood. Blood should draw freely. If it does not,
re-clamp the line and remove the syringe. DO NOT use the catheter for access.
6. If blood aspirates freely, clamp the catheter again. 7. Remove the 10 mL syringe with blood and discard. 8. Attach a 10 mL syringe with NS.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 33 of 39
9. Unclamp and gently flush the line. Never use a smaller syringe. If line does not flush,
re-clamp the line and remove the syringe. DO NOT use the catheter for access.
10. If line flushes, re-clamp and remove the syringe. 11. Attach the catheter to the end of the IV tubing. 12. Unclamp the catheter and begin infusion of NS. Adjust the rate according to the
needs of the patient within the limits of the catheter.
13. Administer medications though IV tubing port if indicated.
PRECAUTIONS:
A. Do not administer medications, flush or aspirate with less than a 10 cc syringe.
Smaller size syringes generate too much pressure and can damage the
catheter.
B. Do not attempt to reinject aspirated blood as it may contain clots.
C. The maximum flow rates for a PICC line is 125 mL/hr for less than size 2.0 French, and
250 mL/hr for catheters over 2.0 size French.
D. Keep patient's arm straight to avoiding kinking the PICC line and obstructing flow. E. Ensure all line connections are secure. F. PICC lines access the patient's central circulation and the risk of infection is high. Avoid
contamination to ports and connections while accessing.
G. Do not administer the following medications through a PICC line:
H. Adenosine – The line may rupture during rapid infusion due to over pressurization.
I. Dextrose 50% – The catheter can be damaged due to the viscosity of the fluid.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 34 of 39
RESTRAINT OF PATIENTS
PURPOSE:
Patient restraints should be utilized only when necessary and in those situations where the
patient is exhibiting behavior that presents a danger to themselves and/or others.
PROCEDURE:
A. Physical Restraint Guidelines:
1. Use the minimum number of physical restraints required to accomplish necessary
patient care and ensure safe transportation (Soft restraints may be sufficient). If law enforcement or additional manpower is needed, call for it prior to attempting restraint procedures. Do not endanger yourself or your crew.
2. Avoid placing restraints in such a way as to preclude evaluation of the patient's medical
B. Physical Restraint Procedure:
1. Place patient face up on long backboard.
2. Secure ALL extremities to backboard. Try to restrain lower extremities first using
restraint devices around both ankles. Next, restrain the patient's arms at his/her sides;
one arm up and one arm down.
3. If necessary, utilize cervical spine precautions (tape, foam blocks, etc.) to control violent
head or body movements.
4. Secure the backboard onto gurney for transport using additional straps. Secure straps to
the upper part of the gurney to avoid restricting the wheeled carriage.
5. Evaluate the patient's respiratory and cardiac status every few minutes to ensure that no
respiratory compromise exists. Monitor SpO2 if possible.
6. DO NOT tighten chest straps to the point that they restrict breathing. 7. Spit hoods placed by law enforcement may be kept on the patient to prevent purposeful
contamination. Monitor the patient's airway.
C. Chemical Restraint Guidelines:
1. Sedative agents may be used to provide a safe method of restraining violently combative patients. These patients may include alcohol and/or drug-intoxicated patients and restless, combative, head-injury patients.
D. Chemical Restraint Procedure:
1. Evaluate the personnel needed to safely attempt restraining the patient.
2. Prepare for possible hypotensive side effects.
3. Administer Haldol 2.5-5.0 mg IV/IO or IM or Midazolam 2-5 mg IV/IO or IM /IN. If
initial dose has no effect, after 10 minutes, give an additional dose of 2.5 mg of Haldol or 2-
5 mg of Midazolam. Maximum dose is 10mg for Haldol and 10.0mg for Midazolam.
Ketamine 2mg/kg IV 4mg/kg IM excited delirium patients.
4. Vital signs should be assessed within the first 5 minutes and thereafter as appropriate. 5. After administration of Haldol, consider diphenhydramine 1 mg/kg IV/IO or deep IM if the
patient shows signs of EPS to a maximum dose 50 mg.
6. Monitor patients ECG and start IV/IO if not already established.
E. Fol ow altered mental status protocol to address possible causes of combativeness.
NOTES & PRECAUTIONS:
A. Patients who are restrained particularly in a prone position are at risk for asphyxia and sudden
death. Constant evaluation of the patient's respiratory status is necessary.
B. Only the minimum amount of restraint is to be used of the patient's chest area. C. Hypoxia may be a cause of combativeness.
D. Use continuous cardiac and ETCO2 monitoring as soon as safe to detect dysrhythmias, QT
prolongation and unexplained changes in waveform capnography.
E. Be prepared to treat seizures.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 35 of 39
When patient is exhibiting respiratory difficulty secondary to secretions in airway or the potential for
aspiration exists.
PROCEDURE:
A. Oral Suctioning
1. Pre-oxygenate patient with 100% oxygen.
2. Assemble equipment: Suction unit with tonsil tip or dental tip, personal protective
equipment (gloves, goggles, gown).
3. Attach required monitoring equipment.
4. Turn suction unit on and confirm mechanical suction is present. 5. Insert tip without suction.
6. Cover thumbhole to begin suction if using a tip other than dental tip. 7. Apply suction until airway is clear. 8. Monitor patient's oxygen saturation. 9. Re-oxygenate patient for at least 2 – 3 minutes between suction attempts.
B. Tracheal Suctioning
1. Pre-oxygenate patient with 100% oxygen. 2. Assemble equipment: Suction unit, correct size suction catheter, sterile rinse, personal
protective equipment (gloves, goggles, gown).
3. Attach required monitoring equipment. 4. If patient is being ventilated with BVM prior to suctioning, have someone else remove the
bag from end of ET tube prior to suction attempt.
5. Insert catheter into the ET tube without applying suction.
6. Advance catheter as far as possible.
7. Withdraw slowly using intermittent suction while rotating catheter.
8. Do not suction more than 15 seconds.
9. Monitor patient's oxygen saturation.
10. Rinse catheter in sterile saline.
11. Re-oxygenate patient for at least 2 – 3 minutes between suction attempts.
C. Suctioning with Meconium Aspirator
1. If meconium is lightly stained and newborn is vigorous do not suction infant.
2. Assemble equipment: Suction unit, appropriate size ET tube, personal protective equipment
(gloves, goggles, gown.)
3. Attach required monitoring equipment. 4. Turn suction unit on and confirm mechanical suction is present. 5. After infant has been intubated, attach meconium aspirator to end of ET tube. 6. Cover thumbhole to begin suctioning while slowly withdrawing the ET tube. 7. Do not suction for more than 15 seconds. 8. Monitor patient's oxygen saturation and heart rate and stop if patient becomes bradycardic. 9. Re-oxygenate patient for at least 2 – 3 minutes between suctioning attempts. 10. If patient has not been intubated and meconium is thick, at the least, aggressive
oropharyngeal suctioning should be carried out with the largest diameter suction device available.
NOTES & PRECAUTIONS:
A. Do not use in patients with obvious skull fracture or severe head/facial injuries with
suspected skull fracture.
B. Do not use in patients with known esophageal varices or who have had caustic
substance ingestion.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 36 of 39
TASER BARB REMOVAL
INDICATIONS:
Taser® barbs should be removed at the request of law enforcement if:
A. Patient has been adequately subdued as not to pose a danger to Fire/EMS personnel.
B. The barbs are not embedded in the face, neck or groin areas.
DEFINITIONS:
Excited Delirium- Condition characterized by a severe disturbance in the level of consciousness
and a change in mental status over a relatively short period of time, manifested by mental and
physiological arousal, agitation, hostility, and heightened sympathetic stimulation. It can result
from mental illness, substance abuse (usually stimulants) or a combination of both.
PROCEDURE:
1. Perform patient assessment. 2. Monitor vitals and LOC. Ensure that vitals are in the normal limits for the situation. 3. Expose the area where Taser barb has implanted under the skin. 4. Cut wires from the barb if still attached. 5. Place thumb and forefinger above and below the barb parallel to the portion of the shaft
implanted in the patient's skin.
6. Spread your thumb and forefinger apart to stretch the skin tightly over the barb. 7. Holding tension, use needle-nose pliers (or similar tool) with gripping strength and grasp
the end of the barb protruding out of the skin near the wire lead and firmly pull out the barb with one quick jerking motion.
8. Assess the skin where the barb was removed. The skin should be cauterized from the
electrical current. Dress the wound to prevent infection.
9. Transport is always recommended for patients who have been tasered and strongly
encouraged for the following conditions: a. Evidence of excited delirium prior to being tasered
b. Persistent, abnormal vital signs c. History or physical findings consistent with amphetamine or hallucinogenic drug use d. Cardiac history e. Altered LOC or aggressive, violent behavior including resistance to evaluation f. Evidence of hyperthermia g. Abnormal, subjective complaints, including chest pain, SOB, nausea, and headaches h. Suspected injuries from falls; consider head and neck injuries.
NOTES & PRECAUTIONS:
A. Patients should be in police custody and monitored by Police for the safety of medical
B. Do not remove Taser® Barbs from the face, neck or groin area. Stabilize the barbs and
transport to the Emergency Department.
C. Tasers® emit two barbs. Make sure both are removed. Treat all barbs as a bio-hazard and
dispose as you would any other sharps.
D. Potential trauma may have occurred before (during a struggle) or after the patient was hit by
the Taser® (patient falls and hits head).
E. Consider whether the patient meets criteria for Altered Mental Status or Poisonings and
Overdoses protocols.
F. CAUTION: Where barbs have wires still connected to the Taser® Gun, shock can still be
Marion & Polk County Regional Treatment Protocols
Procedures – Page 37 of 39
INDICATIONS:
A tourniquet should be used to control hemorrhagic wounds that have not responded
adequately to direct pressure or in situations of significant extremity bleeding with the need for
additional interventions. A tourniquet is used as a last resort to preserve life.
CONTRAINDICATIONS:
A. Non-extremity hemorrhage B. Proximal extremity location where tourniquet application is not practical
PROCEDURE:
Establish a need for a tourniquet meeting the above criteria.
1. Remove the Tourniquet from the trauma bag and carrying pouch. 2. Open the Tourniquet completely and apply to injured extremity. 3. Route the self-adhering band around the injured extremity and pass the free-running
end of the band through the inside slit of the friction adaptor buckle.
4. Pass the band through the outside slit of the buckle utilizing the friction adaptor buckle
which will lock the band in place.
5. Position the Tourniquet above the wound; leave at least 2 inches of uninjured skin
between the Tourniquet and the wound.
6. Pull the free running end of the Self-Adhering Band tight and securely fasten it back on
itself (if applying to an arm wound). Do not adhere the band past the Windlass Clip. - If applying to a leg wound, the Self-Adhering Band must be routed through both sides of the friction adapter buckle and fastened back on itself. This will prevent it from loosening when twisting the Windlass Clip.
7. Twist the Rod until bright red bleeding has stopped. When the situation permits insure
the distal pulse is no longer palpable.
8. Lock the rod in place with the Windlass Clip. 9. Secure the rod with the strap. grasp the Windlass Strap, pull it tight, and adhere it to
the opposite hook on the Windlass Clip.
10. Must note the time applied (on an ePCR or written on tourniquet) and communicate it
with the receiving facility upon arrival.
11. Dress wounds as per standard wound care protocol. 12. Once a tourniquet has been applied, it is not to be removed by prehospital personnel,
unless under direction of OLMC.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 38 of 39
INTRODUCTION:
Portable ventilators provide consistent tidal volume, rate of ventilations and an inspiratory to
expiratory ratio to patients. The ventilator allows a patient to be ventilated without the use of a
BVM, acting as a third caregiver, as well as requiring less oxygen. The use of this device is
encouraged when available.
INDICATIONS:
A. Ventilators may be used on all patients who have an ETT/ Combitube or King Airway in
B. Pre-hospital use of the ventilator may be beneficial in the following situations:
1. Intermittent breaths for CPR 2. May be used for rescue breathing in place of the BVM.
C. Patients less than 5 kg. D. Do not use on unattended patients.
PROCEDURE:
1. Set controls to the initial recommended settings for adult which are:
a. Relief Pressure setting at 40 cmH20 b. Air Mix at 100% c. Frequency set at 12 breaths/min. d. Tidal Volume set at 750 mL
2. Connect to oxygen supply and switch ventilator to on position. 3. Adjust ventilation parameters to suit patient with appropriate tidal volume (5-8 mL/kg)
a. 12 breaths per minute for adults b. 16 breaths per minute for children c. 20 breaths per minute for infants
4. Briefly occlude the patient connection port of the patient valve with the thumb. Check
that the peak inflation pressure reading on the manometer is appropriate for the patient and that the pneumatic audible alarm sounds and the high inflation pressure indicator shows red.
5. Connect patient valve to ET valve or apply face mask to the patient. 6. Check chest movement and inflation pressure manometer to ensure correct ventilation. 7. Check that the green cycle indicator light flashes during each inflation as the pressure
8. Make adjustments as necessary.
A. The ventilator cannot be used in place of initial ET tube confirmation, including
auscultation, visualization and capnometry. Attach the ventilator after confirmation.
B. It is important to pay attention to oxygen tank levels since there are no low oxygen
C. Always ventilate the patient at 100% oxygen for respiratory arrest, CPR and in
contaminated atmospheres.
D. Low pressure alarms may indicate leakage or insufficient tidal volume. E. High pressure alarm may indicate excessive tidal volume, incorrectly positioned airway
or kinked ET tube.
Marion & Polk County Regional Treatment Protocols
Procedures – Page 39 of 39
MEDICATIONS
Marion & Polk County Regional Treatment Protocols
ACETAMINOPHEN (Tylenol®)
OLMC REQUIRED: None
SUPPLIED: 120 mg suppositories
PHARMACOLOGY AND ACTIONS:
Acetaminophen is a non-narcotic analgesic and antipyretic agent. It is thought that these actions are
mediated through inhibition of prostaglandin synthesis both peripherally and centrally.
INDICATIONS:
For the temporary relief of fever.
Hypersensitivity to the medication.
PRECAUTIONS:
Severe or recurrent pain or high continued fever may be indicative of serious illness.
SIDE EFFECTS AND NOTES:
ADULT DOSING (Not used for adults)
PEDIATRIC DOSING
INDICATION DOSE ROUTE NOTES
15 mg/kg suppository up to a max of 1 gram
ACTIVATED CHARCOAL
OLMC REQUIRED:
A. Aspirin and Acetaminophen with time of ingestion more than two hours.
B. All other poisons or ingestions regardless of time from ingestion.
SUPPLIED: 25 – 50 grams
PHARMACOLOGY AND ACTIONS:
Activated charcoal adsorbs toxic substances ingested and inhibits GI adsorption by forming an effective
barrier between the particulate material and the gastrointestinal mucosa. The effect is greatest if used
within one hour of ingestion.
INDICATIONS:
Management of poisoning or overdose of many substances
A. Patients with altered mental status or the inability to maintain their own airway. B. Patients who have aspirated or with a potential for aspiration.
A. Activated charcoal may be ineffective in some ingestions. B. Milk, ice cream and other dairy products will decrease the adsorption capacity substantially. C. Consider anti-emetic in conjunction with Activated Charcoal.
SIDE EFFECTS AND NOTES:
May cause nausea, vomiting, constipation.
ADULT DOSING
INDICATION DOSE ROUTE NOTES
Poisoning/overdose 50
PEDIATRIC DOSING
INDICATION DOSE ROUTE NOTES
Poisoning/overdose 1
ADENOSINE (Adenocard®)
OLMC REQUIRED: None
SUPPLIED: 6 mg/2 mL and 12 mg/4 mL vials or pre-filled syringes
PHARMACOLOGY AND ACTIONS:
Adenosine is a naturally occurring nucleoside that has the ability to slow conduction through the AV
node. Since most cases of PSVT involve AV nodal re-entry, adenosine is capable of interrupting the AV nodal circuit and stopping the tachycardia, restoring normal sinus rhythm. It is eliminated from the circulation rapidly and has a half-life in the blood of less than ten seconds.
INDICATIONS:
To convert PSVT to a normal sinus rhythm, including PSVT that is associated with accessory bypass
tracts (e.g. Wolff-Parkinson-White Syndrome).
A. Second or third degree heart block. B. Sick Sinus Syndrome
C. Known hypersensitivity D. Atrial fibrillation
A. When doses larger than 12 mg are given by injection there may be a decrease in blood pressure
secondary to a decrease in vascular resistance.
B. The effects of Adenosine are antagonized by methylxanthines such as Theophylline and caffeine.
Larger doses of Adenosine may be required.
C. Adenosine effects are potentiated by dipyridamole (Persantine) resulting in prolonged asystole. D. In the presence of carbamazepine (Tegretol), high degree heart block may occur.
E. Adenosine is not effective in converting atrial fibrillation, atrial flutter or ventricular tachycardia.
SIDE EFFECTS AND NOTES:
May cause facial flushing, shortness of breath, chest pressure, nausea, headache and lightheadedness.
ADULT DOSING
INDICATION DOSE
ROUTE NOTES
6 mg, follow with 12 mg if
Use a large proximal IV
site with fluid bolus
PEDIATRIC DOSING
INDICATION DOSE
ROUTE NOTES
0.1 mg/kg, follow with 0.2
Use a large proximal IV
mg/kg x 1 if needed. Do
site with fluid bolus
not exceed adult dosing.
ALBUTEROL (Ventolin®)
OLMC REQUIRED: For use in hyperkalemic patients including crush injury.
SUPPLIED: 2.5 mg /3 mL vial
PHARMACOLOGY AND ACTIONS:
Albuterol is a potent, relatively selective Beta-2 adrenergic bronchodilator and is associated with
relaxation of bronchial smooth muscle and inhibition of release of mediators of immediate sensitivity from cells, especially MAST cells. The onset of improvement in pulmonary function is within 2-15 minutes after the initiation of treatment and the duration of action is from 4-6 hours. Albuterol has
occasional Beta-1 overlap with clinically significant cardiac effects.
A. To treat bronchial asthma and reversible bronchial spasm that occurs with chronic obstructive
pulmonary disease.
B. To treat hyperkalemia.
CONTRAINDICATIONS: None in the prehosptial setting.
A. The patient's rhythm should be observed for arrhythmias. Stop treatment if frequent PVC's
develop or any tachyarrhythmias other than sinus tachycardia appear or if heart rate increases by more than 20 beats/minute.
B. Paradoxical bronchspasm may occur with excessive administration.
SIDE EFFECTS AND NOTES:
Clinically significant arrhythmias may occur, especially in patients with underlying cardiovascular
disorders such as coronary insufficiency and hypertension.
ADULT DOSING
INDICATION DOSE
ROUTE NOTES
Respiratory distress
2.5 mg, repeat as needed
May add Ipratropium with repeated doses.
PEDIATRIC DOSING (Same as adult)
AMIODARONE (Cordarone®)
OLMC REQUIRED: None
SUPPLIED: 150 mg/3 mL vial or pre-filled syringe
PHARMACOLOGY AND ACTIONS:
Amiodarone depresses automaticity of the SA node. It slows conduction and increases refractoriness of
the AV node. Amiodarone increases atrial and ventricular refractory period and prolongs the QT interval. When given IV it is rapidly distributed. No dosage adjustments are needed for patients with renal, liver or heart failure, or advanced age.
