Goteborgsuniversitet.se





Typhoid fever perforation in Kathmandu, Nepal
- A retrospective study of risk factors and antibiotic treatment
Author: Isabel Vigmo, Medical Student at the Sahlgrenska Academy
University of Gothenburg, Sweden
Supervisors: Yogendra Singh, MD, PhD, Professor of Surgical Oncology
Department of Surgery, Tribhuvan University Teaching Hospital, Kathmandu, Nepal
Göran Kurlberg, MD, PhD, Associating Professor of Surgery
Sahlgrenska University Hospital/ Campus Östra, Göteborg, Sweden
Introduction: Typhoid fever is a global health problem, causing significant morbidity and mortality
throughout the developing world. A recent survey estimated a global number of 21.7 million
illnesses and 217.000 deaths in the year 2000. Due to poverty and poor sanitary conditions, typhoid
fever remains a major health problem in Nepal. A frequent complication of typhoid fever is
perforation of the terminal ileum. Mortality rates rating from 20 - 40 % are most commonly
Aims and objectives The aim of this study was to trace risk factors for enteric perforation in
patients with typhoid fever as well as to assess health care factors that influence the outcome of
Methods and materials This retrospective study was conducted at the Tribhuvan University
Teaching Hospital in Kathmandu, Nepal. The inclusion criteria for participation was the diagnosis
ileal perforation due to typhoid fever in adult patients (≥ 16 years). During June 14th 2006 and May
22th 2010, 30 patients underwent surgery for ileal perforation due to typhoid fever and were
therefore enrolled in the study. Using a collection form, data was abstracted from the medical
records of these patients and then analyzed.
Results There were 22 males and eight females (male : female ratio 2.75 : 1) with a mean age of
28.1 ± 10.0 years. The mean duration of symtoms was 14 ± 11.0 days (range 5 - 60 days) and all
patients suffered from fever and abdominal pain. A majority (16) of the perforations occured in the
second week of disease. Primary repair was applied to 18 patients, while 11 patients received wedge
resection and one patient received ileostomy. Reperforation occured in one patient and mortality
developed in two (7 %) patients.
Conclusions Typhoid fever remains a major health problem in Nepal. The leading cause of
mortality in typhoid fever is ileal perforation, which is a complication that possibly could be
prevented. Identification and appropriate antibiotic treatment of typhoid fever would reduce
morbidity and mortality. Early and appropriate surgical intervention is vital for the survival of
typhoid fever perforation.
Keywords : Typhoid fever perforation, Risk factors, Antibiotic treatment
Table of contents
1. Introduction. 1
1.1 Microbiology. 2
1.2 Transmission. 2
1.3 Epidemiology. 3
1.4 Pathogenic mechanisms. 3
1.5 Clinical features. 4
1.6 Diagnostic methods. 4
1.7 Differential diagnosises. 6
1.8.1 Antimicrobial therapy. 6
1.8.2 Surgical management of intestinal perforation. 7
1.9 Study area : Kathmandu, Nepal. 7
2. Problem of interest. 9
3. Aims and objectives. 10
4. Method and materials. 11
6. Case report. 18
7. Discussion. 20
8. Conclusions. 24
9. Acknowledgements. 25
10. References. 26
Typhoid fever is a global health problem causing great suffering in developing countries where
unhealthy living conditions prevail. Also known as enteric fever, this potentially fatal multisystemic
disease is caused by several serotypes of Salmonella Enterica including S. Typhi and S. Paratyphi.
Untreated or suboptimally treated typhoid fever may lead to perforation of the terminal ileum. The
complication requires surgical management and is still associated with considerable morbidity and
mortality [1].
Whereas typhoid fever was a major cause of death in the United States and Europe in the 19th
century, the provision of clean water and good sewage systems led to a dramatic decrease in the
incidence of typhoid in these regions. Today, the disease remains endemic in many areas of the
developing world where poor sanitary conditions remain and facilitate transmission . Typhoid fever
is estimated to cause over 26 million infections and over 200.000 deaths annually. South-central
Asia and south-east Asia have been identified as regions with high incidence of typhoid fever
(>100/ 100.000 cases/year), whereas the rest of Asia, Africa, Latin America and Oceania excluding
Australia and New Zealand are reported to have a medium incidence (10-100 / 100.000 cases /
year) [2]. Most cases of typhoid fever in the developed world are attributed to travellers returning
from endemic areas whilst domestically aquired disease is infrequently reported [3].