A. Ventricular fibrillation B. Ventricular tachycardia with pulses
CONTRAINDICATIONS: None in cardiac arrest.
A. In high concentrations (over 3 mg/mL) Amiodarone can cause phlebitis. Infusion concentrations
should not exceed 2 mg/mL.
B. Amiodarone will precipitate if administered in the same IV line as sodium bicarbonate or
SIDE EFFECTS AND NOTES:
A. In perfusing patients, Amiodarone may cause hypotension, prolonged QT interval,
proarrhythmic effects (Torsades and ventricular fibrillation), severe bradycardia and atrioventricular block.
B. Non-cardiac toxicities are usually related to chronic administration and include pulmonary
infiltrates, hepatic and/or thyroid dysfunction and peripheral neuropathy.
ADULT DOSING
INDICATION DOSE
V-Fib/Pulseless V-Tach
300 mg, repeat once at
V-Tach with pulse
Mix with 100 mL of NS in
Buretrol and infuse over 10
PEDIATRIC DOSING
INDICATION DOSE
Refractory V-Fib/Pulseless
May repeat x 2 to max of
V-Tach with pulse
5 mg/kg over 20 minutes
Mix with 2 mL/kg of NS in
Buretrol. May repeat x 2 to
OLMC REQUIRED: None
SUPPLIED: 81 mg chewable tablets (children's aspirin)
PHARMACOLOGY AND ACTIONS:
Aspirin inhibits prostaglandins and disrupts platelet function for the life of the platelet. It is also a mild
analgesic and anti-inflammatory agent.
INDICATIONS:
In unstable angina and acute myocardial infarction, aspirin has been shown to lower mortality and is
indicated in patients with suspected ischemic chest pain.
A. Allergy to aspirin or aspirin induced asthma. B. History of bleeding disorder (i.e. hemophilia) C. Current ulcer or GI bleeding. D. Suspected aortic dissection.
SIDE EFFECTS AND NOTES:
A. High doses of aspirin can cause ringing in the ears. B. May cause heartburn, nausea and vomiting.
ADULT DOSING
INDICATION DOSE
ROUTE NOTES
Cardiac chest pain
Should be given even if
patient has taken that day. Maximum daily dose is 1 gram.
PEDIATRIC DOSING (Not indicated for pediatric patients.)
ATROPINE SULFATE
OLMC REQUIRED: None
SUPPLIED: 1 mg/10 mL pre-filled syringe, 2 mg/0.7 mL auto-injector pen, 8 mg/20 mL vial
PHARMACOLOGY AND ACTIONS:
Atropine is a muscarinic-cholinergic blocking agent. As such, it has the following effects:
A. Increases heart rate (by blocking vagal influences). B. Increases conduction through the AV node. C. Reduces motility and tone of the GI tract.
D. Reduces action and tone of the urinary bladder (may cause urinary retention). E. Dilates pupils.
A. To increase the heart rate in bradycardia or pacemaker failure. B. To improve conduction in second and third degree heart block. C. As an antidote for some insecticide exposures (e.g. anti-cholinesterases, organophosphates) and
D. To counteract excessive vagal influences causing some bradysystolic and asystolic arrests.
E. For bradycardia not due to hypoxia when using Succinylcholine.
A. Atrial fibrillation and atrial flutter because increased conduction may speed ventricular rate
B. Not used in neonatal resuscitation.
PRECAUTIONS:
Bradycardia in the setting of an acute myocardial infarction is common and probably beneficial. Do not
treat unless there are signs of poor perfusion (low blood pressure, mental confusion).
SIDE EFFECTS AND NOTES:
A. Atropine blocks cholinergic (vagal) influences already present. If there is little cholinergic
stimulation present, effects will be minimal.
B. Remember in cardiac arrest situations, atropine dilates pupils.
ADULT DOSING
INDICATION DOSE
Every 3-5 min. Max 3 mg
As needed after RSI.
Organophosphates 1-2
Call OLMC for frequency.
PEDIATRIC DOSING
INDICATION DOSE
Symptomatic bradycardia
Do not exceed adult dose.
Organophosphates/Toxin 0.02-0.05
Do not exceed adult dose.
Do not exceed adult dose.
Minimum dose 0.1 mg
IM dose 0.02 mg/kg
CALCIUM GLUCONATE
OLMC REQUIRED: Suspected calcium channel blocker overdose except in cardiac arrest.
SUPPLIED: 10% solution 100 mg / 10 mL vial
PHARMACOLOGY AND ACTIONS:
Calcium is the most common cation in the human body. The majority of the body stores of calcium are
located in bone. It plays an important role in many physiologic functions and is essential for proper nerve and muscle function.
A. Suspected calcium channel blocker overdose B. Hyperkalemia
A. Hypercalcemia and hypercalciuria (hyperthyroidism, Vitamin D overdose, bone metastases).
B. Patients on Digoxin.
A. Extravasation of Calcium salts will cause necrosis of tissue. The IV should be secured and free
blood return into the syringe should be checked 2-3 times during administration. If extravasation does occur, immediately stop administration.
B. Administer slowly (no faster than 2mL/min) and stop if patient complains of distress. Inject
using a small needle in a large vein.
C. Calcium Gluconate will precipitate if mixed with Sodium Bicarbonate. Flush catheter completely
before administering one medication after another.
SIDE EFFECTS AND NOTES:
A. Rapid injection of Calcium Gluconate may cause vasodilatation, decreased blood pressure,
bradycardia, cardiac arrhythmias, syncope and cardiac arrest.
B. One vial of 10 mL Calcium Gluconate 10% contains 1 gram of calcium gluconate salt . 27.2
mg/mL of elemental calcium.
ADULT DOSING
INDICATION DOSE ROUTE NOTES
Hyperkalemia, calcium
Slow IV/IO over 5-10
Use a proximal port.
channel blocker overdose
PEDIATRIC DOSING
INDICATION DOSE ROUTE NOTES
Hyperkalemia, calcium
0.6-1.0 mL/kg. Max
Slow IV/IO over 5-10
Use a proximal port.
channel blocker overdose
CARDIZEM (DILTIAZEM®)
OLMC REQUIRED:
Patients under the age of 15.
Systolic BP less than 100 mmHg
SUPPLIED: 25 mg/ 5 mL vial, or pre-load syringe
PHARMACOLOGY AND ACTIONS:
Diltiazem is a calcium channel blocker that slows AV nodal conduction making it an excellent agent for
slowing Atrial Fibrillation or Atrial Flutter and the conversion of PSVT. It will achieve these goals in less
than 7 minutes in over 80% of patients. It is metabolized by the liver primarily and excreted by the
kidneys.
A. As a second line agent for regular PSVT that has failed to respond to the 6 mg, 12 mg dosing of
B. In the patient with Chronic Atrial Fibrillation or Flutter with a rapid ventricular response (more
than 120 bpm average) AND 1. Who has unstable symptoms of cardiac ischemia (chest pain) AND
2. Has a blood pressure greater than 100 mm Hg systolic.
A. Sick sinus syndrome, or second or third degree AV block in the absence of an artificial
B. Systolic BP below 100 mmHg. Call OLMC for advice. C. Wolff-Parkinson-White Syndrome or patients with a wide-complex tachycardia.
A. Diltiazem will rarely convert PSVT, but will slow the rate, which will improve cardiac output. B. Drug interactions can be seen with Cyclosporin and Tegretol by increasing their levels. This
information should be passed onto the receiving facility.
SIDE EFFECTS AND NOTES:
A. Hypotension is rare and will respond to Calcium Gluconate or saline. B. If sustained hypotension after administration of Diltiazem, try IV fluids and give 10 cc Calcium
Gluconate IV/IO over 1-2 minutes if condition not improved.
C. Site reactions include: itching, burning, flushing, arrhythmias.
ADULT DOSING
INDICATION DOSE
Atrial Fibrillation or
0.25 mg/kg slow IV over
If no response after 15
minutes, give 0.35 mg/kg
PSVT failing to
respond to Adenosine
PEDIATRIC DOSING
INDICATION DOSE
Atrial Fibrillation or
Call OLMC for advice.
PSVT failing to
respond to Adenosine
DEXAMETHASONE (DECADRON®)
OLMC REQUIRED: None
SUPPLIED: 10 mg/1mL vial, 4mg/1mL vial, 20mg/5mL vial
PHARMACOLOGY AND ACTIONS:
Dexamethasone is a long acting, synthetic steroid that suppresses acute and chronic inflammation. In
addition, it potentiates vascular smooth muscle relaxation by beta-adrenergic agonists and may alter airway hyperactivity.
A. Moderate to severe asthma/COPD B. Anaphylaxis after Epinephrine therapy
CONTRAINDICATIONS:
Do not use in patients with known hypersensitivity to corticosteroids.
PRECAUTIONS:
May cause hypertension.
SIDE EFFECTS AND NOTES:
May cause nausea, vomiting, headache, or dizziness.
ADULT DOSING
INDICATION DOSE ROUTE NOTES
Respiratory distress,
PEDIATRIC DOSING
INDICATION DOSE ROUTE NOTES
Respiratory distress,
DEXTROSE
OLMC REQUIRED: None
SUPPLIED: 25 g / 50 mL pre-filled syringe and/or 25 g / 250 mL bag (0.1 g/mL)
PHARMACOLOGY AND ACTIONS:
Glucose is the body's basic fuel. It produces most of the body's quick energy. Its use is regulated by
insulin which stimulates storage of excess glucose outside the bloodstream, and glucagon, which mobilizes stored glucose into the bloodstream.
A. Altered Mental Status and CBG less than 60 for adults and pediatrics and CBG less than 40 for
B. Unconscious patient when history is unobtainable.
CONTRAINDICATIONS: None
A. Extravasation of Dextrose is likely to cause necrosis of tissue. IV should be secured and free
blood return of blood into the syringe or tubing should be checked 2-3 times during administration. If extravasation does occur, immediately stop administration.
B. Report any extravasation to receiving hospital personnel and document on the Prehospital Care
SIDE EFFECTS AND NOTES:
Hyperglycemia may complicate or worsen a number of medical conditions (e.g. myocardial infarction and stroke). D50 should be given whenever hypoglycemia is documented by blood glucose meters. If these findings are not available, the EMT should use judgment based on signs and history.
ADULT DOSING
INDICATION DOSE
Can give orally.
Hypoglycemia (Altered Mental Status)
Titrate to effect
PEDIATRIC DOSING – patients 10-35 kg, CBG less than 60
INDICATION DOSE
May repeat once. Can
Hypoglycemia (Altered
give orally. Titrate to
PEDIATRIC DOSING – patients less than 10 kg, CBG less than 40
INDICATION DOSE
May repeat once. Can
Hypoglycemia (Altered
give orally. Titrate to
DIAZEPAM (VALIUM®)
FOR USE BY HAZMAT TEAM – OR – CHEMPACK ONLY
OLMC REQUIRED: None
SUPPLIED:
Auto-injector: 10 mg in 2 mL; disposable Auto-injector vial; 5 mg/mL concentration Multi-dose vial: 5 mg/mL; 10 mL vial
PHARMACOLOGY AND ACTIONS:
A. Diazepam is a benzodiazepine with anxiolytic, anticonvulsant, and sedative properties. B. Increases the inhibitory processes in the cerebral cortex.
C. Diazepam appears to act on areas of the limbic system, thalamus, and hypothalamus, inducing
anxiolytic effects. Its actions are due to the enhancement of GABA activity. Can be administered PO, IV, IO, IM, or PR.
D. The onset of action is 1-5 minutes for IV administration and 15-30 minutes for IM
administration. The duration of diazepam's peak pharmacological effects is 15 minutes to 1 hour
for IM, IV, or IO administration.
INDICATIONS:
Status seizures secondary to suspected or confirmed nerve agent / organophosphate poisoning
CONTRAINDICATIONS:
A. Acute alcohol or drug intoxication B. Known hypersensitivity C. Myasthenia gravis
A. Do not inject Auto-injector intravenously.
B. Use with caution (but do not withhold) in patients with preexisting cardiac disease, HTN or
SIDE EFFECTS AND NOTES:
A. Decreased level of consciousness
B. Respiratory depression
C. Hypotension
D. Impaired motor functions
When administering diazepam, prepare for advanced airway management should respiratory depression occur. Adverse effects of diazepam can be reversed with a benzodiazepine receptor antagonist such as Flumazenil (Romazicon).
ADULT DOSING
INDICATION DOSE
Status seizure due to nerve Auto-injector:
Repeat as needed q 10-
agent / organophosphate
IM; outer thigh only 15 min
Multi-dose vial:
Multi-dose vial:
PEDIATRIC DOSING
INDICATION DOSE
Age: over 30 days-5 years:
Status seizure due to nerve
0.2-0.5 mg slow IV every 2-5 minutes
agent / organophosphate
Maximum dose: 5 mg
Age: over 5 years:
1 mg slow IV every 2-5 minutes
Maximum dose: 10 mg
DIPHENHYDRAMINE (Benadryl®)
OLMC REQUIRED: None
SUPPLIED: Varies
PHARMACOLOGY AND ACTIONS:
Diphenhydramine is an antihistamine which blocks the action of histamines released from cells during an
allergic reaction. It has direct CNS effects, which may be stimulant, or more commonly depressant, depending on individual variation. Diphenhydramine also has an anticholinergic and anti-Parkinsonian effect which is used to treat acute dystonic reactions to antipsychotic drugs (e.g. Haldol®, Thorazine®,
Compazine®, Inapsine®). These reactions include oculogyric crisis, acute torticollis, and facial grimacing.
A. The second-line drug in anaphylaxis and severe allergic reactions (after epinephrine).
B. To counteract acute dystonic and dysphoric reactions to anti-psychotic drugs.
CONTRAINDICATIONS: None
A. May have an additive effect with alcohol or other CNS depressants.
B. Although useful in acute dystonic reactions it is not an antidote for anti-psychotic toxicity or
C. May cause hypotension when given IV.
SIDE EFFECTS AND NOTES:
Diphenhydramine is rarely necessary in the field. It is not the first-line drug for allergic reactions, but
may be useful for long transports.
ADULT DOSING
INDICATION DOSE ROUTE NOTES
IV/IO or deep IM
Extrapyramidal Symptoms
PEDIATRIC DOSING (same as adult)
DOPAMINE (Intropin®)
OLMC REQUIRED: None
SUPPLIED: 800 mg/10 mL vial or 400 mg/10 mL vial or pre-mix bag
PHARMACOLOGY AND ACTIONS:
Dopamine is the chemical precursor of norepinephrine which occurs naturally in humans and which has
both alpha and beta receptor stimulating actions. Its actions differ with the dosage given:
1-2 mcg/kg/min – dilates renal and mesenteric blood vessels. No effect on heart rate of blood
2-10 mcg/kg/min – beta effects on the heart which usually increase cardiac output without
increasing heart rate or blood pressure.
10-20 mcg/kg/min – alpha peripheral effects cause peripheral vasoconstriction and increased
20-40 mcg/kg/min – alpha effects reverse dilatation of renal and mesenteric vessels with
resultant decreased flow.
A. Primary indication is cardiogenic shock.
B. May be useful in other forms of shock, except hypovolemic.
CONTRAINDICATIONS: Hypovolemic shock
A. May induce tachyarrhythmias, in which case infusion should be decreased or stopped.
B. High doses may cause extreme peripheral vasoconstriction. Conversely, low doses may cause a
decreased blood pressure due to peripheral dilatation.
C. Should not be added to Sodium Bicarbonate or other alkaline solutions since Dopamine will be
inactivated in alkaline solutions.
SIDE EFFECTS AND NOTES:
A. The most common side effects include ectopic beats, nausea, and vomiting. B. Angina has been reported following treatment. C. Tachycardia and arrhythmias are less likely than with other catecholamines. D. Can precipitate hypertensive crisis in susceptible individuals (i.e. patients on MAO inhibitors
such as Parnate, Nardil or Marplan).
E. Consider hypovolemia and treat this with appropriate fluids before administration of Dopamine. F. Dopamine is best administered by infusion pump if available. Monitor closely.
ADULT DOSING
INDICATION DOSE ROUTE NOTES
Shock (excluding
5 mcg/kg/min IV/IO drip. Increase by 5
mcg/kg/min every 5 minutes to max of 20
mcg/kg/min or until desired effect is achieved.
PEDIATRIC DOSING (same as adult)
EPINEPHRINE (Adrenalin®)
OLMC REQUIRED: For additional doses.
SUPPLIED: 1:10,000 – 1 mg / 10 mL pre-filled syringe; 1:1,000 – 30 mg / 30 mL vial; 1 mg/mL amps
PHARMACOLOGY AND ACTIONS:
Epinephrine is a catecholamine with both alpha and beta effects. In general the following cardiovascular
responses can be expected: increased heart rate, increased myocardial contractile force, increased systemic vascular resistance, increased arterial blood pressure, increased myocardial oxygen consumption, increased automaticity. Epinephrine is also a potent bronchodilator.
INDICATIONS:
Epinephrine is indicated in the following situations: VF, asystole, pulseless electrical activity, pulseless
VT, systemic allergic reactions, asthma, children 6 months to 6 years with audible stridor at rest.
CONTRAINDICATIONS: None
A. Epinephrine increases cardiac work load and can precipitate angina, MI, or major dysrhythmias in
individuals with ischemic heart disease.
B. Wheezing in an elderly person is pulmonary edema or pulmonary embolus until proved
SIDE EFFECTS AND NOTES:
A. IM administration is preferred over subcutaneous injection. B. May cause anxiety, tremor and headache. C. Cardiac side effects include tachycardia, PVCs, angina and hypertension.
ADULT DOSING
INDICATION DOSE ROUTE
VF/pulseless VT, Asystole,
I mg 1:10,000 IV/IO
PEA Anaphylaxis, asthma
0.3 mg 1:1000 IM or
For patients over 40 y/o
0.3 mg 1:10,000 IV/IO
with known cardiac history, 0.15 mg
PEDIATRIC DOSING
INDICATION DOSE
VF/ pulseless VT, Asystole,
0.01 mg/kg 1:10,000
Repeat every 3-5
PEA, Bradycardia
0.01 mg/kg 1:1000 IM to a
Contact OLMC for
Anaphylaxis, asthma
max single dose of 0.3 mg
additional doses in
Respiratory distress –
5 mL of 1:1000 via nebulizer
Contact OLMC for
audible stridor at rest 6 mo
additional doses
ETOMIDATE (Amidate®)
OLMC REQUIRED: None
SUPPLIED: Varies
PHARMACOLOGY AND ACTIONS:
Etomidate is a hypnotic drug without any analgesic activity. Intravenous injection of Etomidate produces
hypnosis characterized by rapid onset of action; usually within one minute. Duration of hypnosis is dose dependent but relatively brief, usually 3-5 minutes.
INDICATIONS:
As an induction agent for use in rapid sequence intubation.
CONTRAINDICATIONS:
Etomidate is contraindicated in patients who have a known hypersensitivity to the drug.
PRECAUTIONS:
Excessively rapid injection may cause a fall in blood pressure.
SIDE EFFECTS AND NOTES:
A. The most frequent adverse reactions are transient venous pain on injection and transient
skeletal muscle movements.
B. Etomidate may also cause nausea and/or vomiting.
ADULT DOSING
INDICATION DOSE ROUTE NOTES
Induction agent for RSI
0.3 mg/kg injected
PEDIATRIC DOSING (same as adult)
FENTANYL (Sublimaze®)
OLMC REQUIRED: None
SUPPLIED: 100 micrograms / 2 mL vial
PHARMACOLOGY AND ACTIONS:
Fentanyl is a potent synthetic opioid analgesic that produces analgesia and sedation. It is about 50-100
times more potent than Morphine on a weight basis. Onset of action when given is 2-3 minutes. Peak effect occurs at 3-5 minutes and lasts 15-45 minutes.
A. Pain. B. Suspected ischemic chest pain unresponsive to Nitroglycerin.
A. Known allergy to Fentanyl.
B. A systolic blood pressure of less than 100 mm/Hg. C. Respiratory rate less than 14 breaths per minute, oxygen saturation of less than 90%, or
significant respiratory depression. For pediatric patients, vital signs should be maintained within normal age-appropriate limits.
A. Fentanyl can cause respiratory depression that is reversible with Naloxone. Respiratory
depression can also be exacerbated by underlying lung disease and the use of other respiratory depressant drugs (benzodiazepines, alcohol, cyclic antidepressants). Have Naloxone and
respiratory support available when administering Fentanyl.
B. If administered rapidly and in very large doses, Fentanyl can cause muscle spasm and chest wall
rigidity. The only reliable treatment for this is neuromuscular blockade.
C. The action of Fentanyl is prolonged and its elimination is slower in the elderly. Smaller
maintenance doses are advisable.
D. Fentanyl must be used cautiously in patients who have already received Morphine for
prehospital analgesia.
SIDE EFFECTS AND NOTES:
A. If hypotension develops, it is usually responsive to Naloxone administration. If hypotension
continues, follow Shock protocol.
B. Check and document vital signs and patient response after each dose. C. The goal of Fentanyl administration is patient comfort, not the total elimination of pain but the
reduction in the perception of pain by the patient.
ADULT DOSING
INDICATION DOSE ROUTE NOTES
50-100 mcg IV/IO/IM/IN, repeat 25-50 mcg q 3-5 min as needed to a max of 400 mcg. If additional amounts required, contact OLMC.
PEDIATRIC DOSING
INDICATION DOSE ROUTE NOTES
1 mcg / kg IV/IO/IM/IN, repeat with 0.5-1 mcg/kg q 3-5 min as needed to a max of 4 mcg/kg. Do not exceed adult dosing.
GLUCAGON
OLMC REQUIRED: Beta blocker overdose
SUPPLIED: 1 mg vial of powder / 1 mL vial of dilutent
PHARMACOLOGY AND ACTIONS:
Glucagon is a hormone that causes glucose mobilization in the body. It works opposite to insulin, which
causes glucose storage. It is released at times of insult or injury when glucose is needed and mobilizes glucose from body glycogen stores. Return to consciousness should be within 20 minutes of an IM dose if patient is hypoglycemic.
A. Known hypoglycemia (preferably demonstrated by blood glucose determination) when patient is
confused or comatose and dextrose is not available or an IV cannot be started.
B. Possible Beta blocker overdose per OLMC.
CONTRAINDICATIONS:
None.
IV Dextrose is the treatment of choice for hypoglycemia in the patient who cannot tolerate oral glucose. The use of Glucagon is restricted to patients who are seizing, comatose, combative, or with collapsed veins and in whom an IV cannot be started.
SIDE EFFECTS AND NOTES:
A. Nausea and vomiting may occur with administration. B. Persons with no liver glycogen stores (malnutrition, alcoholism) may not be able to mobilize any
glucose in response to Glucagon.
ADULT DOSING
INDICATION DOSE ROUTE NOTES
Beta blocker overdose
1 mg IV/IO; contact OLMC for additional dosing.
PEDIATRIC DOSING
INDICATION DOSE ROUTE NOTES
0.02 mg / kg IM to a maximum of 1 mg
Beta blocker overdose
Contact OLMC for dosing.
ORAL GLUCOSE
OLMC REQUIRED: None
SUPPLIED: 15 grams glucose in 37.5 grams of gel tube
PHARMACOLOGY AND ACTIONS:
Glucose is the body's basic fuel and it produces most of the body's quick energy. Its use is regulated by
insulin that stimulates storage of excess glucose from the bloodstream and glucagon that mobilizes stored glucose into the bloodstream.
INDICATIONS:
Oral glucose is indicated in the conscious patient where a suspicion of hypoglycemia exists or a blood
glucose measurement indicates a low blood glucose level.
CONTRAINDICATIONS:
Do not give to patients who cannot adequately protect their own airway.
To give solutions orally, a patient must be continually assessed for the ability to protect his or her own airway.
SIDE EFFECTS AND NOTES:
A. Research suggests that hyperglycemia may complicate, or worsen, a number of medical
conditions (i.e. myocardial infarction, stroke). Oral glucose should be given to a conscious patient whenever hypoglycemia is documented by blood glucose meter. If these objective findings are not available, the EMT should use judgment based on signs and history.
B. Effects will be delayed in the elderly and people with poor circulation. C. May be more tolerable if administered with liquid between dosages. D. Patient's condition may require more than one dose of oral glucose.
ADULT DOSING
INDICATION DOSE ROUTE NOTES
PEDIATRIC DOSING (same as adult)
HALOPERIDOL (Haldol®)
OLMC REQUIRED: None
SUPPLIED: 5 mg / 1 mL vial
PHARMACOLOGY AND ACTIONS:
Haldol is the first of the Butyrophenone series of major tranquilizers. Has pharmacological property
similar to the phenothiazine class of drugs. Haldol is a major sedative. Haldol is also an antipsychotic drug of high-potency, strong tranquilizer.
INDICATIONS:
For use as chemical restraint for managing uncontrollable, violent or acutely agitated patients when
other means prove ineffective.
A. Severe CNS depression
B. Patients with history of drug OD C. Patients with Seizure disorder
D. Known hypersensitivity to drugs in Butyrophenone family E. Patients with systolic BP less than 100 mm Hg
PRECAUTIONS:
Patients at risk for cardiac dysrhythmias (e.g. history of cocaine use, amphetamine use or cardiac
history/event or seizures) should not receive Haldol. Institute continuous cardiac monitoring as soon as safe to detect dysrhythmias and QT prolongation.
Treat ventricular tachy-dysrhythmias according to relevant protocols.
SIDE EFFECTS AND NOTES:
A. Dosage should be reduced in setting of impaired hepatic or renal functions, the elderly and
debilitated patients.
B. Monitor for hypotension and hyperthermia
C. Be prepared to treat seizures.
ADULT DOSING
INDICATION DOSE ROUTE NOTES
Chemical restraint
2.5-5 mg IV/IO/IM to max of 10 mg
PEDIATRIC DOSING (DO NOT USE)
SFD SWAT-MEDIC ONLY HEMOSTATIC AGENT
OLMC REQUIRED: None
SUPPLIED: Various sizes of bandages
PHARMACOLOGY AND ACTIONS:
The hemostatic agent bandages have a positive charge and attracts red blood cells which have a negative
charge. The red blood cells create a seal over the wound as they are drawn into the bandage, forming a
very tight adherent seal. By quickly sealing the wound, the bandage reduces blood loss, and thus demand
for blood transfusion products such as red blood cells or plasma. These bandages are cleared with an
antibacterial barrier protecting from a wide range of harmful organisms, including the antibiotic resistant
Staphylococcus aureus (MRSA), Enterococcus faecalis (VRE) and Acinetobacter baumannii.
INDICATIONS:
The hemostatic dressing is for the external temporary control of severely bleeding wounds intended for
emergency use. In addition, the hemostatic bandage also controls bleeding following hemodialysis.
CONTRAINDICATIONS:
A. There are no known contraindications for use of hemostatic bandages. B. There have been no known allergic reactions (shellfish allergies).
A. Be cautious around ocular injuries, but do not withhold for uncontrolled bleeding. B. Do not save unused product; reuse will cause risk of infection and loss of efficacy.
SIDE EFFECTS AND NOTES:
Slightly overlap the bandages when the wound is larger than the bandage itself.
Bandages may be cut or torn to fit the application and/or wound site.
TREATMENT:
A. Monitor vital signs. B. Prepare patient for rapid transport. C. Treat for hypovolemic shock including a 500 cc fluid bolus. May repeat to keep a normal
mental status and a systolic BP of 80.
1. Use direct pressure to control bleeding. 2. Use pressure dressing to control bleeding. 3. If these fail to control bleeding use a tourniquet on extremity wounds. 4. Apply hemostatic agent to wounds in conjunction with tourniquet or to wounds
where a tourniquet is unable to be applied.
A. Wipe away excess blood from wound. B. Place hemostatic agent in the wound. C. Completely cover the wound and apply direct pressure for at least 2 minutes. D. If possible, bring in hemostatic packaging to assist hospital staff with removal
HYDROXOCOBALAMIN (Cyanokit®)
OLMC REQUIRED: Second 5 gram dose
SUPPLIED: 2.5 grams powder in vial for reconstitution with 100 mL NS (kit has two vials)
PHARMACOLOGY AND ACTIONS:
Hydroxocobalamin (vitamin B12a) is an effective antidote in the treatment of cyanide poisoning based on
its ability to bind cyanide ions. Each Hydroxocobalamin molecule can bind one cyanide ion to form cyanocobalamin (vitamin B12), which is then excreted in the urine. Cyanide is an extremely potent toxic poison. In the absence of rapid and adequate treatment exposure to a high dose of cyanide can
result in death within minutes due to inhibition of cytochrome oxidase resulting in arrest of cellular respiration.
Cyanide poisoning or smoke inhalation with suspected cyanide poisoning due to the presence of coma, persistent hypotension or cardiorespiratory arrest.
Do not administer Hydroxocobalamin and Sodium Thiosulfate to the same patient.
PRECAUTIONS:
Hydroxocobalamin has physical (particulate) and chemical incompatibilities with many medications and it
is best to administer other drugs or products (e.g. blood) through a separate intravenous line.
SIDE EFFECTS AND NOTES:
A. The most frequently occurring side effects are chromaturia (red colored urine) and erythema
(skin redness) which occur in nearly all patients.
B. Other reported serious side effects include allergic reactions, temporary increases in blood
pressure, nausea, headache and infusion site reactions.
D. Because of its deep red color, Hydroxocobalamin has also been found to interfere with certain
laboratory tests based on light absorption including co-oximetric measurements or carboxyhemoglobin, methemoglobin and oxyhemoglobin.
ADULT DOSING
Each 2.5 gram vial should be reconstituted with 100 mL of Normal Saline
INDICATION DOSE
Cyanide poisoning or smoke
Monitor for clinical
inhalation with suspected
5 grams IV or IO over 15 minutes. Contact
cyanide poisoning (See
OLMC regarding second 5 gram dose
Indications above)
PEDIATRIC DOSING (DO NOT USE)
Each 2.5 gram vial should be reconstituted with 100 mL of Normal Saline
INDICATION DOSE
Cyanide poisoning or smoke
Monitor for clinical
inhalation with suspected
70 mg/kg IV or IO over 15 minutes.
cyanide poisoning (See
Contact OLMC regarding second dose.
Indications above)
IPRATROPIUM BROMIDE (Atrovent®)
OLMC REQUIRED: None
SUPPLIED: 0.5 mg / 2.5 mL vial
PHARMACOLOGY AND ACTIONS:
Ipratropium is an atropine derivative used for inhalation therapy. For severe asthma, Ipratropium taken
in addition to a short acting beta agonist (such as Albuterol) can provide greater bronchodilation and clinical benefit than the beta agonist alone. It has no anti-inflammatory effects and does not decrease bronchial hyper-responsiveness.
INDICATIONS:
As a supplement to Albuterol in patients with asthma and COPD.
CONTRAINDICATIONS:
Do not use in patients with severe glaucoma.
Ipratropium in the meter dose inhaler and auto-inhaler formulations should not be administered to individuals allergic to soy lecithin or related food products (e.g. soy beans, peanuts). The nebulized
formulation may be administered to these patients.
SIDE EFFECTS AND NOTES:
A. Dry mouth B. Pharyngeal irritation C. Increased intra-ocular pressure in glaucoma patients
ADULT DOSING
INDICATION DOSE ROUTE
Combine with doses of
Albuterol. Max 2 doses.
PEDIATRIC DOSING (Same as adult)
KETAMINE HYDROCHLORIDE
OLMC REQUIRED: None
SUPPLIED: 100 mg/mL 5 mL vial
PHARMACOLOGY AND ACTIONS:
A dissociative anesthetic causing a trance-like state.
The only anesthetic producing: analgesia/pain relief, hypnosis and sleep, amnesia (short-term memory loss).
INDICATIONS:
A second-line induction agent indicated when Etomidate is unavailable or contraindicated.
Excited delirium patients
A. Brain masses or hydrocephalus.
B. Acute globe injury or glaucoma (increases intraocular pressure). C. Patients with schizophrenia.
D. When significant increase in BP might prove harmful as in aortic dissection, acute myocardial
infarction, angina, intracranial hemorrhage.
A. Ketamine will increase blood pressure and heart rate.
B. Like all induction agents, Ketamine should be administered before paralytics.
SIDE EFFECTS AND NOTES:
A. Many patients sedated with Ketamine do not close their eyes. B. Produces dose-related increase of heart rate and blood pressure. C. Produces a significant vagolytic response; this partially accounts for its ability to cause bronchial
smooth muscle relaxation.
ADULT DOSING
INDICATION DOSE ROUTE
RSI – induction agent
Administer over 1 minute.
PEDIATRIC DOSING (Same as adult)
LIDOCAINE (Xylocaine®)
OLMC REQUIRED: See contraindications
SUPPLIED: 100 mg / 5 mL of 2% solution in pre-filled syringe
PHARMACOLOGY AND ACTIONS:
Lidocaine depresses the automaticity of Purkinje fibers, raising stimulation threshold in the ventricular
muscle fibers which makes the ventricles less likely to fibrillate. It has little anti-arrhythmic effect on the atrial muscle in normal doses. The effect of a single bolus on the heart disappears in 10-20 minutes due to redistribution in the body. Metabolic half-life is about 2 hours.
A. Recurrent ventricular fibrillation B. Stable ventricular tachycardia or recurrent ventricular tachycardia if clinical condition is not
rapidly deteriorating
C. Following successful defibrillation or cardioversion from ventricular fibrillation or ventricular
D. PVCs in a suspected ischemic event E. Pain management after IO placement
Lidocaine is contraindicated in the following perfusing pts situations without OLMC approval: A. Systolic BP is less than 90 mmHg
B. Heart rate is less than 50 beats per minute C. Periods of sinus arrest are present D. Second or Third degree heart blocks are present
A. Lidocaine is not recommended in the treatment of supra-ventricular arrhythmias. B. If administering maintenance dosing and the patient begins seizing, stop the Lidocaine dosing and
treat per Seizure protocol.
C. All Lidocaine doses after the initial bolus must be reduced to 0.375 mg/kg (1/4 of the initial
dose) in patients with CHF, shock, hepatic disease, or in patients greater than 70 y/o.
SIDE EFFECTS AND NOTES:
A. CNS side effects include sleepiness, dizziness, disorientation, confusion, and convulsions. B. Hypotension C. Toxicity is more likely in elderly patients (over 70 yrs old).
ADULT DOSING
INDICATION DOSE
Recurrent VF, Stable VT,
Bolus:1.5 mg/kg to a max of
See Side Effects above for
3 mg/kg maintenance; 0.75
when to reduce dosing.
mg/kg every 10 minutes
Pain management after IO
0.5 mg/kg to a max of 50 mg
Administer and wait 30-60
seconds before flushing with
PEDIATRIC DOSING (Same as adult)
MAGNESIUM SULFATE
OLMC REQUIRED: Pediatric dosing for asthma
SUPPLIED: 1 gram (50%) / 2 mL vial
PHARMACOLOGY AND ACTIONS:
Magnesium is a cation that is present in human cells and intercellular fluids. It acts as an anti-arrhythmic
agent and may convert ventricular fibril ation and ventricular tachycardia.
A. In cardiac arrest after defibrillation, Epinephrine, Lidocaine and Amiodarone in the treatment of
ventricular fibrillation and pulseless ventricular tachycardia.
B. For the treatment of seizures in women with pre-eclampsia/eclampsia. C. In severe asthma as a smooth muscle relaxant and inhibitor of histamine.
CONTRAINDICATIONS: None
In the non-arrest patient, Magnesium Sulfate may cause hypotension, bradycardia, decreased reflexes and respiratory depression.
SIDE EFFECTS AND NOTES:
See precautions.
ADULT DOSING
INDICATION DOSE
Ventricular fibrillation/tachycardia 2 grams over 1-2 minutes IV/IO
4 grams over 10 minutes IV/IO via Buretrol
4 grams over 10 minutes IV/IO via Buretrol
PEDIATRIC DOSING
Ventricular fibrillation/tachycardia 25-50 mg/kg over 1-2 minutes IV / IO. Max dose of 2 grams.
Contact OLMC for use in this situation. Usual dose is 25-50 mg/kg over 10 min. Max 2 grams.
25-50 mg/kg over 10 min IV / IO Max dose 2 grams.
DILUTING FOR IV ADMINISTRATION
Dilute each gram (2 mL) of Magnesium Sulfate in 8 mL of Normal Saline. (Example: 1 gram (2 cc) – mix in 8 ccs of NS; 2 grams (4 cc) – mix in 16ccs of NS; 4 grams (8 cc) – mix in 32 ccs of NS)
MIDAZOLAM (Versed®)
OLMC REQUIRED: None
SUPPLIED: 10 mg / 2 mL vial or 5 mg / 2 mL vial
PHARMACOLOGY AND ACTIONS:
Midazolam is a benzodiazepine with potent sedative, anti-anxiety and anticonvulsant properties. It also
causes significant anterograde amnesia when administered IV.
A. Status seizure (any seizure that has lasted longer than 2 minutes or two consecutive seizures
without regaining consciousness)
B. To relieve anxiety and produce amnesia during cardioversion, pacing or paralytic intubation.
CONTRAINDICATIONS:
In seizures, do not give unless patient is actively seizing.
PRECAUTIONS:
Midazolam causes respiratory depression and/or hypotension especially if administered rapidly. Monitor
patient closely.
SIDE EFFECTS AND NOTES:
A. Common side effects include drowsiness, hypotension, respiratory depression and apnea. These
are like more likely to occur in the very young and the elderly. Rarely, patients may experience paradoxical agitation.
B. Respiratory depression is more likely in patients who have taken other CNS depressant drugs
such as opioids, alcohol and barbiturates or when given rapidly.
C. Midazolam is metabolized in the liver and excreted by the kidney. Doses should be adjusted
accordingly in patients with underlying hepatic or renal diseases and low flow states such as congestive heart failure.
ADULT DOSING
INDICATION DOSE
Seizures, pacing, restraint
2-5 mg IV/IN/IO/IM
May repeat once.