Kathmandu, the capital city of Nepal, has previously been coined a typhoid fever capital of the
world [4]. Whereas typhoid fever in rural areas is not uncommon, the living conditions of the urban
centre of Kathmandu predispose for the constant transmission of the disease to a greater extent.
Typhoid fever is largely an endemic disease but it has an epidemic potential. The disease also has a
very high social and economic impact because of the hospitalization of patients with acute disease
and the complications and loss of income attributable to the duration of the clinical illness.
If typhoid fever remains untreated, or if the antibiotic treatment is inadequate, the disease can cause
fatal complications such as intestinal bleeding and ileal perforation. Despite the high morbidity,
relatively little is known about risk factors for enteric perforation in patients with typhoid fever.
Furthermore, the emergence of multi-drug resistant strains of S. Typhi and S. Paratyphi makes
treatment increasingly difficult. Given the morbidity and mortality and considering the reduced
effectiveness of commonly used antibiotics there is an increasing urgency to control the disease and
its complications.
The majority of cases of typhoid fever are attributed to the gram-negative bacterium Salmonella
Enterica serotype Typhi (S. Typhi). It is a rod-shaped facultative bacteria which belongs to the
family Enterobacteriaceae. Only humans are affected by this highly virulent and invasive pathogen.
For every ten cases of S. Typhi infection, there are one or two cases of paratyphoid fever, caused by
the human-adapted S. Enterica serotypes Paratyphi A, Paratyphi B and Paratyphi C [5]. Whereas S.
Paratyphi A infections occur in a significant and rising number, S. Paratyphi B and S. Paratyphi C
are relatively uncommon [6].
The genes for virulence factors cluster in Salmonella pathogenesis islands (SPIs) and their
expression is generally limitied to specific host compartments. SPI-1 encodes a type three secretion
system (TTSS) that translocates bacterial virulence factors into host cells during infection. This
system is essential for the invasion of epithelial cells and it is activated under the conditions present
in the intestinal lumen. SPI-2 is then required for the survival and replication of the bacteria in the
host cells. Unlike most other Salmonella serotypes, S. Typhi expresses the Vi Polysaccaride capsule.
It is an important virulence factor that increases the infectivity of S. Typhi and the severity of the
disease [7].
Humans are the only natural host and reservoir. Typhoid fever is transmitted the fecal- oral route by
ingestion of food or water contaminated by excreta of patients and asymptomatic carriers and is
therefore most common in areas with poor sanitary facilities. The highest incidence occurs where
water supplies serving large populations are contaminated with faeces. The risk of infection
increases as the size of inocula increases and the infectious dose varies between 103 – 108 organisms
given orally [3].
Food often eaten outside the home and handled by chronic or transient infected persons is also a
common source of infection. Other risk factors for increased transmission include recent typhoid
fever in the household, a lack of toilets in the household, drinking unboiled water, not using soap
for hand washing, and sharing food from the same plates as others.
Between 1 % and 5 % of patients with acute typhoid infection have been reported to become
chronic carriers of the infection in the gall bladder, depending on age, sex and treatment regimen
Typhoid fever is a global health problem causing great morbidity and mortality. In 2000, typhoid
fever caused an estimated 21.7 million illnesses and 217.000 deaths throughout the world, whereas
paratyphoid fever caused an estimated 5.4 million illnesses [9]. However, its real impact is difficult
to estimate since the clinical picture is easily confused with those of many other febrile diseases.
The indian subcontinent has previously been reported to have the highest incidence of typhoid fever
worldwide [3]. A recent multicentre study in five Asian countries – China, India, Indonesia, Pakistan
and Vietnam- showed that incidence rates were significantly higher in the south Asian sites
(Pakistan and India) than in South East and North East Asian sites (Vietnam, Indonesia, China)
Infections with S. Paratyphi A are emerging at a rapid rate and are responsible for a growing
proportion of typhoid fever in a number of Asian countries [2]. A study performed by Maskey et al
in Patan Hospital in Kathmandu confirms this trend; the proportion of enteric fever cases caused by
S. Paratyphi A increased fron 17.5 % in 1993 to 34 % in 2003 [11].
Multiple studies have highlighted the seasonal variation of typhoid fever. In Nepal, the heaviest
burden is during the monsoon months from June to August where both the average temperatures
and rainfall peak [4]. This association is believed to be due to contamination of water supply caused
by an overwhelmed and inadequate sewage system.
1.4 Pathogenic mechanisms
After ingesting contaminated food or water, typhoid organisms pass through the pylorus and reach
the small intestine. They penetrate the mucosal epithelium and arrive in the lamina propria, where
they elicit an influx of macrophages that ingest the bacterium but generally does not kill them.
Some bacteria remain within macrophages in the lymphoid tissue of the small intestine, whereas
others are drained into mesenteric lymph nodes. The current opinion is that the typhoid baceria then
reach the bloodstream by drainage via mesenteric lymph nodes to the ductus thoracicus and then the
general circulation. Within 24 hours the bacteria reach an intracellular location in the organs of the
reticuloendothelial system, that is, the spleen, liver and bone marrow. They then reside here during
the incubation period, usually 8-14 days [8]. Clinical symtoms start with bacteremia.
Typhoid fever is not a typical diarrhoeal disease and the intestinal pathology is characterized by
interstitial inflammation with predominantly mononuclear infiltrates, while neutrophils are scarce
[5] . S. Typhi therefore has the ability to cause a systemic infection without stimulating a significant
local inflammatory response but is transferred from the gastrointestinal lumen to the
reticuloendothelial system in a relatively undetected way [12].
The mechanism of intestinal perforation in typhoid fever is hyperplasia and necrosis of Peyer's
patches in the terminal ileum. The lymphoid aggregates of Peyer's patches extend from the lamina
propria to the submucosa, so that in the presence of hyperplasia the distance from the luminal
epithelium to the serosa is bridged by lymphoid tissue. Tissue damage in Peyer's patches occures,
resulting in ulceration, bleeding, necrosis and, in extreme cases, perforation. Both bacterial factors
and the host inflammatory response are believed to contribute to the consequent tissue damage [6].
1.5 Clinical features
The symtoms of typhoid fever can vary broadly in severity and may be altered by co-existing
morbidities and early administration of antibiotics [13]. Since typhoid fever and paratyphoid fever
most often present with similar clinical symtoms and signs, the accurate diagnosis relies on
laboratory confirmation [14].
Acute non-complicated typhoid fever is generally characterized with the sudden onset of prolonged
fever, headache, abdominal discomfort and malaise. The disease often causes diarrhea, especially in
younger children, whereas older children and adults commonly present with unaffected bowel
motions or constipation. Bronchitic cough is common in the early stage of the illness. During the
period of fever, up to 25% of patients show exanthema, on the chest, abdomen and back [8]
The most common complications are sepsis and dehydration. Around 10 % of the patients also
develop intestinal bleeding and ileal perforation [15]. The symtoms of intestinal perforation and
peritonitis is characterized by abdominal tenderness, rebound tenderness, guarding and subsequent
abdominal rigidity. This complication is usually accompanied by a sudden rise in pulse rate and
hypotension. If untreated, ileal perforation leads to generalized peritonitis, septicemia as well as
fluid- and electrolyte derangements.
1.6 Diagnostic methods
Isolation of the causative organism remains the most effective diagnostic method in suspected
typhoid fever. The investigation of choice is therefore the culturing of blood or bone marrow.
However, whilst considered routine, blood culturing of S. Typhi is expensive and requires specialist
facilities and personell. In addition, S. Typhi and S. Paratyphi A are not alwas culturable even if
good microbiological facilities are available [12].
Bone marrow is the most sensitive culture route but this is an invasive procedure and is seldome
performed outside specialist hospitals. Stool and urine becomes positive after the first week of
infection, but their sensitivity is significantly lower then the culturing of blood or bone marrow.
The diazo test is a simple bed side urine test that can be of great value in the early diagnosis of
typhoid fever. It is particularly valuable in developing countries where laboratories are primitive.
The principle of the test is a red colouration given by the foam of the urine when mixed with diazo
reagent. The test is positive in about 90 % of typhoid cases and previous administration of
antibiotics does not interfere with the sensitivity of the diazo test [16].
The first typhoid diagnostic was the Widal test, which measures antibodies against O and H
antigens of S. Typhi. This test is simple to perform, but has only moderate sensitivity and
specificity. Another diagnostic challenge is the fact that many patients reach microbiological
facilities at a late stage of infection or may be self treated with antimicrobials.