Post-Intubation Sedation
1-2 mg IV/IO/IN or 2 mg IM
PEDIATRIC DOSING
Seizures, pacing
0.1 mg/kg IV/IO/IN to a max of 2.5 mg
May repeat once after 5
minutes. Call OLMC for
additional doses.
Post-Intubation Sedation
0.1 mg/kg IV/IO to a max of 2.5 mg
MORPHINE SULFATE
OLMC REQUIRED: None
SUPPLIED: Varies
PHARMACOLOGY AND ACTIONS:
Morphine is a narcotic analgesic that induces drowsiness, mental clouding and mood changes. It also
increases venous capacitance, decreases venous blood return (preload) and reduces systemic vascular resistance at the arteriolar level (afterload). This may lead to decreases in myocardial oxygen demand. Onset of action when given IV is 2-3 minutes and peak effect occurs at 7-10 minutes. Duration is 3-5
A. Suspected ischemic chest pain unresponsive to nitroglycerin.
B. Pain due to burns or musculoskeletal injury.
A. Known allergy to morphine or sulfates (Sulfa drugs are not sulfates.)
B. Blood pressure less than 100 mmHg systolic. C. Respiratory rate less than 14 breaths per minute or oxygen saturation less than 90%. For
pediatric patients, vital signs should be maintained within the normal age-appropriate range.
A. Morphine causes respiratory depression that is reversible with Naloxone. This respiratory
depression is exacerbated by underlying lung disease (COPD, etc.) and other depressant drugs
(Valium, alcohol, cyclic anti-depressants). Naloxone and respiratory support must be available when using Morphine.
B. If hypotension develops it is usually responsive to Naloxone administration. If hypotension
persists, follow Shock protocol.
C. Use caution in patients with trauma or pain of the head or abdomen.
SIDE EFFECTS AND NOTES:
A. The goal of Morphine administration is patient comfort (not the total elimination of pain but
reduction in perception of pain by the patient).
B. Morphine is a Schedule II controlled substance. Follow your agency's Controlled Substance
policy or procedure for control and monitoring of use.
ADULT DOSING
INDICATION DOSE
ROUTE NOTES
2-5 mg, repeat as
Maximum dose is 20 mg
PEDIATRIC DOSING
0.1 mg/kg, repeat as
Do not exceed adult dosing.
NALOXONE (Narcan®)
OLMC REQUIRED: None
SUPPLIED: Varies
PHARMACOLOGY AND ACTIONS:
Naloxone is an opioid antagonist which competitively binds to opioid receptor sites but which exhibits
almost no pharmacologic activity of its own. Duration of effect is 1-4 hours.
A. Reversal of opioid effects, particularly respiratory depression, due to opioid drugs either
ingested or injected or administered in the course of treatment. Opioid drugs include Fentanyl, Morphine, Demerol, Dilaudid, Percodan, Codeine.
B. Diagnostically in coma of unknown etiology to rule out or reverse opioid depression.
Do not use in neonates.
A. In patients physically dependent on opioids, violent withdrawal symptoms may occur. Be
prepared to restrain the patient.
B. Some opioid intoxications may require up to 8 mg of Naloxone to reverse symptoms (e.g.
Methadone, designer drugs).
SIDE EFFECTS AND NOTES:
A. The duration of some opioids is longer than Naloxone, repeat doses may be necessary.
Monitor the patient closely. Patients who have received Naloxone must be transported to the
hospital because coma may reoccur when Naloxone wears off.
B. Side effects are rare. Do not hesitate to use if indicated. C. If no effect is seen from Naloxone administration, consider other causes of coma.
ADULT DOSING
INDICATION DOSE
ROUTE NOTES
Reversal of opioid effects,
0.5 mg IV/IO/IN. Repeat every 2 minutes up to 2 mg titrating to
coma of unknown etiology respirations. If no IV/IO, give 2 mg IM.
If no response to initial dose, may repeat at 2 mg every 5 min (IV/IO/IN or IM) up to a maximum of 8 mg.
PEDIATRIC DOSING
Reversal of opioid effects,
0.1 mg/kg IV/IO/IM/IN up to 2 mg. May repeat every 3-5 minutes up to 2
coma of unknown etiology mg/dose. Do not exceed adult dosing. Do not use in neonates.
OLMC REQUIRED: See contraindications
SUPPLIED: 0.4 mg metered dose spray, 0.4 mg tablets in dark bottle
PHARMACOLOGY AND ACTIONS:
Nitroglycerin is an organic nitrate and is a vasodilating agent. Its cardiovascular effects include: reduced
venous tone (causing pooling of blood in the peripheral veins and decreased return of blood to the heart), decreased peripheral resistance, and dilation of coronary arteries. It also is a general smooth muscle relaxant.
A. Chest pain thought to be related to cardiac ischemia B. Pulmonary edema
A. Blood pressure less than 100 mmHg systolic. B. Patients who have taken Viagra® (sildenafil citrate) or Levitra® (vardenafil HCl) within 24 hours,
or who have taken Cilais® (tadalafil) within 48 hours. Contact OLMC for direction.
A. Generalized vasodilatation may cause profound hypotension and reflex tachycardia. B. IV should be established prior to administration in patients who have not taken Nitroglycerin
previously or who have a potential for hemodynamic instability.
C. Use with caution in patients with borderline blood pressure or with inferior/right side MI.
SIDE EFFECTS AND NOTES:
A. Common side effects are headache, flushing or dizziness. B. Because nitroglycerin causes generalized smooth muscle relaxation, it may be effective in
relieving chest pain caused by esophageal spasm.
ADULT DOSING
INDICATION DOSE
ROUTE NOTES
Chest pain/pulmonary edema
0.4 mg SL every 5 minutes as long as systolic BP is greater than 100 mmHg
PEDIATRIC DOSING (Contact OLMC.)
ONDANSETRON (Zofran®)
OLMC REQUIRED: Patients less than two years old
SUPPLIED: 4 mg/2 mL vial, 4 mg oral tablet, 4 mg/2 mL pre-filled syringe
PHARMACOLOGY AND ACTIONS:
Ondansetron is a potent, highly selective serotonin (5-HT3) receptor agonist. Its precise mode of
action in the control of nausea is not known. Pharmacologic agents and other triggers may cause release of 5-HT3 receptors. Ondansetron blocks the initiation of this reflex. Ondansetron is commonly used in the treatment of nausea in patients who are receiving chemotherapy or as a postoperative
nausea treatment. Peak plasma concentrations of the drug occur 10 minutes after IV administration, and 40 minutes after IM injection. Both routes have the same elimination half life of 4 hours.
Prevention and control of uncomplicated nausea and vomiting
CONTRAINDICATIONS:
Known hypersensitivity to Zofran or similar medications
A. Hypersensitivity reactions have been reported in patients who have exhibited hypersensitivity to
other 5-HT3 medications (Anzemet®, Kytril®)
B. Patients with bowel obstruction should be monitored closely following administration.
C. Ondansetron may precipitate if mixed with alkaline solutions. D. Monitor ECG for signs of QT prolongation after administration.
SIDE EFFECTS AND NOTES:
A. The most common side effects include headache, dizziness, drowsiness and shivers. B. Body aches, agitation, dysuria, hypotension and rash have also been reported in a very small
number of patients.
ADULT DOSING
INDICATION DOSE ROUTE NOTES
Give slowly over two
minutes if giving IV/IO.
PEDIATRIC DOSING
For ages 2 and older
Give slowly over two
minutes if giving IV/IO.
Do not exceed 4 mg.
Contact OLMC for use in patients under 2 years old except for children in spinal immobilization or children receiving chemotherapy. In this instance, give 2 mg IV/IO/IM in children between 6 mos-2 yrs.
OLMC REQUIRED: None
SUPPLIED: Various (D cylinder contains 415 liters at 2,000 psi)
PHARMACOLOGY AND ACTIONS:
Oxygen added to the inspired air raises the amount of oxygen in the blood and the amount delivered to
the tissues. Breathing in most patients is regulated by small changes in acid/base balance and CO2 levels
and it takes a large drop in oxygen concentration to stimulate respiration.
A. Suspected hypoxemia or respiratory distress from any cause B. Acute chest pain in which cardiac ischemia or myocardial infarction is suspected C. Shock from any cause D. Major trauma E. Carbon monoxide poisoning
CONTRAINDICATIONS: None
A. If the patient is not adequately breathing, the appropriate treatment is ventilation with oxygen –
not just supplemental oxygen.
B. In a small percentage of patients with chronic lung disease, administration of oxygen will
decrease respiratory drive. Do not withhold oxygen because of this possibility. Be prepared to assist ventilations if needed.
SIDE EFFECTS AND NOTES:
A. Non humidified oxygen is drying and irritating to mucous membranes. B. Restlessness may be an important sign of hypoxia. C. Oxygen toxicity is not a risk in acute administration. D. Nasal cannula prongs work equally well on nose and mouth breathers.
ADJUNCT FLOW INSPIRED
2-6 liters per minute
Non-rebreather mask
10-15 liters per minute
15-25 liters per minute
PRALIDOXIME CL (Protopam/2-PAM®)
FOR USE BY HAZMAT TEAM OR CHEMPACK
OLMC REQUIRED: For IV/IO use
SUPPLIED: 600 mg/2 mL auto-injector, 1 gram powder vial – reconstitute with 20 mL NS
PHARMACOLOGY AND ACTIONS:
The principal action of Pralidoxime is to reactivate cholinesterase which has been inactivated by an
organophosphate pesticide or related compound. The drug's most critical effect is in relieving paralysis
of respiratory muscles. Atropine is always required concurrently to block the effect of acetylcholine.
A. As an antidote in the treatment of poisoning due to organophosphate pesticides and chemicals.
B. Control of overdose by anticholinesterase drugs (e.g. treatment of myasthenia gravis).
CONTRAINDICATIONS:
None in the emergency setting.
A. Rapid IV injection may cause tachycardia, laryngospasm, muscle rigidity and transient
neuromuscular blockade. Administration should be done slowly and preferably by infusion.
B. Pralidoxime is a relatively short acting drug; repeat dosing may be necessary.
SIDE EFFECTS AND NOTES:
Dizziness, blurred vision, diplopia, headache, drowsiness, nausea, tachycardia and muscle weakness have
been reported following administration.
ADULT DOSING
INDICATION DOSE
ROUTE NOTES
Refer to Haz-Mat Protocol – Organophosphate Poisoning for dosing.
PEDIATRIC DOSING
Refer to Haz-Mat Protocol – Organophosphate Poisoning for dosing.
PROPARACAINE (Alcaine®)
OLMC REQUIRED: See notes on Adult Dosing below.
SUPPLIED: 0.5% solution in 15 mL bottle
PHARMACOLOGY AND ACTIONS:
Proparacaine hydrochloride is a short-acting local anesthetic of the ester type with an onset of action
within 30 seconds. Duration is up to 15 minutes.
INDICATIONS:
Superficial foreign bodies or chemical burns to the eye.
A. Known hypersensitivity to any component of the solution. B. Penetrating eye injury
PRECAUTIONS:
Systemic effects are rare with topical use.
SIDE EFFECTS AND NOTES:
Instillation of Proparacaine in the recommended concentration and dosage produces little or no initial
irritation, stinging or burning, these effects may occur several hours after use.
ADULT DOSING
INDICATION DOSE
ROUTE NOTES
Foreign body or burn to eye
One drop in the affected for initial anesthetic effect. If effect is not felt within one minute, three additional one drop doses may be given at one-minute intervals. If no anesthetic effect is felt after the fourth drop, consult OLMC. For transports longer than 15 minutes, if eye pain returns, 1-4 additional drops may be given as previously done to continue anesthetic effect. Contact OLMC for transports greater than 30 minutes.
PEDIATRIC DOSING (Same as adult)
ROCURONIUM (Zemuron®)
OLMC REQUIRED: None
SUPPLIED: 10 mg/mL, 10 mL or 5 mL vials
PHARMACOLOGY AND ACTIONS:
Rocuronium is a non-depolarizing neuromuscular blocking agent causing skeletal muscle relaxation.
Neuromuscular blockade occurs within 1-2 minutes. Time to recovery is 30-60 minutes.
A. For sustained neuromuscular blockade in the intubated patient.
B. As the first line agent for Rapid Sequence Induction in the patient where Succinylcholine is
contraindicated.
CONTRAINDICATIONS:
Known sensitivity to Rocuronium
A. Rocuronium has no affect on consciousness and must be used with a sedative or induction
B. Patients with renal or hepatic failure may experience prolonged paralysis. C. Due to the prolonged duration of action, the endotracheal tube or pharyngeal tube must be
continually monitored to ensure correct placement with ETCO2 and pulse oximetry.
SIDE EFFECTS AND NOTES:
Rocuronium can be used to maintain paralysis even if intubation was performed without Succinylcholine.
ADULT DOSING
INDICATION DOSE
ROUTE NOTES
Maintenance of post-intubation
PEDIATRIC DOSING (Same as adult)
SODIUM BICARBONATE
OLMC REQUIRED: Tri-cyclic anti-depressant overdose, crush injury, hyperkalemia
SUPPLIED: 50 mEq/50 mL pre-filled syringe
PHARMACOLOGY AND ACTIONS:
Sodium Bicarbonate is an alkalotic solution which neutralizes acids found in the blood. Acids are
increased in the blood when body tissues become hypoxic due to cardiac or respiratory arrest. Acidosis depresses cardiac contractility and cardiac response to catecholamines, and makes the heart more likely to fibrillate and less likely to defibrillate. In the non-perfusing patient Sodium Bicarbonate
has been shown to increase the intracellular acidosis and worsen acid/base balance, thus it is not recommended in the routine cardiac arrest sequence.
A. To control arrhythmias or asystole in cyclic antidepressant overdose or hyperkalemia. B. Acidosis caused by prolonged cardiac arrest.
A. Addition of too much bicarbonate may result in alkalosis that is difficult to reverse and may
cause as many problems in resuscitation as acidosis.
B. May increase cerebral acidosis, especially in diabetics who are ketotic. C. Do not mix Sodium Bicarbonate with calcium preparations. Slowly flush one drug from the
catheter before administering the other.
SIDE EFFECTS AND NOTES:
Each amp of Sodium Bicarbonate contains 50 mEq of sodium. This may increase intravascular volume
and hyperosmolarity resulting in cerebral impairment.
ADULT DOSING
INDICATION DOSE ROUTE NOTES
Cyclic antidepressant
1 mEq/kg IV or IO
V-Fib/Pulseless VT, asystole
1 mEq / kg IV or IO, may repeat every 10 minutes at 0.5 mEq / kg
Entrapment/Crush injury
Mix 100 mEq in 1000 cc of NS; administer 200 cc/hour
PEDIATRIC DOSING (Same as adult)
Use same dosing as for adult with exception of hyperkalemia, call OLMC for dosing in that situation. For children less than 10 kg (1 yr), dilute by one-half with Normal Saline prior to administration.
SODIUM THIOSULFATE 25%
OLMC REQUIRED: All situations
SUPPLIED: 12.5 grams/50 mL vial
PHARMACOLOGY AND ACTIONS:
Sodium Thiosulfate is used as an antidote for cyanide poisoning. The primary mechanism of cyanide
detoxification involves the conversion of cyanide to the thiocyanate ion, which is relatively non-toxic. This reaction involves the enzyme rhodanese which is found in many body tissues but with the major activity in the liver. The body has the capability to detoxify cyanide; however, the rhodanese enzyme
system is slow to respond to large amounts of cyanide. The rhodanese enzyme reaction can be accelerated by supplying an exogenous source of sulfur. This is commonly accomplished by
administering Sodium Thiosulfate.
INDICATIONS:
Cyanide poisoning
None in cyanide poisoning
A. Sodium Thiosulfate is essentially non-toxic. However, some animal studies showed that a
constant infusion of Sodium Thiosulfate led to hypovolemia which was considered due to an
B. It is not known whether Sodium Thiosulfate can cause fetal harm when administered to a
pregnant woman and should only be administered in this setting if clearly needed.
SIDE EFFECTS AND NOTES:
Sodium Thiosulfate is administered as a slow push over 10 minutes. Consider using a Buretrol or similar
device.
ADULT DOSING
INDICATION DOSE ROUTE NOTES
Cyanide poisoning
50 mL slow IV over 10-20 minutes
PEDIATRIC DOSING
INDICATION DOSE ROUTE NOTES
Cyanide poisoning
1.65 mL/kg slow IV over 10-20 minutes
SUCCINYLCHOLINE (Anectine®)
OLMC REQUIRED: None
SUPPLIED: 200 mg/10 mL vial
PHARMACOLOGY AND ACTIONS:
Succinylcholine is a short acting motor nerve depolarizing skeletal muscle relaxant. It competes with
acetylcholine to combine with cholinergic receptors in the motor end plate causing depolarization inhibiting neuromuscular transmission. After intravenous injection paralysis is obtained within 1-2 minutes and persists for approximately 4-6 minutes. Effects then start to fade and return to normal. It
has no effect on consciousness. Muscle relaxation begins in the eyelids and jaw, then progresses to the limbs, abdomen, diaphragm and finally intercostals muscles. Succinylcholine is hydrolyzed by plasma
pseudocholinesterase and is excreted by the kidneys.
INDICATIONS:
To achieve temporary paralysis where endotracheal intubation is indicated.
A. Hypersensitivity to the drug. B. Major burns and crush injuries between 48 hours and 6 months old. C. Stroke or spinal cord injuries with profound residual deficits between 48 hours and 6 months
D. Hyperkalemia will worsen following the administration of Succinylcholine and may precipitate
ventricular dysrhythmias or even cardiac arrest. This is especially true in patients with pre-existing long term paralysis, crush injury, severe burns over 24 hrs old, kidney failure, neuromuscular disease or skeletal muscle myopathy. Its use is contraindicated if these exist.
A. Succinylcholine shall not be administered unless personnel trained and authorized in this
procedure are present and ready to perform the procedure.
B. Oxygen, ventilation equipment and resuscitation drugs shall be readily available. C. Succinylcholine produces paralysis, but does not alter a person's level of consciousness.
Paralysis in the conscious patient is very frightening, therefore, sedation should be provided to
the patient during the procedure. Verbal explanations should be provided to the patient during the procedure, even if you do not think they can hear you.
SIDE EFFECTS AND NOTES:
In rare individuals, because of pseudocholinesterase deficiency, paralysis may persist for a prolonged
period of time. Be prepared to continue to assist ventilations as needed.
ADULT DOSING
INDICATION DOSE
Rapid sequence intubation
Dose for 6 yrs old and older
PEDIATRIC DOSING
INDICATION DOSE
Rapid sequence intubation
Dose for less than 6 yrs old
VASOPRESSIN
OLMC REQUIRED: None
SUPPLIED: 20 units/1 mL vial
PHARMACOLOGY AND ACTIONS:
Vasopressin is a non-peptide hormone made in the posterior pituitary. Its primary role is water
regulation with a secondary role as vasoconstriction. It increases gastrointestinal and uterine motility and platelet aggregation. Vasopressin also results in the secretion of ACTH, aldsoterone, and factor VIII.
INDICATIONS:
As the first line pressor agent in cardiac arrest.
CONTRAINDICATIONS:
Hypersensitivity to the medication
PRECAUTIONS:
None in cardiac arrest
SIDE EFFECTS AND NOTES:
None in cardiac arrest.
ADULT DOSING
INDICATION DOSE
V-Fib/Pulseless VT, Asystole, PEA
PEDIATRIC DOSING (Not used in pediatrics)
VECURONIUM (Norcuron®)
OLMC REQUIRED: None
SUPPLIED: 10 mg vial of powder and 10 mL vial of dilutent solution
PHARMACOLOGY AND ACTIONS:
Vecuronium is a non-depolarizing neuromuscular blocking agent causing skeletal muscle relaxation. It
reversibly binds the acetylcholine receptor, blocking the action of acetylcholine. Neuromuscular blockade occurs within 2-3 minutes. Time to recovery is 30-45 minutes. Vecuronium metabolism is 5-35% renal with the remainder done in the liver.
A. For sustained neuromuscular blockade in the intubated patient. B. As the first line agent for Rapid Sequence Induction in the patient where Succinylcholine is
contraindicated.
CONTRAINDICATIONS:
None
A. Due to the prolonged duration of action, the endotracheal or pharyngeal tube must be
continually monitored to ensure correct placement with ETCO2 and pulse oximetry.
B. Patients with renal or hepatic failure may experience prolonged paralysis.
C. Vecuronium has no affect on consciousness and must be used with a sedative or induction
SIDE EFFECTS AND NOTES:
A. Vecuronium exhibits minimal side effects and does not substantially affect heart rate or rhythm,
systolic or diastolic blood pressure, mean arterial pressure, cardiac output, or systemic vascular resistance.
B. Vecuronium can be used to maintain paralysis even if intubation was performed without
Succinylcholine.
ADULT DOSING
INDICATION DOSE
RSI and maintenance of post-intubation
PEDIATRIC DOSING (Same as adult)
OPERATIONS
Marion & Polk County Regional Treatment Protocols
GENERAL PATIENT CARE GUIDELINES
TREATMENT:
A. Assess scene safety and use appropriate PPE
a. Gloves b. Masks c. Masks with Face shields d. Safety Glasses e. HEPA Masks f. Gowns
B. Begin initial patient assessment and determine chief complaint. C. Secure airway and start Oxygen as needed per General Airway Management protocol. D. Monitor vital signs and SpO2. E. Monitor ECG, ETCO2 and obtain CBG readings as appropriate. F. Establish IV/IO access as required. G. Obtain pain severity scale if applicable. H. Follow appropriate Patient Care Treatment Protocol if patient's chief complaint or
assessment findings change.
NOTES & PRECAUTIONS:
If patient is unable to provide medical history, check for medical bracelets and necklaces which can
provide critical medical information and treatment.
KEY CONSIDERATIONS:
*Medical history
*Medical Bracelets
*Medications and Allergies
PANDEMIC:
In the event of a pandemic event utilize proper PPE and contact the following Agencies for direction
as needed:
-Marion County Public Health (MCPH): 503-588-5357
-Center for Disease Control (CDC): 1-770-488-7100, 24/7 Emergency Line
Marion & Polk County Regional Treatment Protocols
Operations – Page 1 of 27
ADVANCED DIRECTIVES/POLST/ DNR ORDERS
PURPOSE:
This EMS system believes in respect for patient autonomy. The patient with decision-making
capacity has the right to accept or refuse medical intervention. This includes the right to specify, in
advance, patient preferences when the person is no longer able to communicate wishes.
PROCEDURE:
The EMS system shall honor POLST forms, Advance Directives and other Do Not Resuscitate
orders (DNR) under the following circumstances:
A. Do Not Attempt Resuscitation: In the pulseless and apneic patient who does not meet
the criteria of the DEATH IN THE FIELD protocol, but is suspected to be a candidate for withholding resuscitation, BLS protocols will be followed until one of the following occurs:
1. The EMT sees a written DNAR order, which should be honored and resuscitation
2. The patient's physician is contacted and directs EMTs to discontinue resuscitation. 3. The EMT sees a valid Advance Directive or Directive to Physician which directs them
not to continue resuscitation.
4. The patient's attorney-in-fact (PAHC or DPAHC) directs the EMTs not to resuscitate the
5. OLMC directs the EMTs not to continue resuscitation. 6. If a person who is terminally ill appears to have ingested medication under the provisions
of the Oregon Death with Dignity Act. (See Section F below)
B. Advance Directives: DNAR (DNR) orders only apply if the patient is in cardiopulmonary
arrest. If the patient's PAHC, DPAHC, or other Advance Directive is available to convey the patient's wishes, and the EMTs have seen a copy of the document, the EMTs must honor the treatment preferences as expressed.
C. Physician Orders for Life-Sustaining Treatment (POLST): If a POLST form is
available and it clearly expresses the patient's wishes and is signed by a physician, nurse practitioner or physician's assistant, EMTs shall honor the patient's treatment care preferences as documented in the EMS section of the POLST. [Cite: OAR 847-035-030 (7)] If an electronic registry is available and the POLST form is not immediately available, EMTs may also follow orders documented in the electronic POSLT registry.
D. Oregon POLST Registry Emergency Number: 1-888-476-5787
E. If there are questions regarding the validity, or enforceability, of the health care instruction,
begin BLS treatment and contact OLMC.
F. It is always appropriate to provide comfort measures as indicated.
Marion & Polk County Regional Treatment Protocols
Operations – Page 2 of 27
ADVANCED DIRECTIVES/POLST/ DNR ORDERS
F. Death with Dignity Act: If a person who is terminally ill and appears to have ingested
medication under the provisions of the Oregon Death with Dignity Act, the EMT should:
1. Provide comfort care as indicated.
2. Determine who called 9-1-1 and why (i.e. to control symptoms or because the person
no longer wishes to end their life with medications).
3. Establish the presence of DNAR orders and/or documentation that this was an action
under the provisions of the Death with Dignity Act.
a. DNAR orders are present, and
b. There is evidence that this is within the provisions of the Death with Dignity Act,
DEFINITIONS:
A. Do Not Attempt Resuscitation Order (DNAR): An order written by a physician
stating that in the event of cardiopulmonary arrest, cardiopulmonary resuscitation will not be administered. DNAR orders apply only if the patient is pulseless and apneic.
B. Health Care Instruction: A document executed by a person to indicate the person's
instructions regarding health care decisions.
C. Advance Directive: A document that contains a health care instruction or a power of
attorney for health care.
D. Living Will: A document that may confirm an Advance Directive or Directive to Physician
informing her/him that if the patient has a terminal illness and death is imminent, the patient would not wish to be placed on artificial life support that will only prolong the process of dying. In general, the traditional Living Will document alone is not helpful in the out-of-hospital setting because of its multiple restrictions and lack of clarity on when it should take effect.
E. Attorney in Fact: An adult appointed to make health care decisions for a person.
F. Power of Attorney for Health Care (PAHC): Power of attorney document that
authorizes an attorney-in-fact to make health care decisions for a person when the person is
G. Physician Orders for Life-Sustaining Treatment (POLST): The POLST is a
voluntary form that was developed to document and communicate patient treatment
preferences across treatment settings. It includes a section for documentation of DNAR
orders and a section communicating patient preferences for EMS care. While these forms
are most often used to limit care, they may also indicate that the patient wants everything
medically appropriate done. Read the form carefully! When signed by a physician (MD
or DO), nurse practitioner, or physician's assistant, the POLST is a medical order and the
EMTs are directed to honor it in their Scope of Practice
Marion & Polk County Regional Treatment Protocols
Operations – Page 3 of 27
CRIME SCENE RESPONSE
PURPOSE:
Law enforcement agencies stress that their first priority on any crime scene is the preservation of
life with reconstruction of the crime scene second. EMS personnel can be of assistance by adhering
to the following guidelines regarding crime scene response.
PROCEDURE:
A. Response and Arrival
1. Be conscious of physical and weather conditions around the site. Tire tracks of suspect
vehicles are often located in or adjacent to a driveway.
2. Limit the number of personnel allowed onto the scene. Consult with police on the
scene to direct placement of vehicles and route of personnel onto the scene.
B. Access and Treatment
1. Select a single route to the victim. Maintaining a single route decreases the chance of
altering or destroying evidence or tracking blood over a suspect's footprints.
2. Note the location of furniture, weapons, and other articles, and avoid disturbing them. If
they need to be moved, someone should note the location the article was moved from, by whom it was moved, and where it was placed.
3. Remove from the scene all EMS generated debris that is contaminated with blood or
body fluid and dispose of through established channels.
4. Be conscious of any statements made by the victim or other persons at the crime scene.
Write down what these statements were and report to the investigating officers.
5. Note the specific garments worn by the patient at the time of treatment. It is also
important not to tear the clothing off or cut through any holes, whether made by a knife, bullet, or other object.
6. The victim should be placed on a clean sheet when ready for transport. At the hospital,
please try to obtain the sheet once the victim is moved off of it, fold it carefully in on itself, and give it to the investigating officers. This is especially important in close contact crimes such as rape, serious assault and death cases.
C. Documentation
1. A detailed report is important in case you are later called to testify in court. An incident
report should be completed and should cover your observations, conversations with
family or witnesses, location of response vehicles and equipment, furniture, weapons, clothing that has been moved, items that were handled and your route to the victim.
2. An Unusual Event Report may be helpful for you to complete. This is a protected
document and if you are called to court may be used by you to refresh your memory of aspects of the call that are not included in the Patient Care Report.
3. Do not offer your opinions or evaluations about the crime scene.
REMINDER:
Any location can be, or become, a crime scene. When responding, and upon arrival, if something
does not appear to be right, notify police. If you suspect a crime scene and police are not present,
secure area and document what you see.
Marion & Polk County Regional Treatment Protocols
Operations – Page 4 of 27
DEATH IN THE FIELD
PURPOSE:
The purpose of the Death in the Field Protocol is to define under what conditions medical care can
be withheld or stopped once it has been started.
PROCEDURE:
Resuscitation efforts may be withheld if:
A. The patient qualifies as a "DNR". (See Advance Directives Protocol) B. The patient is pulseless and apneic in a mass casualty incident or multiple patient scene where the
resources of the system are required for the stabilization of living patients.
C. The patient is decapitated. D. The patient has rigor mortis in a warm environment. E. The patient is in the stages of decomposition. F. The patient has skin discoloration in dependent body parts (dependent lividity).
TRAUMATIC ARREST:
1. A victim of trauma (blunt or penetrating) who has no vital signs in the field may be declared dead on
scene. If opening the airway does nort restore vital signs/signs of life, the patient should NOT be
transported unless there are extenuating circumstances.
2. A cardiac monitor may be beneficial in determining death in the field when you suspect a medical
cause or hypovolemia: A narrow complex rhythm (QRS less than .12) may suggest profound hypovolemia, and may respond to fluid resuscitation.
3. At a trauma scene, the paramedic should consider the circumstances surrounding the incident,
including the possibility that a medical event (cardiac arrhythmia, seizure, and hypoglycemia) preceded the accident. When a medical event is suspected, treat as a medical cardiac event. VF should raise your index of suspicion for a medical event.
4. In instances prior to transport where the patient deteriorates to the point that no vital signs (i.e.
pulse/respiration) are present, a cardiac monitor should be applied to determine if the patient has a viable cardiac rhythm. A viable rhythm especially in patients with penetrating trauma may reflect hypovolemia or obstructive shock (tamponade, tension pneumothorax) and aggressive care should be continued.
MEDICAL CARDIAC ARREST:
1. Patients with persistent/refractory Ventricular Fibrillation should be transported, except when Advanced
Directives, DNR orders, or other withholding resuscitative efforts apply. If in doubt, contact OLMC.
2. Patients with Pulseless Electrical Activity (PEA) who have not responded to ACLS care for PEA may be
determined to be dead in the field (if ETCO2 less than 20) after consultation with OLMC.
3. When a patient's cardiac rhythm deteriorates to asystole or an agonal rhythm despite appropriate
cardiac resuscitative measures (including establishing an effective airway, administering epinephrine and/or vasopressin, adequate CPR and checking the rhythm in at least three leads) the PIC may determine that the patient is dead in the field.
4. If the patient's ECG shows asystole or agonal rhythm upon initial monitoring, and after checking the
rhythm in at least three leads, the patient, in the paramedic's best judgment, would not benefit from resuscitation: a. The PIC should determine DIF and notify the Medical Examiner or Law Enforcement;
b. Begin BLS procedures, and contact OLMC with available patient history, current condition, and
with a request to discontinue resuscitation.
Marion & Polk County Regional Treatment Protocols
Operations – Page 5 of 27
DEATH IN THE FIELD
NOTES & PRECAUTIONS:
1. ORS allows a layperson, EMT or Paramedic to determine "Death in the Field"
2. The EMT is encouraged to consult OLMC if any doubt exists about the resuscitation potential
of the patient.
3. A person who was pulseless or apneic and has received CPR and has been resuscitated, is not
precluded from later being a candidate for solid organ donation.
4. ETCO2 may be a useful adjunct in the decision to terminate resuscitation with PEA. An ETCO2
of 10 or less in patients in PEA after 20 minutes of ACLS resuscitation does not correlate with
5. Survival from trauma arrest is low, but not completely zero.
6. If person has been identified as an organ donor, contact the Medical Examiner as soon as is
reasonably possible.
7. ORS 146.090 Deaths requiring investigation.
a. The Medical Examiner shall investigate and certify the cause and manner of all human
i. Apparently homicidal, suicidal or occurring under suspicious or unknown
ii. Resulting from the unlawful use of controlled substances or the use or abuse of
chemicals or toxic agents;
iii. Occurring while incarcerated in any jail, correction facility or in police custody;
iv. Apparently accidental or following an injury;
v. By disease, injury or toxic agent during or arising from employment;
vi. While not under the care of a physician during the period immediately previous
vii. Related to disease which might constitute a threat to the public health; or
viii. In which a human body apparently has been disposed of in an offensive manner.
8. ORS 146.103 Removal of body, effects or weapons prohibited without consent
a. In a death requiring an investigation, no person shall move a human body or body
suspected of being human, or remove any of the effects of the deceased or
instruments or weapons related to the death without permission of a medical
examiner, deputy medical examiner or the district attorney.
b. No person shall move or remove any of the items specified in subsection (1) of this
section if the medical examiner or district attorney objects.
c. A medical examiner, district attorney or deputy medical examiner shall take custody
of or exercise control over the body, the effects of the deceased and any weapons,
instruments, vehicles, buildings or premises which the medical examiner, district
attorney or deputy medical examiner has reason to believe were involved in the
death, in order to preserve evidence relating to the cause and manner of death.
d. In a death requiring investigation, no person shall undress, embalm, cleanse the
surface of the body or otherwise alter the appearance or the state of the body
without the permission of the medical examiner or the district attorney.
Marion & Polk County Regional Treatment Protocols
Operations – Page 6 of 27
PROCEDURE:
A. A patient care report shall be generated for each identified patient and shall be completed on
an approved Agency EMS patient care form.
B. Documentation shall include, at least:
1. The patient's presenting problem. 2. Vital signs with times. 3. History and physical findings as directed in by individual protocols. 4. Treatment(s) provided, and time(s). 5. If monitored, ECG strip and interpretation. 6. Any change in the condition of patient. 7. OLMC contact:
a. Include physician name b. Time of contact c. Orders received from physician
C. A copy of the Prehospital Care Report must be left at the receiving hospital whenever a
patient is transported per ORS 333-250-0044.
D. If a patient refuses treatment and/or transport, refer to Refusal and Informed consent
Marion & Polk County Regional Treatment Protocols
Operations – Page 7 of 27
GRIEVING PEOPLE
PURPOSE:
These guidelines are intended to provide paramedics with direction during the difficult emotional
times immediately before and following death. They suggest words to say, steps to take, and
directions for dying patients and grieving family and friends. They are not intended to rigidly codify
or regulate how one should respond during this intensely personal experience, rather, the following
recommendations and resources are offered as a guide.
RESUSCITATION PHASE:
A. As time allows, give accurate and truthful updates about the patient's prognosis. If available,
assign one person to interact with and support family members.
B. Consider gently removing children from the resuscitation area. C. Depending upon the emotional state of family members, consider allowing them to watch
and/or participate in a limited and appropriate way.
D. If family or friends were doing CPR prior to your arrival, commend their efforts. E. If family or friends are disruptive consider removing them or try assigning simple tasks, such
as helping bring in the stretcher, holding doors open, telling other family about the event
and calling the doctor or minister.
F. Be respectful. Make requests. Don't give orders.
ONCE DEATH IS DETERMINED:
A. Treat the recently dead with respect. B. Tell family and friends of the death honestly. Use the words "death" or "dead". Avoid using
euphemisms such as "passed away" or "gone".
C. Avoid using past tense terms when speaking to survivors of the recently dead.
D. Allow family and friends to express their emotions. Listen to them if they want to talk but
don't push them.
E. Give factual information.
F. Genuine warmth and compassion will be more helpful than almost anything else for survivors.
Don't feel it necessary to say the "right" things. Listening often provides grieving people with the most comfort.
FOCUSING ON THE SURVIVORS:
A. See to it that survivors have a support system present before you leave. Consider calling
TIP through EMS Dispatch, if available in your jurisdiction. Call friends, family, clergy, or
neighbors to be with them. Respect the survivor's wishes to be alone.
B. Explain the next steps to them after you have pronounced death. This will include the
police coming to make reports, possibly the medical examiner, and the possible need for an
autopsy in certain instance.
C. Contact the Medical Examiner's office before moving or altering the body (as soon as
D. Allow family and friends to say their good-byes if possible.
E. A chaplain may be helpful in assisting with survivors. It is advisable to call early, as the
chaplains do not have code-3 capabilities.
F. Help survivors make decisions such as which people should be called. If they ask you to
make calls, try to comply, mention the need to find a funeral home, if one has not already
been chosen. Clergy may also be helpful with this decision.
Marion & Polk County Regional Treatment Protocols
Operations – Page 8 of 27
GRIEVING PEOPLE
DEATH OF A CHILD:
A. Do not accuse the parents of abuse or neglect, but take careful note of the patient
surroundings and the general physical condition of the child.
B. Do not be overly silent, which may imply guilt to the parents. C. Ask the parents only necessary questions and do not judge or evaluate them. Do not tell
them what they "should have" been doing before your arrival.
D. Remind parents to arrange for child care of other children. E. Listen carefully to their statements and answer only with accurate information. F. If there is a police investigation, tell the parents that this is only routine. G. Successful management of child deaths requires supportive, compassionate and tactful
DYING PATIENTS:
A. Look for Advance Directives and/or POLST forms and attempt to honor patient preferences
for or against treatment. Always provide comfort measures.
B. Be aware of your own fears regarding death. C. Be ready to admit when a dying person reminds you of a loved one. If a particular person is
too disturbing, have your partner or other members of the responding team take over.
D. Treat dying persons with warmth and understanding. Do not avoid them. Allow them to
discuss their situation, but do not push them to talk.
E. Many dying people are not upset by discussions of death as long as you do not take away all of their hope. F. Touching a dying person is important. Use words like "death". Do not use meaningless
G. Ask the person how you might help. Patients can frequently guide you to make them feel more comfortable. H. Give factual information.