Due to these reasons, the diagnosis of typhoid fever is often made on clinical criteria in resource-
poor countries.
Leukopenia is known to be a common laboratory finding aminst patients with typhoid fever who do
not have ileal perforation [6]. This may be explained by a shift of leukocytes from the circulation
into the marginal or, alternatively, by a bone marrow depression. A study performed by Khan et al
has identified leukopenia due to absolute neutropenia with a relative lymphocytosis as a laboratory
finding highly suggestive of typhoid fever [17].
Typhoid fever without ileal perforation is also associated with trombocytopenia, which can be
caused by a decreased production of trombocytes, an increased destruction by the
reticuloendothelial system, or DIC [18].
Additional investigation is needed when ileal perforation due to typhoid fever is suspected.
Abdominal ultrasonography, plain abdominal radiography and paracentesis are the recommended
diagnostic methods. Typical findings for ileal perforation include free abdominal fluid,
hepatosplenomegaly, air-fluid levels, and free air under the diaphragm [6]. Fecaloid or purelent
fluid upon paracentesis is also consistent with this diagnosis. In some cases, the diagnosis is
ultimately based on the typical intraoperative findings of a perforation of the antimesenteric border
of the terminal ileum. A definitive confirmation of the diagnosis is obtained by an ulcer edge biopsy
and a lymph node culture.
1.7 Differential diagnosises
As previously stated, the diagnosis of typhoid fever is often based on clinical features in developing
countries. This poses a problem, since typhoid fever may mimic many common febrile illnesses.
The differential diagnosises include malaria, sepsis with other bacteria pathogens and infections
caused by intracellular organisms such as tuberculosis and brucellosis. Viral infections, such as
dengue fever and acute hepatitis should also be taken into consideration [13]. Furthermore,
infections with Rickettsia Typhi are common in Kathmandu and offer similar clinical symtoms on
presentation [4].
Typhoid fever perforation should be considered in patients presenting with a high fever for a long
period and an acute abdomen. Common differential diagnosises are perforated appendicitis,
duodenal ulcer or other forms of acute abdomen.
General principles for the management of typhoid fever include adequate rest, hydration and
correction of fluid-electrolyte imbalance. Antipyretic treatment is also required. More than 90 % of
patients can be managed at home with oral antibiotics and reliable care [8]. Regular follow-ups are
recommended in order to detect complications and clinical relapse [13].
1.8.1 Antimicrobial therapy
The prognosis for a patient with typhoid fever is dependent on the rapidity of diagnosis and
accurate treatment. Appropriate antibiotic treatment is critical in curing typhoid with minimal
complications. Furthermore, the outcome is also affected by the patient's age, general state of
health and nutrition.
The current recommendation for the treatment of uncomplicated typhoid fever is the use of
fluoroquinolones as a first line therapy. Fluoroquinolones, such as ofloxacin, ciprofloxacin,
fleroxacin and perfloxacin are highly active and generally well tolerated when compared to the
former first-line drugs chloramphenicol, ampicillin, amoxicillin and trimethoprim-
A challenge in the therapy of typhoid fever is the changing of antibiotic sensitivity patterns and the
emergence of high level resistant cases. Multi-drug resistance (MDR) strains of S. Typhi – that is,
resistance to chloramphenicol, ampicillin, amoxicillin and trimethoprim-sulfamethoxazole, have
emerged. The widespread use of fluoroquinolones has also been associated with decreased
susceptibility as well as resistance to this class of drugs [2].
In the case of intestinal perforation due to typhoid fever, the administration of metronidazole and
gentamicin or ceftriaxone is recommended if a fluoroquinolone is not being used to treat leakage of
intestinal bacteria into the abdominal cavity [8].
1.8.2 Surgical management of ileal perforation
Patients with typhoid fever perforation most commonly show an imbalance in blood electrolytes
and acid-base levels. Anemia is common and may be partially masked by dehydration. For these
reasons, the patients have to go through hemodynamic stabilization before they undergo surgery.
Studies have shown that proved that mortality and morbidity can be decreased if the patient
undergoes a sufficient preoperative resuscitaion [19, 20].
All patients with generalized peritonitis must undergo surgery. The intestinal perforation has to be
closed and a thorough peritoneal lavage needs to be carried out [19].