Marion & Polk County Regional Treatment Protocols
Operations – Page 9 of 27
HAZARDOUS MATERIALS
PURPOSE:
Paramedics may be first on the scene of a hazardous materials situation because of shorter
response times or no knowledge of dispatch that hazardous materials are involved. This protocol is
intended to guide paramedics who do not normally function in hazardous materials scenes. If the
scene you are responding to is a known or suspected (based on information from dispatch)
hazardous materials situation, stage and wait for the hazardous materials personnel. When you
have arrived at the scene and find out during scene assessment that hazardous materials are
involved, stage and wait for the hazardous materials personnel. All scenes (MVA, Industrial, etc.)
should be considered as being a potential hazardous materials situation. The following approach
procedure should be used:
PROCEDURE:
a. Be cautious all times.
b. The reported location may be inaccurate, response into a contaminated area might
c. Approach upwind and upgrade if possible.
d. Position vehicle well away from the incident.
e. Communicate your actions to the 9-1-1 Center.
f. Remember: contaminated and/or exposed response personnel may add to the
overall problem and reduce their effectiveness to help.
2. If at any time you suspect a hazardous materials situation:
a. Confirm that fire and police have been notified. The agency responsible for
hazardous materials response may respond with different levels of personnel and
equipment based upon the information received. Do not always expect a hazardous
materials team to respond.
b. If you are a first-in responder, the first priority is scene isolation.
c. If you believe that you or your vehicle is contaminated, stage in an isolated area.
KEEP OTHERS AWAY! KEEP UNNECESSARY EQUIPMENT FROM BECOMING
Person in Charge
1. If the paramedic is the first medical person on the scene, he/she should assume the role of
PIC (medically) until a "hazardous materials trained paramedic" (HMP) arrives. If at all
possible the Incident Command Structure should be implemented.
2. The HMP will direct all care.
3. The HMP will determine the method of transport of the exposed patient (air vs. ground).
4. The HMP will determine who will provide care during transport (HMP may remain in that
position during transport).
Marion & Polk County Regional Treatment Protocols
Operations – Page 10 of 27
HAZARDOUS MATERIALS
Patient Care for the Contaminated Patient
i. Types of incidents which may require decontamination of the patient:
b. Biological hazards
d. Toxic substances
ii. Contamination can occur though:
c. Direct contact
iii. Determine the hazardous substance involved and provide treatment as directed by the Haz-
Mat Paramedic (HMP). In the absence of an HMP, consult Poison Control through OLMC.
iv. The hazardous materials team must be contacted about removal of contaminated clothing
and packaging of the patient with regard to your protection and the patient's.
D. Ambulance Preparation
1. The HMP shall determine the process needed for ambulance preparation.
2. Remove any supplies and equipment that will be needed for patient care.
3. Seal cabinets and drape interior, including floor and squad bench, with plastic (available from
hazardous materials team).
E. Transport and Arrival at the Hospital (if requested by "HMP")
1. If an ambulance has transported a patient from an incident that is subsequently determined
to involve hazardous materials exposure, scene personnel must immediately relay all
relevant information to the transporting unit(s) and/or receiving facility(s) involved (via EMS
dispatch or OLMC).
2. OLMC and the receiving hospital should be contacted as soon as possible. The paramedic
should communicate the material involved, degree of exposure, decontamination
procedures used and patient condition.
3. The ambulance should park in an area away from the emergency room or go directly to a
decontamination center or area.
4. Patient(s) should not be brought into the emergency department before paramedics receive
permission from the hospital staff.
5. Once the patient(s) has been released to the hospital, follow the HMP's direction and if
necessary double bag the plastic sheeting used to cover the gurney and the floor. Double
bag any equipment, which is believed to have become contaminated.
6. After unloading the patient from the ambulance, check with the "HMP" to see where the
ambulance can be safely decontaminated and whether or not there is equipment available
for this purpose. Do not begin decontamination without direction from the "HMP". After
consultation with the Hazardous Materials Team leader the HMP may recommend that the
ambulance be decontaminated.
7. Following decontamination recommendations from the "HMP", decontaminate the
ambulance and personnel before returning to the incident scene. When returning to the
incident scene, bring bags containing contaminated materials, equipment, clothing, etc., and
turn them over to the "HMP".
Marion & Polk County Regional Treatmen
Operations – Page 11 of 27
HAZARDOUS MATERIALS
Paramedic/EMT Exposure
1. If a paramedic/EMT is exposed or is concerned with the possibility of exposure, medical help
should be sought immediately.
2. Report all exposures to the HMP, Poison Center, and supervisor, and the on-call OHDP
3. Follow your agencies guidelines for Communicable Disease: Bloodborne/Airborne
Pathogens), including appropriate Personnel Exposure Report.
4. Do not return to service until cleared to do so by the HMP or Poison Center.
FOR ADDITIONAL INFORMATION SEE THE HAZMAT PROTOCOL
Marion & Polk County Regional Treatment Protocols
Operations – Page 12 of 27
HOSPICE PATIENT RESPONSE
GUIDELINE:
1. Initiate patient assessment and provide appropriate basic life support comfort care
2. Validate whether the patient is on Hospice- If YES, contact the Hospice RN via phone;
contact Hospice MD if conflict arises.
3. Obtain POLST documentation and familiarize yourself with the prescribed orders. 4. Once in contact with Hospice RN, report the patient's condition and your general
impression of the situation.
5. If available, consider involving the patient's family in the treatment discussion plan, remaining
sensitive to the emotional toll of the terminal diagnosis.
6. In coordination with the Hospice RN, determine if management of dyspnea, pain, nausea and
injuries can be treated with Hospice resources and avoiding transport.
7. Provide transport to hospital under the following conditions:
a. Hospice requests and authorizes transport (confirm this) b. Patient/Family (with the capacity to do so) demands transport even after being informed
that the transport and hospital treatment may not be covered by hospice/insurance and a financial obligation will result
8. DO NOT automatically transport in the following cases:
a. Care facility staff (not actual Hospice RN) says they won't/can't care for patient. b. Care facility staff or family states they "don't know what to do" with patient. c. Hospice RN advises that they can't or won't come into see the patient- If this
happens, contact the Hospice MD
9. When in doubt, or if you require additional consultation, contact OLMC.
DEFINITIONS:
Hospice- interdisciplinary program of supportive care to people and families in the final phase of a
terminal illness with a focus on comfort and quality of life, rather than cure or life prolongation;
emphasis on keeping the patient "home".
Terminal Illness-single diagnosis defined by attending MD and hospice medical director as the
primary reason for terminal prognosis; may include metastatic cancer, as well as end-stage cardiac,
pulmonary, liver, or kidney disease. In addition, terminal diagnosis may be:
Dementia: end-stage dementia (bedbound, non-verbal) and moderate dementia with
other medical problems that will no longer be treated aggressively (if other problems
Failure to Thrive: progressive functional, nutritional compromise, and/or Body Mass
Index less than 22.
Debility: defined by Medicare as a combination of multiple organ system problems; all
medical problems and any medical treatment is considered part of Debility diagnosis.
Any other diagnosis with documentation supporting a 6 month or fewer prognosis.
Marion & Polk County Regional Treatment Protocols
Operations – Page 13 of 27
MEDICAL CONTROL OF SCENE
PURPOSE:
The purpose of this protocol is to describe who is in charge of patient care on the scene of medical
emergencies and how to resolve disputes with other medical professionals in attendance. This
protocol does not apply to MCI/MPS events where ICS is established.
PROCEDURE:
A. EMTs/Paramedics/Prehospital Providers On-Scene: The first arriving, highest certified EMT will
be the Person-In-Charge (PIC) and will assume responsibility for directing overall patient care.
The team approach to patient care assessment and treatment should be utilized by the PIC.
B. When a higher level EMT arrives, in an EMS role, that individual shall assume the role of PIC,
after receiving verbal report from the initial PIC.
C. The responsibilities of the PIC directing overall patient care include:
1. Assuring that treatment, operations, and communications follow protocols.
2. Coordinating patient care activities. This PIC must watch over the entire patient care
scene activities and be sure that the patient care activities are being accomplished in a
rapid, efficient, and appropriate manner.
3. Directing other EMTs to establish airway management, start IVs, etc.
4. Establishing the appropriate time to be spent at the scene for doing patient care.
5. Determining when transportation of the patient is to occur.
6. Performing medical coordination with all agencies and personnel.
D. The PIC directing overall patient care will be held responsible and accountable for patient care
activities performed at the scene and be identified on all patient care reports.
E. If a patient requires transport and the first arriving PIC is from a non-transporting agency,
provision of patient care will be turned over to the transporting EMT-P or flight personnel
1. The patient is placed on the transport unit's gurney, OR
2. At a time agreed upon by both EMTs, continued patient care will then become the
responsibility of the transporting unit. There will be a verbal agreement anytime transfer
of care from one EMT (EMT-P) to another takes place.
Paramedic Direction On Scene:
EMTs and Paramedics take medical direction from:
1. Physician Supervisors.
2. Regional Protocols.
3. On-Line Medical Control (OLMC) as directed in protocols.
Physician On Scene Policy, (within office):
When EMS is called to a physician's office, the EMTs and paramedics should receive information
from the physician and attempt to provide the service requested by the physician.
While in the physician's office, the physician shall remain in charge of the patient. The EMTs and
paramedics may follow the direction of the physician as long as it is within the Scope of Practice
and protocols of the PIC. Anytime there is a conflict between a physician's orders and the
protocols, OLMC shall be contacted.
Once the patient is in the ambulance, unless the physician accompanies the patient, paramedics shall follow the protocols.
Marion & Polk County Regional Treatment Protocols
Operations – Page 14 of 27
MEDICAL CONTROL OF SCENE
Physician On-Scene Policy, (outside office):
A. Any physician (MD or DO) at the scene of an emergency may be qualified to provide assistance
to EMTs and paramedics and shall be treated with professional courtesy.
B. A licensed physician requesting control of patient care at the scene shall be:
1. Thanked for the offer by the PIC.
2. Advised that the EMTs and Paramedics work under regional protocols and On-Line
Medical Control.
3. Advised that we are not permitted to relinquish medical control to a physician on the
scene without agreement from On-Line Medical Control.
C. If the physician requesting control is not the patient's "physician of record," EMTs and
paramedics shall be authorized to proceed under the direction of the physician ONLY IF ALL THREE OF THE FOLLOWING PROVISIONS ARE MET:
1. OLMC is contacted and authorizes transfer of patient care.
2. The physician agrees to accompany the patient to the hospital in the ambulance.
3. The physician agrees to complete and sign the appropriate patient care report.
D. If communication with OLMC cannot be established, care may be provided only according to
approved ALS protocols. No direction from an on scene physician may be accepted.
Disputes On-Scene Between EMTs or Other Medical Professionals:
A. Disagreements about care should be handled in a professional manner and shall not detract
from patient care.
B. To the extent possible, the ALS and BLS protocols shall be followed and provide the basis for
resolving disputes.
C. If an unresolved dispute continues between EMTs or other medical professionals concerning the
care of a patient, OLMC shall be contacted.
D. If a dispute arises which results in transfer of patient care from one PIC to another, the
approximate time of the transfer shall be included on the patient care report.
E. DISPUTES SHALL NOT APPEAR ON PATIENT CARE REPORTS. Written "Unusual Event
Forms" should be completed pursuant to any dispute arising at the scene.
Marion & Polk County Regional Treatment Protocols
Operations – Page 15 of 27
ON-LINE MEDICAL CONTROL
PURPOSE:
This protocol describes the steps an EMT should follow in contacting On-Line Medical Control
(OLMC), and describes the contents of the various reports.
PROCEDURE:
A. Calls to OLMC: EMTs shall contact OLMC or the Receiving Hospital by radio or
telephone in the following situations:
As required by the protocols
As required for trauma services
When On-Line Medical Control is needed
B. All scenes involving OLMC contact:
One person at the scene must be designated as the contact person in charge of
communications. The EMT designated as "in charge" of communications shall
contact OLMC or the Receiving Hospital by the time transport has begun, including
all air ambulance transports
If BLS responders have initiated OLMC communications, ALS responders shall
continue to use that medical direction source
C. When requesting OLMC, the following information must be relayed
Unit number, identity and certification level of person making contact
Location of the call, street address if appropriate
Purpose of call (Identify the protocol being followed)
Age and sex of patient
Patient's chief complaint
Brief history, prior medical history, medications, and allergies
Pertinent physical findings
Treatment at scene
ETA, including loading time
Marion & Polk County Regional Treatment Protocols
Operations – Page 16 of 27
ON-LINE MEDICAL CONTROL
D. When contacting the Receiving Facility for trauma system patients, the following
information must be relayed: 1.
Unit number, identity and certification level of person making contact
Number of patients. Follow Multiple Patient Scene or Mass Casualty Incident protocol, if applicable.
Age and sex of the patients
Trauma System entry criteria (be as specific as possible)
Trauma Band number(s)
Patient's vital signs, specify if not taken or not present
Approximate ETA of patient(s) to Trauma Center; include loading time if appropriate
Unit number and mode of transport
Patient destination based on incident location or request
Marion & Polk County Regional Treatment Protocols
Operations – Page 17 of 27
REFUSAL AND INFORMED CONSENT
PURPOSE:
To establish the process of obtaining informed consent.
To define which persons may be left at the scene because they are not considered in need of
To describe the process of obtaining and documenting patient refusal.
PROCEDURE: (Refer to Refusal Flow sheet)
A. Determine if there is an "Identified Patient":
Pt contact is defined as an EMT in conjunction with a response in an Ambulance doing any of
the following seeing or hearing a patient, having direct contact with patient or patients
caregiver (verbal or physical), or being directly or indirectly involved in the patients care plan
Determine "No Patient Identified" if the person meets ALL of the following criteria:
No significant mechanism of injury.
No signs of traumatic injury.
No acute medical condition.
No behavior problems that place the patient or others at risk.
Person is NOT less than 18 years of age.
Person is NOT the 911 caller.
Identified Patient who is refusing medical care or transport:
Determine if the patient appears to have impaired decision making capacity. Consider
conditions that may be complicating the patient's ability to make a decision:
Head injury.
Drug or alcohol intoxication.
Toxic exposure.
Psychiatric problems.
Language barriers (consider translator or ATT language line through dispatch).
Serious medical conditions.
C. Identified Patient WITH decision making capacity who refuses needed treatment and/or
1. Explain the risks and possible consequences of refusing care and/or transport.
2. If a serious medical need exists, contact OLMC for physician assistance.
3. Enlist family, friends, or law enforcement to help convince patient.
4. If patient continues to refuse, complete the Patient Refusal Information Form and have
them sign it. Follow Documentation Protocol.
D. Identified Patient WITH IMPAIRED decision making capacity:
1. Treat and transport any person who is incapacitated and has a medical need.
2. Patients with impaired decision making capacity should NOT sign a release form.
3. With any medical need, make all reasonable efforts to assure that the patient receives
medical care. Attempt to contact family, friends, or law enforcement to help.
4. If deemed necessary, consult with OLMC and consider chemical or physical restraint per
Restraining of Patients Protocol.
Marion & Polk County Regional Treatment Protocols
Operations – Page 18 of 27
REFUSAL AND INFORMED CONSENT
All instances of an identified patient, with or without impaired decision making capacity, must be
fully documented on a Patient Care Form with an attached signed refusal form. The following is
considered minimum documentation criteria:
General appearance and level of consciousness (mental status).
History, vital signs, and physical exam.
Presence of any intoxicants.
Assessment of the person's decision making capacity.
Risks explained to patient.
Communication with family, friends, police, and/or OLMC.
GUIDELINES & DEFINITIONS:
A.
Decision Making Capacity: The ability to make an informed decision about the need for medical
Accurate information given the patient regarding potential risks associated with refusing
treatment and/or transport.
The persons perceived ability to understand and verbalize these risks.
The person's ability to make a decision that is consistent with his/her beliefs and life goals.
B. Impaired Decision Making Capacity: The inability to understand the nature of the illness or
injuries, or the risks and consequences of refusing care.
C. Emergency Rule: EMTs may treat and/or transport under the doctrine of implied consent a
person who requires immediate care to save a life or prevent further injury. Minors may be
treated and transported without parental consent if a good faith effort has been made to
contact the parents or guardians regarding care and transport to a hospital, and the patient, in
the opinion of EMTs, needs transport to a hospital. When in doubt, contact OLMC.
D. Required OLMC Contact: EMTs are strongly advised to contact OLMC for the following refusal
Suspected impaired decision making capacity.
Suspected serious medical condition such as:
o Respiratory distress.
o Sustained abnormal vital signs.
o Compromised airway.
o Uncontrolled bleeding.
o Suspected cervical spine injury.
o Infants under 3 months of age.
o Cardiac dysrhythmia.
o Poisons and overdoses.
o First time seizures.
o Suspected intoxication or impairment
Suspected abuse situation involving a minor or the elderly.
Any unconscious or altered mental status (individual or parent/guardian for a minor).
Conflict on scene regarding refusal of care.
Minor without a parent or guardian who is refusing care.
Marion & Polk County Regional Treatment Protocols
Operations – Page 19 of 27
REFUSAL AND INFORMED CONSENT
ASSESS PATIENT'S MEDICAL NEED
NO IDENTIFIED PATIENT
IDENTIFIED PATIENT
No significant mechanism.
No visible signs of traumatic injury
No known acute medical condition
ASSESS ABILITY TO MAKE DECISIONS
No identifiable behavior problems.
NOT less than 18 years old.
Consider:
DID NOT request medical assistance.
Head injury.
Drug or alcohol intoxication.
Medical conditions (e.g., hypoglycemia).
Toxic exposure.
No Patient Care Report required.
Psychiatric problems.
Follow Documentation Protocol.
Language barriers.
ABLE TO MAKE DECISIONS
UNABLE TO MAKE DECISIONS
Ambulance transport advised, but
(Impaired Capacity)
-ACTION-
-ACTION-
Refusal Form Required
DO NOT have patient sign
Refusal Form
Explain risks of refusal.
If serious medical need exists, contact
Treat & transport if medical emergency
exists. Use Restraining of Patients
Enlist family, friends, police, etc. to
protocol if needed.
counsel patient.
Make all reasonable efforts to assure
Complete Refusal Form and obtain
patient gets medical care.
patient signature.
Consult OLMC.
Follow Documentation protocol.
MINIMUM DOCUMENTATION
OLMC CONTACT STRONGLY
For ALL Identified Patients
Impaired decision-making capacity.
General appearance & level of
Suspected serious medical condition.
History, vital signs, & physical exam.
Suspected abuse – child or elderly.
Presence of any intoxicants.
First-time seizures (all).
Assessment of patient's decision-
Scene conflict regarding medical care.
making capacity.
Minor without guardian refusing care.
Any risks that were explained to the
Suspected intoxication or impairment
Communications with family, police,
Marion & Polk County Regional Treatment Protocols
Operations – Page 20 of 27
PURPOSE:
To establish guidelines for the evaluation and treatment of personnel assigned to the Rehabilitation
PROCEDURE:
A. Upon assignment to REHAB, individuals will undergo an initial medical evaluation consisting of
blood pressure, pulse and temperature measurements. All medical evaluations will be recorded
on standard forms as outlined in the Emergency Incident Rehabilitation Training Bulletin.