A variety of surgical procedures can be utilized in the treatment of enteric perforation: simple
closure, resection of the perforation and closure, wedge resection and closure, resection of the
affected terminal ileum and end-to-end anastomosis, loop or end-ileostomy, resection with
ileotransverse colostomy, and resection with right colectomy [21].
In the case of a single perforation, primary repair is suggested after debridement of the perforation
borders. Multiple perforations pose greater problems and the recommended surgical intervention is
a resection of the affected intestine segment followed by an end-to-end anastomosis. Ileostomy is
the suggested procedure for patients in a critical general condition with severe peritonitis [1].
Right hemicolectomy is only recommended in cases of caecal perforation [21].
1.9 Study area : Kathmandu, Nepal
Nepal, officially the Federal Democratic Republic of Nepal, is a landlocked country in South Asia
and the worlds youngest republic. The World Bank calculates that Nepal has a gross per capita
income of 340 US $, which classifies the country as the poorest in Asia. Approximately 55 % of the
population lives below the international poverty line of US $ 1.25 per day. Poverty and political
instability have had obvious harmful effects on infrastructure; while 89 % of the population now
has access to improved water sources, only 45 % of the population has access to improved sanitary
conditions [22]. In 2007, the World Health Organization has estimated the life expectancy at birth to
63 years and the healthy life expectancy to 55 years [23].
The population of Nepal has increased over the past decade to 28.1 million people, a development
that is particularly prominent in the capital city Kathmandu, where people flock in the search of
employment. Almost two million people live in Kathmandu, which therefore is the most populated
area of the country with a density of 1,100 people per square kilometer. Predisposing factors for
typhoid fever are appearant in large parts of Kathmandu, where living conditions faciliate constant
transmission of the disease [4].
The Tribhuvan University Teaching Hospital (TUTH) is a 482-bed tertiary care centre situated in
the centre of Kathmandu. The hospital provides emergency and elective out-patient and in-patient
services and is one of the biggest hospitals in the country. This study was conducted in the
Department of Surgery of the TUTH during May - June 2010.
2. Problem of interest
The most frequent complication of typhoid fever, and also the principal cause of mortality, is
perforation of the terminal ileum. The complication requires surgical management and remains a
significant surgical problem. Early intervention is crucial and mortality rates increase as the delay
between perforation and surgery lengthens. Perforation rates are reported with wide variation.
Butler et al reviewed 15,980 cases of typhoid fever in the worlds literature and reported an overall
2.8 % perforation rate [24] . Mortality rates between 20 - 40 % are most commonly reported for
ileal perforation [24 [36]. Relatively little is known about risk factors for enteric perforation in
patients with typhoid fever. Male gender, short duration of symtoms and leukopenia has previously
been reported as independent risk factors in a case- control study performed by Hosoglu et al [6].
Appropriate antibiotic treatment is critical in curing typhoid fever with minimal complications.
When patients diagnosed with typhoid fever recieve an adequate antibiotic treatment, the
development of enteric perforation will be prevented [1]. However, the emergence of S. Typhi and
S. Paratyphi bacteria with decreased susceptibility as well as resistance to recommended antibiotics
pose a therapheutic problem and increase the risk of complications. The impact on the disease
burden by multi-drug resisance and the emergence of recent fluoroquinolone resistance has not been
estimated. It is however clear that the median time to fever clearance, relapse rates and faecal
carriage rates are higher in patients infected with drug resistant strains than for those with drug
sensitivite strains [3]. In addition, it increases the pool of patients and carriers who can potentially
transmit the disease.
3. Aims and objectives
The aim of this study was to trace risk factors for enteric perforation in patients with typhoid fever
as well as assess health care factors that influence the outcome of these patients.
− To determine age, sex and geographical distribution of the patients− To evaluate various parameters such as temperature, pulse and white blood cell count on
− To asses socioeconomic status defined by occupation− To study how antibiotic treatment influences the outcome of these patients − To study the seasonal variation of ileal perforation due to typhoid perforation
4. Methods and materials
This retrospective study was conducted at the Tribhuvan University Teaching Hospital in
Kathmandu, Nepal. The inclusion criteria for participation was the diagnosis ileal perforation due to
typhoid fever in adult patients (≥ 16 years). During June 14th 2006 and May 22th 2010, 29 patients
underwent surgery for ileal perforation due to typhoid fever and were therefore enrolled in the
study. Using a collection form, data was abstracted from the medical records of these patients.