B. Medical Treatment or a resting period will be determined according to the following Triage
Criteria based on Entry Findings:
1. If initial exam findings include:
Heart Rate over 110, Temperature greater than 100.6 F
Any type of injury
Shortness of breath
* Immediately assign person to Medical Unit
2. If initial exam findings include:
Heart Rate over 120 and Temperature greater than 100.6 F or ETCO 10% or greater Blood Pressure over 160 systolic or less than 100 systolic or over 110 diastolic
regardless of temperature.
Temperature greater than 100.6 F regardless of other vital signs.
* Continue to observe in Rest and Refreshment Area and reassess in 20
minutes.
3. If, after 20 minutes, re-assessment exam findings include:
Heart Rate over 100 Blood Pressure greater than 160 systolic or less than 100 systolic or over 110 diastolic Temperature greater than 100.6 F
ETCO 10% or greater
* Continue to observe in Rest and Refreshment Area and reassess in 10
minutes.
4. If, after an additional 10 minutes, re-assessment exam findings include:
Heart Rate over 100
Blood Pressure greater than 160 systolic or less than 100 systolic or over 90 diastolic
Temperature greater than 100.6 F
ETCO 10% or greater
* Immediately assign person to Medical Unit
5. If, after initial or second re-assessment, exam findings include:
Heart Rate over 60 and less than 100 Systolic Blood Pressure over 100 and less than 160 Diastolic Blood Pressure over 60 and less than 100 Temperature between 97.6 F and 99.6 F ETCO <10%
* Personnel may be returned to Staging for re-assignment.
Marion & Polk County Regional Treatment Protocols
Operations – Page 21 of 27
REHAB ENTRY FLOWCHART
PRIMARY ASSESSMENT:
Any Injuries?
DIRECT TO
Shortness of Breath?
MEDICAL UNIT
Chest Pain? Temperature > 100.6°F, with a Pulse > 110.
SECONDARY ASSESSMENT:
AGGRESSIVE TREATMENT
Temperature > 100.6°F, Regardless of other Vitals.
IN REST and REFRESHMENT
Pulse > 120 and a Temperature > 100.6°F.
Systolic BP < 100 or > 160.
Diastolic BP > 100.
ETCO 10% or greater
REASSESS VITALS AFTER
20 MINUTES:
Temperature < 100.6°F.
Pulse 60 – 100.
NORMAL REHAB IN REST and
Systolic BP 100 – 160.
REFRESHMENT AREA:
Diastolic BP 60 – 100.
ETCO 10% or greater
Minor Nutritional Support.
Rehydration.
Minimum of 20 Minutes in Rehab.
REASSESS VITALS AFTER
Reassess Vitals after 20 Minutes
10 MINUTES:
Temperature < 100.6°F.
Pulse 60 – 100.
Systolic BP 100 – 160.
Diastolic BP 60 – 100.
VITALS NEEDED TO
ETCO 10% or greater
RETURN TO STAGING
Pulse 60 – 100.
DIRECT TO
Systolic BP 100 – 160.
MEDICAL UNIT
Diastolic BP 60 – 100.
Temperature 97.6°F – 99.6°F. ETCO <10%
Marion & Polk County Regional Treatment Protocols
Operations – Page 22 of 27
REPORTING OF SUSPECTED CHILD ABUSE
PURPOSE: To establish guidelines for the reporting of suspected child abuse.
A. Abuse: The non-accidental assault or physical injury to a child. This may include mental abuse,
sexual abuse, neglect, etc.
B. Child: An unmarried person under the age of 18.
C. Public or Private Officials: physicians, including residents and interns, firefighter or EMT among
PROCEDURE:
It is the policy of the State of Oregon to require mandatory reporting of suspected child abuse.
A. D UTY TO REPORT CHILD ABUSE (ORS 419B.010)
Public or private officials All EMS PERSONNEL ARE REQUIRED have a duty to report child
abuse REGUARDLESS OF ON DUTY STATUS. Such an official whoANY EMT WHO has re asonable cause to believe that a child has either been abused or witnessed abuse of another child
or adult, or who comes into contact with someone who has abused a child, shall report the
contact to a law enforcement agency, i.e., any city or municipal police department, any county
sheriff's office, the Oregon State Police AS SOON AS POSSIBLE, or DHS (Department of Human
Services, formerly SCF) or the County Juvenile Department. DHS: 503-378-6704, or 1-800-232-
3020. Passing the report only to a Nurse or Physician does not meet the requirement
of the Statute or the Protocol.
B. C ONTENT OF REPORT (ORS 419B.015)
Paramedic must file an Unusual Event Report with the EMS Office within 12 hours as outlined in
the Documentation Protocol. If there is imminent danger to health or life, notify police, the Chief
Officer and use your agency notification procedure. The report must contain, if known, the
following information:
- The names and addresses of the child and parents/person responsible for the child's care.
- The child's age.
- The nature and extent of abuse (including any evidence of previous abuse).
- The explanation given for the abuse.
- Any information the official believes may be helpful in establishing the cause of abuse or the
perpetrator's identity.
IMMUNITY OF PERSONS MAKING REPORTS (ORS 419.025):
Persons who acting in good faith and upon reasonable grounds, report child abuse are immune from
civil and criminal liability.
Marion & Polk County Regional Treatment Protocols
Operations – Page 23 of 27
REPORTING OF SUSPECTED ELDER ABUSE
PURPOSE:
To es
tablish guidelines for the reporting of suspected elder abuse or abuse of a resident in a long term care
OCEDURE:
ere are two separate elder abuse reporting requirements; a general reporting requirement which applies
to patients outside long-term care facilities and a special reporting requirement, which applies to patients of
long-term care facilities.
FINITIONS:
Abuse: The non-accidental physical injury to an elderly person or patient of a long term cares facility.
Abuse also includes:
1. Outside long-term care facilities:
a. Neglect means the withholding of services necessary to maintain health and well being.
Treatment solely by spiritual means is not neglect; however, the person must be voluntarily
under the care of an accredited practitioner or in accordance with the practices of a
recognized church or religion. Abandonment, including desertion or willful forsaking of
an elderly person or withdrawal or neglect of duties and obligations owed an elderly person
c. Willful infliction of physical pain or injury.
2. Inside long-term care facilities:
a. Il egal or improper use of the patient's financial resources for personal profit or gain.
b. Sexual contacts by force, threat, duress or coercion by an employee, agent or other
c. Use of derogatory names, phrases, harassment, intimidation, punishment or involuntary
Elderly person - Any person 65 years of age or over.
Long-term care facility - Any licensed skilled nursing facility or intermediate care facility.
OCEDURE:
DUTY TO REPORT ELDER ABUSE AND PATIENT ABUSE IN A LONG TERM CARE FACILITY
An EMT who has reasonable cause to believe that an elderly person has been abused, or who comes into contact with someone who has abused an elderly person, shall file an Unusual Event Report with the EMS
Office within 12 hours as outlined in the Documentation Protocol. If there is imminent danger to health
or life, notify police, or DHS . DHS: 503-304-3400 or 1-800-232-3020. Passing the report only to
a Nurse or Physician does not meet the requirement of the Statute or the Protocol.
B. CONTENT OF REPORT
The elder abuse report must contain, if known, the following information:
- The names and addresses of both the elderly person and anyone responsible for his/her care.
- The nature and extent of abuse including any evidence of previous abuse.
- The explanation given for the abuse.
- Any information the official believes may be helpful in establishing the cause of abuse.
IMMUNITY OF PERSONS MAKING REPORTS
ersons participating in good faith in making a report of elder abuse and who have reasonable grounds for
making it are immune from civil and criminal liability including participation in any judicial proceeding re sulting from their report. Persons making such a report of abuse of a patient in a long term care facility in
addition have immunity from any criminal liability that might otherwise be incurred or imposed with respect to making such a report.
Marion & Polk County Regional Treatment Protocols
Operations – Page 24 of 27
TRAUMA SYSTEM
PATIENT ENTRY:
The paramedic is required to report the reason for patient entry to the Receiving Facility. Patients are to
be entered into the Trauma System if they meet the following criteria:
Physiological Criteria
Glasgow Coma Scale less than or equal to13; or
Systolic blood pressure less than 90; or
Respiratory rate less than 10 or greater than 29 (less than 20 in infants less than one year); or
need for ventilatory support
Anatomical Criteria
All penetrating injuries to head, neck, torso and extremities proximal to elbow and knee
Chest wall instability or deformity (e.g. flail chest); or
Two or more proximal long-bone fractures; or
Crushed, degloved, or mangled extremity; or
Amputation proximal to wrist and ankles; or
Suspected pelvic fracture; or
Open or depressed skull fracture; or
Motor or sensory deficit
Mechanism of Injury
Adults: greater than 20 feet (one story is equal to 10 ft.); or
Children: greater than 10 feet or 2-3 times the height of the child; or
High-Risk Auto Crash
• Intrusion, including roof greater than 12 inches into passenger compartment; or greater
than 18 inches anywhere on vehicle; or
• Ejection (partial or complete) from automobile; or
• Death in same passenger compartment; or
• Vehicle telemetry data consistent with high risk of injury; or
Auto vs. Pedestrian/Bicyclist Thrown, Run Over, or with significant (greater than
20mph) impact; or
Motorcycle or ATV Crash greater than 20 mph
Special Populations (Comorbidities)
Older adults: Risk of injury or death increases after age 55; or
SBP less than 110 might represent shock after 65 years; or
Low impact mechanisms (e.g. ground level falls) may result in severe injuries; or
Children: Should be triaged preferentially to pediatric-capable trauma centers; or
Anticoagulation and Bleeding Disorders
• Patients with head injury are at high risk for rapid deterioration; or
• Without other trauma mechanism: triage to burn facility; or
• With trauma mechanism: triage to trauma center; or
Pregnancy greater than 20 Weeks; or
EMS Provider Judgment
Salem Hospital Specific Reminders
Near Drowning (suspected spinal injury)
Near Hanging
GLF, GCS less than or equal to 14 with suspected head injury, age 55 and on
Anticoagulation (ASA / Ibuprofen not included)
Marion & Polk County Regional Treatment Protocols
Operations – Page 25 of 27
TRAUMA SYSTEM
II. MEDICAL DIRECTION:
A. Off-line medical direction for trauma patients is controlled by the Treatment Protocols.
B. OLMC is provided. OLMC may override off-line medical direction. Any instances where
this occurs will be reported to the EMS Office.
III. COMMUNICATIONS:
A. Communications with Receiving Facility (RF):
The following information will be provided:
1. Unit number and the location of the incident.
2. Number of patients.
3. Age and sex of the patients.
4. Trauma system entry criteria and vital signs.
5. Glasgow Coma Scale.
6. ETA to Trauma Center.
7. Patient destination based on incident location or request.
B. Communications from Receiving Facility or OLMC to the paramedics in the field
will be as follows:
1. RF will inform the paramedic if more information is needed by the trauma
2. RF will inform the paramedic if the chosen trauma center is unable to receive the
patient and will assist in designating an alternate destination.
3. In the event that there are multiple Trauma System entries, RF will assist the
paramedic at the scene in determining the destinations of all patients.
IV. TRAUMA CENTER DESTINATION:
A. Hospital Levels: LEVEL II – Salem Hospital, Good Samaritan Hospital (Corvallis),
LEVEL III – Samaritan Albany Hospital, LEVEL IV – Samaritan Lebanon Hospital, Santiam
Memorial Hospital, Silverton Hospital
B. Patients or Guardians Request: If the alert, competent patient or his/her competent
guardian demands transport to a specific hospital, the EMT must honor that request and
notify the TCC immediately.
C. Multiple Patients: From the same scene, all patient destinations are to be that
assigned by the above service areas unless the designated Trauma Center advises the
TCC that the facility cannot accept additional patients. In this instance, the Trauma
Communications Center (TCC) will assist the paramedic in determining patient
destinations. Hospital trauma patient numbers: Albany – 2, Corvallis – 10, Lebanon – 2,
Riverbend (Springfield) – 15, Salem – 15, Santiam – 2, Silverton – 3.
D. Diversion Toto Local Hospital: If the paramedic is unable to establish an airway, the
patient should be transported to the nearest acute care facility. In the event this occurs,
the Receiving Trauma Facility should be notified of the diversion.
Marion & Polk County Regional Treatment Protocols
Operations – Page 26 of 27
TRAUMA SYSTEM
V. MODE OF TRANSPORT:
An air ambulance should be used when it would reduce total pre-hospital time by 10 minutes
or greater. This is usually achieved whenever the ground transport time will exceed 20
minutes (Scene is more than 15 miles from Salem, or other circumstances exist).
VI. PATIENT EVALUATION PROTOCOL:
A. Treatment Priority Should Be Approached In This Order:
1. Airway Maintenance (including control of the cervical spine)
3. Control of circulation and hemorrhage
4. Treatment of shock
5. Neurological examinations
6. Complete secondary survey.
7. Cardiac monitor should be considered specifically if blunt torso trauma is present, a
medical issue was the possible cause of injury or if there is concern for dysrhythmia.
8. Splinting of fractures
THESE ARE POTENTIALLY CRITICALLY INJURED PATIENTS; TREAT THEM AS
VII. SCENE TIME:
After gaining access to the patient, scene time should not exceed ten minutes for any patient
who is entering the Trauma System. Plan to start IV/IOs and initiate other care once en-route
to the hospital if necessary.
Marion & Polk County Regional Treatment Protocols
Operations – Page 27 of 27
MULTIPLE PATIENT SCENE
MASS CASUALITY INCIDENT
Marion & Polk County Regional Treatment Protocols
MCI & MPS – General Guidelines
1. TRIAGE PROCEDURE
Triage is defined as the sorting of, and allocation of treatment to, multiple
patients and disaster victims according to a system of priorities designed
to maximize the number of survivors. The function of Triage is most
useful and needed in large disaster settings where local resources are
MPS- Multiple Patient Scene. Defined as more than 5 and less than10
patients on an incident.
MCI- Multiple Casualty Incident. Defined as 10 or more identified patients.
Triage is performed by the assigned team(s), whose knowledge of triage
will allow them to quickly evaluate and place the victim into an
appropriate category. The "START" and "RPM" system of triage is the
initial triage procedure for MCI/MPS scenes for jurisdictions covered by
these protocols. The START Flow Chart is located in this section and
provides a quick reference guide for triage in the field.
In general, cardiopulmonary resuscitation of patients should not be
initiated unless staffing allows for immediate treatment of all immediate
and delayed patients.
START- Simple Triage and Rapid Transport
RPM- Respirations, Perfusion, Mental Status
Marion County Fire Defense Board MCI Plan can be referenced for
further details of MCI incidents.
START TRIAGE FLOW CHART
Rev. 9/2010 Marion & Polk County Regional MCI Treatment Protocols – Page 1 of 1
Marion & Polk County Regional Treatment Protocols
DECONTAMINATION- General Guidelines
DECONTAMINATION ZONE
Note: All victims suspected of ingestion or significant exposure to hydrogen cyanide
solution require decontamination. Others may be transferred immediately to the Support
A. Decontamination Operations
1. Request HAZMAT for assistance with decontamination operations
2. Notify hospital of pending transport(s) and self-reporting of contaminated victims
B. Triage – Considerations for high-priority decon
1. Priority 1: Victims closest to point of release
2. Priority 2: Victims reporting exposure to vapor or aerosol
3. Priority 3: Victims with evidence of liquid on skin or clothing
4. Priority 4: Victims with serious medical symptoms (SOB, seizures, etc.)
1. Ambulatory victims who are able and cooperative should assist with their own
decontamination.
a. Instruct victims to rapidly remove contaminated clothing, jewelry, and
other personal items while flushing exposed skin and hair with high-flow,
low-pressure water
b. If available, wash with soap
c. Rinse thoroughly with water
d. Double bag contaminated clothing and personal belongings; track belongings if
e. Irrigate exposed or irritated eyes with plain water or saline for 5 minutes.
f. Continue eye irrigation during other basic care or transport.
g. Remove contact lenses if present and easily removable without additional
trauma to the eye
D. Non-Ambulatory Victims
1. Victims unable to walk through gross decon or remove their own clothing will
need to be assisted by 2-4 personnel
a. Place the victim on a backboard
b. Remove and collect clothing, jewelry, and other personal items; track
belongings if feasible.
c. Flush exposed skin and hair with high-flow, low-pressure water, starting at the
head and moving towards the feet
d. If soap is available, wash all skin folds, genitals, hair, underarms, etc., avoiding
vigorous irritation of any burns, abrasions, or other injuries
e. Protect airway from secondary contamination; consider the use of BVM to
provide oxygen and for water-tight seal
f. One person (usual y at the head) should be assigned to EMS/Airway care to
during decontamination
E. Transfer to Support Zone as soon as decontamination is complete.
Marion & Polk County Regional Haz-Mat Treatment Protocols
DECONTAMINATION- General Guidelines
Marion & Polk County Regional Haz-Mat Treatment Protocols
HYDROGEN CYANIDE
CAS 74-90-8
UN 1051
Synonyms include formic anammonide and formonitrile. Aqueous solutions are referred to as
hydrocyanic acid and prussic acid. Hydrogen cyanide is very volatile, producing potentially lethal concentrations at room temperature. At temperature below 78°F, hydrogen cyanide is colorless or pale blue liquid
(hydrocyanic acid); at higher temperatures, it is a colorless gas. It has a faint bitter almond odor
and a bitter burning taste. It is soluble in water. Hydrogen cyanide is lighter than air.
PRECAUTIONS
A. Persons whose clothing or skin is contaminated with cyanide containing solutions can
secondarily contaminate personnel by direct contact or through off-gassing vapor.
1. Avoid dermal contact with cyanide-contaminated victims and their bodily fluids.
2. Take special care with victims who may have ingested cyanide, as
cyanide salts dissolve in the stomach and react with hydrochloric acid
to produce hydrogen cyanide gas. Transport patients in vehicles with
windows opened and/or good ventilation. These patients who meet
Death in the Field criteria should be considered a Hot Zone.
3. Victims exposed only to hydrogen cyanide gas do not pose contamination risks to
B. Hydrogen cyanide is a volatile flammable liquid at room temperature; as a gas, it is
flammable and potentially explosive.
C. Hydrogen cyanide is absorbed well by inhalation and can produce death within minutes.
1. Substantial absorption can occur through intact skin if vapor concentration is
2. Exposure by any route may cause systemic effects.
HEALTH EFFECTS
HCN is classified a systemic (chemical) asphyxiant. Cyanides interfere with the intracellular
utilization of oxygen resulting in cel ular dysfunction and cel death. Effects are most profound
and first evidenced in the CNS and cardiovascular system. Initial symptoms may include CNS
excitation and cardiovascular compensation fol owed by depression/col apse of both systems.
ROUTES OF EXPOSURE
1. Hydrogen cyanide is readily absorbed from the lungs; symptoms of poisoning
begin within seconds to minutes
2. The odor of cyanide does not provide adequate warning of hazardous concentrations.
Perception of the odor is a genetic trait (20% to 40% of the general
population cannot detect hydrogen cyanide); also rapid olfactory fatigue
can occur.
B. Skin/Eye Contact: Exposure to hydrogen cyanide can cause skin and eye irritation and
can contribute to systemic poisoning with delayed symptoms.
C. Ingestion of hydrogen cyanide solutions or cyanide salts can be rapidly fatal
Marion & Polk County Regional Haz-Mat Treatment Protocols
HYDROGEN CYANIDE
SIGNS AND SYMPTOMS
A. Signs and symptoms usual y develop rapidly. Initial symptoms are nonspecific and include
excitement, dizziness, n/v, HA and weakness.
B. Progressive signs and symptoms may include: Drowsiness, tetanic spasm, convulsions,
hal ucinations and loss of consciousness.
C. Cardiovascular – Can cause various life threatening dysrhythmias. D. Respiratory
1. Victims may complain of shortness of breath and chest tightness 2. Pulmonary findings may include rapid breathing and increased depth of respiration 3. As poisoning progresses, respirations become slow and gasping; cyanosis may be
present, and pulmonary edema may develop
RESCUER PROTECTION
A. Respiratory protection: Pressure demand self-contained breathing apparatus (SCBA) is
recommended in response situations that involve exposure to potentially unsafe levels
of hydrogen cyanide
B. Skin protection: Chemical protective clothing is recommended because both hydrogen
cyanide vapor and liquid can be absorbed thorough the skin to produce systemic
DECONTAMINATION ZONE
A. Refer to Decontamination page. B. Transfer to Support Zone as soon as decontamination is complete.
SUPPORT ZONE
A. Be certain that victims have been decontaminated properly. Additional decontamination
may be required for exposed skin and eyes.
B. Decontaminated victims or those exposed only to vapor, pose no serious risks of
secondary contamination to rescuers. In these cases, Support Zone personnel require
no specialized protective gear.
Marion & Polk County Regional Haz-Mat Treatment Protocols
HYDROGEN CYANIDE
TREATMENT
Patients who rapidly regain consciousness and who have no other signs or symptoms may not
require antidote treatment. Patients who remain comatose or develop shock should be
treated promptly with the antidotes per OLMC direction. In cases of ingestionemesis and
activated charcoal are contraindicated.
A. High flow oxygen, establish IV access, apply cardiac monitor and secure protected airway
following Airway Management protocol.
B. If Cyanide Toxicity is suspected based on findings (soot in mouth, nose or oropharynx,
know exposure) and patient is comatose, in cardiac or respiratory arrest, or has persistent
hypotension despite fluid resuscitation: 1. Administer Hydroxocobalamin (Cyanokit®) 5 g IV as an infusion and monitor for
clinical response. Contact OLMC for advice regarding a second 5 g dose.
2. If Hydroxocobalamin (Cyanokit®) is not available, then administer Sodium Thiosulfate
50 ml of 25% solution over 10-20 minutes. Pediatric dose is 1.65 ml/kg
3. Do NOT administer Hydroxocobalamin (Cyanokit®) and Sodium Thiosulfate to the
4. Treat other presenting symptoms per appropriate protocol. 5. Initiate emergent transport to appropriate facility. 6. Patients in shock or having seizures should be treated according to existing protocols.
These patients may be seriously acidotic; consider giving sodium bicarbonate 50 mEq,
with OLMC direction.
C. MULTI-CASUALTY TRIAGE (MPS or MCI) - Patients who have only brief inhalation
exposure and mild or transient symptoms may be discharged.
Marion & Polk County Regional Haz-Mat Treatment Protocols
HYDROGEN FLOURIDE
CAS 7664-39-3
UN 1052 (Anhydrous)
UN 1790 (Solution)
Synonyms include fluoric acid, hydrofluoride, hydrofluoric acid, and fluorine monohydride.
Hydrogen fluoride is a colorless, corrosive fuming liquid or gas (boiling temperature 67oF) with
a strong irritating odor. It is usually shipped in cylinders as a compressed gas. Hydrogen
fluoride readily dissolves in water to form colorless hydrofluoric acid solutions. Dilute
solutions are indistinguishable from water. It is present in a variety of over-the-counter
products at concentrations of 6% to 12%.
PRECAUTIONS
A. Victims whose clothing or skin is contaminated with HF liquid, solution or condensed vapor,
can secondarily contaminate response personnel by direct contact or through off-gassing
B. Inhalation hazards result not only from HF gas but also from fumes arising from
concentrated hydrogen fluoride liquid or from the patient's bodily fluids.
C. Rapid flushing of exposed areas with water is critical. HF is water-soluble.
HEALTH EFFECTS
The toxic effects of hydrogen fluoride are due primarily to the fluoride ion. The fluoride ion
combines with endogenous calcium and magnesium to form insoluble calcium fluoride and
magnesium fluoride.
A. This results in cell destruction and local bone demineralization
B. Life threatening hypocalcemia, hypomagnesemia, and hyperkalemia can occur.
C. The adverse action of the fluoride ion may progress for several days
ACUTE EXPOSURE
A. RespiratoryDue to HF's water solubility, effects of exposure generally occur in the
upper airway including the glottis. However, people incapacitated in large clouds of HF can
have severe deep lung injury.
1. Mild effects mucous membrane irritation, cough and narrowing of the bronchi.
2. Severe effects.
a. Almost immediate narrowing and swelling of the throat, causing upper airway
b. Lung injury may evolve rapidly or may be delayed in onset for 12 to 36 hours.
c. Pulmonary edema and constriction of the bronchi. Partial or complete lung col apse
d. Pulmonary effects can result even from splashes on the skin.
B. DermalDepending on the concentration and duration of exposure, skin contact may
produce pain, redness of the skin, and deep, slow healing burns with symptoms delayed up
to 24 hours. HF can penetrate tissues deeply, causing both local cellular destruction and
systemic toxicity.
Marion & Polk County Regional Haz-Mat Treatment Protocols
HYDROGEN FLOURIDE
C. Ocular
1. Mild effects rapid onset of eye irritation
2. More severe effects may result from even minor hydrofluoric acid splash include,
sloughing of the surface of the eye, swelling of the structures of the eye, and cell death
due to lack of blood supply. Potential y permanent clouding of the eye surface may
develop immediately or after several days
D. Gastrointestinal
1. A small amount of ingested HF is likely to produce systemic effects including acid-base
imbalance and may be fatal.
2. Ingestion of hydrofluoric acid may cause corrosive injury to the mouth, throat and
esophagus as well as inflammation and bleeding of the stomach.
3. Nausea, vomiting, diarrhea and abdominal pain may occur
E. Electrolyte disturbancesexposure by any route may result in systemic effects:
hypocalcemia and/or hypomagnesemia and/or hyperkalemia.
PREHOSPITAL MANAGEMENT
Rescuer Protection
A. SCBA is recommended in response situations that involve exposure to potential y unsafe
levels of hydrogen fluoride
B. Skin protection: Chemical protective clothing, i.e. level A or level B, is recommended
because skin exposure to either vapor or liquid may cause severe consequences.
DECONTAMINATION ZONE
A. Victims exposed only to hydrogen fluoride gas or vapor who have no skin or eye irritation
do not need decontamination, they may be transferred immediately to the Treatment Area.
B. Rescuer Protection: If exposure levels are determined to be safe, personnel wearing a lower
level of protection than that worn in the Hot Zone may conduct decontamination.
C. ABC Reminders
1. Quickly ensure a patent airway anticipate airway edema.
2. Stabilize the cervical spine with a c-col ar and a backboard if trauma is suspected
3. Administer supplemental O2
4. Assist ventilation with a bag-valve-mask device if necessary
D. Basic decontamination
1. Victims who are able and cooperative may assist with their own decontamination
a. RADLY REMOVE CONTAMINATED CLOTHING while flushing exposed
skin and hair with plain water for 15 minutes.
b. If either of the treatments recommended below is available, water flushing may be
reduced to 5 minutes and the treatment should be started immediately.
1. 2.5 G calcium gluconate in 100 ml of water soluble lubricant such as KY Jelly,
2. OR, ml of 10% calcium gluconate per ounce of KY Jelly
c. Double bag contaminated clothing and personal belongings
Marion & Polk County Regional Haz-Mat Treatment Protocols
HYDROGEN FLOURIDE
2. Irrigate exposed or irritated eyes with plain water or saline or 5 minutes
a. Continue eye irrigation during other basic care or transport
b. Remove contact lenses if present and easily removable without additional trauma to
3. In case of ingestion, do not induce emesis or administer activated charcoal
a. Victims who are conscious and able to swallow should be given 4 to 8 ounces of water
b. If available, also give 2 to 4 ounces of an antacid containing magnesium (e.g., Maalox,
Milk of Magnesia) or calcium (e.g., TUMS)
4. As soon as basic decontamination is complete, move the victim to the Treatment Area.
TREATMENT
Be certain that victims have been decontaminated properly. Treatment Area personnel require
no specialized protective gear if victims have undergone decontamination.
A. ABCs, C-spine (PRN), Pulse Oximetry, ECG obtain baseline QT interval a (may be of
benefit for this).
B. Treat patients who are or have per existing protocols.
C. Observe for signs of hypocalcemia and contact OLMC regarding treatment with Calcium
1. ECGprolonged Q-T interval or QRS or ventricular dysrhythmias. 2. OtherMuscular tetany. This is probable after ingestion of even small amounts of HF.
D. For inhalation victims.
1. Administer 2.5% Calcium Gluconate by nebulizer. Mix 1cc of 10% Calcium Gluconate
with 3ccs of Normal Saline into the nebulizer.
2. If wheezes are present consider use of Albuterol per Respiratory Distress protocol.
E. Minor Burns.
1. Initially, the health care provider should wear rubber or latex gloves to prevent
secondary contamination
2. Vigorously massage the burned areas with Calcium Gluconate gel2 ml of 10% Calcium
Gluconate per ounce of KY Jelly
3. Continue this procedure until pain is relieved or more definitive care is rendered
F. Hand Exposure
1. Subungual (under the nail) burns often do not respond to immersion treatment. The
treatment for hand burns requires expert assistance; consult with OLMC
2. Treatment of hand exposures can be accomplished by placing Calcium Gluconate gel
into an exam glove and placing the glove on the affected hand.
G. Optical ExposureIrrigate exposed eyes with a 1% aqueous solution of Calcium
Gluconate (10 ml of 10% solution in 90 ml of sterile saline in Buretrol) using a nasal cannula.
1. Up to 500 ml over 1 to 2 hours may be used
2. If Calcium Gluconate is not available, use normal saline for irrigation.
MULTI-CASUALTY TRIAGE (MPS or MCI)
Consult with the OLMC for advice regarding triage of multiple victims. Persons who have had
only minor or brief exposure to hydrogen fluoride gas or vapor and are initially asymptomatic
are not likely to develop complications. See Multiple Toxic Exposure Protocol. Marion & Polk County Regional Haz-Mat Treatment Protocols
IDENTIFICATION
CAS 56-38-2
UN 2783
Synonyms include Alkron, Al eron, Danthion, DNTP, DPP, Ethyl Parathion, Etilon, E-605,
Stathion, Sulphos, and Thiophos. The term organophosphate (OP) is general y understood to mean an organic derivative of
phosphoric or similar acids. There are many different OPs and they differ to some extent in
their properties. Many OPs inhibit an enzyme known as acetylcholinesterase. This is a class
effect of OPs, but not al OPs (e.g. glyphosate) demonstrate this effect. Inhibitors of
acetylcholinesterase affect certain nerve junctions in animals, as well as parasympathetic effector
sites (the heart, lungs, stomach, intestines, urinary bladder, prostate, eyes and salivary glands).
By inhibiting the enzyme acetylcholinesterase, OPs prevent the nerve junction from functioning
properly.
PRECAUTIONS
A. Organophosphates are highly contaminating.
B. Victims whose skin or clothing is contaminated with liquid or powdered organophosphate
can secondarily contaminate response personnel by direct contact or off gassing of solvent
C. Clothing and leather goods (e.g., belts or shoes) cannot be reliably decontaminated; they
should be incinerated.
D. Special care should be taken to avoid contact with the vomitus of a patient who has
ingested organophosphate.
PHYSICAL PROPERTIES
A. At room temperature, organophosphate powders or combustible liquids.
B. Organophosphates are almost insoluble in water, slightly soluble in petroleum oils, and
miscible with many organic solvents. Accordingly, most commercial products contain
hydrocarbon solvents.
C. Organophosphates have low vapor pressures, thus significant inhalation is unlikely at normal
temperatures (Exception: Dichlorvos (a.k.a. DDVP and Vapona) when in a poorly ventilated
confined space). However, the hydrocarbon solvents remain volatile and flammable, as well
as possessing toxic properties.
ROUTES OF EXPOSURE
1. Toxic inhalation of organophosphate vapor is unlikely at ordinary temperatures because
of its low volatility, but toxic effects can occur after inhalation of organophosphate
2. The hydrocarbon solvents (most commonly toluene and xylene) used to dissolve
organophosphate are more volatile than organophosphate itself, and toxicity can result
from inhalation of solvent vapor as well
B. Skin/Eye ContactOrganophosphates are rapidly absorbed through intact skin or eyes,
contributing to systemic toxicity.
C. IngestionAcute toxic effects. May be rapidly fatal.
Marion & Polk County Regional Haz-Mat Treatment Protocols
HEALTH EFFECTS
A. Introduction:
1. Organophosphates are known as cholinesterase inhibitors. Normally, the
neurotransmitter acetylcholine (ACh) is broken down by acetylcholinesterase (AChE).
Organophosphates inhibit the activity of AChE and thus ACh is not broken down. The
resulting accumulation of ACh overstimulates ACh receptors (aka cholinergic
receptors) within the central and peripheral nervous systems. The toxic effects of
organophosphates result from this overstimulation of ACh receptors. There are two
types of ACh receptors, muscarinic and nicotinic.
2. Signs and symptoms of poisoning vary according to age, dose, and concentration
a. CNS effectsIrritability, nervousness, giddiness, fatigue, lethargy, impairment of
memory, confusion, slurred speech, visual disturbance, depression, impaired gait,
convulsions, loss of consciousness, coma, and respiratory depression. CNS effects
can be some of the earliest symptoms.
b. PNS Effectsnicotinic and muscarinic stimulation can provide opposing effects. In
general, nicotinic signs and symptoms predominate early in organophosphate
poisoning, while muscarinic signs and symptoms predominate later.
1. Muscarinic effects SLUDGE (Salivation, Lacrimation, Urination,
Defecation, Gastroenteritis, Emesis), or DUMBELS (Diarrhea, Urination,
Miosis, Bradycardia, Bronchorrhea, Bronchospasm, Emesis, Lacrimation,
Salivation, Secretion, Sweating)
2. Nicotinic effects MTWHF (Mydriasis, Tachycardia, Weakness,
Hypertension, Hyperglycemia, Fasciculations, Flaccidity)
PREHOSPITAL MANAGEMENT
• HOT ZONE
A. Respiratory Protection: SCBA is recommended in response situations that involve exposure
to potential y unsafe levels of organophosphates.
B. Skin Protection: Chemical-protective clothing is recommended because organophosphates
are rapidly absorbed through the skin and may cause systemic poisoning.
• DECONTAMINATION ZONE
All victims suspected of organophosphate ingestion, or substantial exposure to aerosolized
organophosphates, or who have skin or eye exposure to liquid or powdered
organophosphates require thorough decontamination.
BASIC DECONTAMINATION
Follow Decontamination General Guidelines. Then, move the victim to the Treatment Area
upon completion.
SIGNS AND SYMPTOMS
A. Mild poisoning HA, n/v, abdominal cramps, diarrhea.
B. Moderate poisoning: Generalized muscle weakness and twitching, slurred speech, pinpoint
pupils, excessive secretions, and shortness of breath.
C. Severe poisoning: Seizures, skeletal-muscle paralysis, respiratory failure, and coma. Marion & Polk County Regional Haz-Mat Treatment Protocols
TREATMENT
A. Secure protected airway in cases of respiratory compromise per Airway Management
B. There is no contra-indication to the use of paralytic agents is in this setting, however both
Succinylcholine and Vecuronium will have a significantly sustained duration of
paralysis in the presence of organophosphates.
C. The initial intravenous dose of atropine in adults should be determined by the severity of
symptoms. In seriously poisoned patients, very large doses may be required. Alterations of
pulse rate and pupil ary size are unreliable indicators of treatment adequacy. Atropine
works only to correct muscarinic effects.
a. Mild poisoning1 mg.
b. Moderate poisoning1 to 2 mg.
c. Severe poisoning2 to 5 mg.
Doses should be repeated every 5 minutes until excessive secretions and sweating have been
controlled. Note: The Mark 1 Auto-Injector atropine is 2 mg; Duodote is 2.1 mg
D. Administer Pralidoxime (2-PAM), if profound weakness or paralysis present. E. Moderate symptoms1,200 mg (2 amps Mark 1 injector) F. Severe symptoms1,800 mg (3 amps Mark 1 injector)
G. CAUTION: When administering 2-PAM intravenously, administer at rate of less than 200
mg/minute, (4 mg/minute for children).
H. Patients who are comatose, hypotensive, have seizures or cardiac dysrhythmias should be
treated according to ALS protocols.
TRANSPORT TO MEDICAL FACILITY
A. Report to OLMC, and the receiving medical facility, the condition of the patient, treatment
given, and estimated time of arrival at the medical facility.
B. If organophosphate has been ingested:
1. Prepare the ambulance in case the victim vomits toxic material.
2. Prepare several towels (or other absorbent material) and open plastic bags to quickly
clean up and isolate vomitus
MULTI-CASUALTY TRIAGE (MPS or MCI)
Patients who have histories or evidence suggesting substantial exposure and all persons who
have ingested organophosphate should be transported to a medical facility for evaluation.
A. Others may be discharged from the scene after their names, addresses, and telephone
numbers are recorded.
B. They should be advised to seek medical care promptly if symptoms develop or recur.
C. Request Chem-Pack from Salem Fire.
PEDIATRIC PATIENTS:
Atropine: In children, dosages range from 0.02 to 0.05 mg/kg.
Pralidoxime: Pediatric dose: 25 to 50 mg/kg and must be given slowly via IV (4 mg/min.)
Marion & Polk County Regional Haz-Mat Treatment Protocols
Source: http://www.staytonfire.org/Files/EMS%20Protocols.pdf
Rapid oscillometric blood pressure measurement compared toconventional oscillometric measurementSteven A. Yarowsa, Ketul Patelb and Robert Brooka Background There have been few reports studying the necessary interval between blood pressure Oscillometric blood pressure measurement has largely measurements, after the initial rest period.
Observatorio de Comercio Exterior Informe de omercio Actualización Exterior Intercambio con Principales Socios Departamento de Economía Gabriel Molteni Gonzalo De León Martín Mattiazzi Panorama General En el mes de febrero de 2014 las exportaciones argentinas fueron de 5393 millones de dólares, lo que representó un crecimiento de 3,1% en relación al mes anterior. En la comparación interanual se detectó una caída de 6,1%, explicada por un descenso en las cantidades vendidas de 5% y una disminución de los precios de 1 por ciento.