In order to determine risk factors for ileal perforation in patients with typhoid fever, the following
parameters were examined: age, sex, district, clinical presentation, duration of symtoms, month of
the year, temperature and pulse on admission, antibiotic treatment, duration of hospital stay, white
blood cell count (WBC), lymphocytes, neutrophils, histopathology, widal test, operative findings,
type of surgery, district, occupation and outcome of the patient.
Duration of symtoms were defined in days from the onset of symtoms to hospital admission.
The diagnosis was in most cases based on a pre-perforation history consistent with typhoid fever
combined with typical operative findings of antimesenteric ileal perforation.
5. Results
There were 30 patients with the intraoperative diagnosis of typhoid ileal perforation during the
study period. The mean age of the patients was 28.1 ± 10.0 years. There were 22 males and eight
females, giving a male : female ratio of 2.75 : 1 (Figure 1). The mean duration of symtoms was 14.1
± 11.0 days (range 5-60 days) and all patients suffered from fever and abdominal pain (Table 1).
Age and sex
No. of patients
A majority (53 %) of the perforations occured in the second week of disease. (Table 2) The Widal
test was performed in seven patients only and proved positive in three of these cases. A
histopathologic confirmation of the diagnosis was obtained in six patients.
Most common symtoms in patients with typhoid fever perforations
No. of patients (%)
Duration of illness *
Duration in weeks
* Defined as the time from onset of disease until surgical
Takycardia (>100 bpm) was present in 15 patients and fever (>38.0 °C) in 10 patients. (Table 3)
The mean white blood cell count (WBC) was 10074 ± 4814 cmm³ (range 2200 – 22300 cmm³).
Four patients (13 %) showed leukopenia (WBC <4000 cmm³) , 15 (50 %) showed leukocytosis
(WBC > 11000 cmm³) and 11 (37 %) showed normal WBC. The mean neutrophil count was 73.3 ±
15.5 % (range 25 – 93%) and the mean lymphocyte count 23.3 ± 14.8 % (range 3- 70 %).
Laboratory findings and vital signs on admission
No. of patients %
Leukocyte count Within normal range
Temperature < 36.0 °C
Pulse ≥ 100 bpm
Leukopenia defined as WBC <4000 cmm³ and leukocytosis defined as WBC > 11000
cmm³. Information on puls on admission was missing for one patient.
The majority of perforations were located more than 30 cm from the ileoceacal junction (range 10-
120 cm) (Figure 2) and most perforations were smaller than 2 x 2 cm (Figure 3).
Site of perforation
Size of perforation
No. of patients
A single perforation was seen in 28 patients, wheras one patient had multiple perforations and one
patient had a sealed perforation (Table 4) . The applied surgery types were: primary repair to 18
patients (60%), Wedge resection and anastomosis to 11 patients (37 %) and resection and ileostomy
to one patient (3 %).
Type of surgery and mortality
PC
No. of perforations
contamination Mild
PC: Primary closure; WRA: wedge resection and anastomosis; RI: resection and ileostomy. Peritoneal
contamination was accepted as severe when the drainage amount was more than 1,000 ml, whereas
the contamination was accepted as moderate when the amount was between 500 and 1,000 ml,
and mild when the amount was less than 500 ml. Information of some factors was missing for two
Complications requiring a second surgical treatment developed in two patients (7 %). A
perianastomotic abscess with interloop adhesions and pyoperitoneum developed in one patient and
one patient suffered a reperforation as well as multiple abscesses and adhesions.
Mortality developed in two patients (7%). The cause was sepsis with ARDS and bronchopulmonal
fistula and septic chock with ARDS, respectively.
The mean duration of stay in the hospital was 12.8 ± 9 days (This information was lacking for two
Prior unspecified antibiotic treatment had been given to eight patients before hospital admission.
In the hospital, most patients were administred a combination antibiotic therapy consisting of
metronidazol and a third generation cephalosporin, most commonly Ceftriaxone (Table 5).
The two patients that deceased had both received antibiotic treatment prior to hospital admission. In
the hospital, they received combination therapys consisting of Klindamycin, Amikacin,
Vankomycin and Cefotaxim, Metron, Clavum and Amikacin, respectively.
Outcome of different antibiotic regimes
cephalosporin + generation
cephalosporins + Other
Third generation cephalosporins e.g. Ofromax, Ceftriaxone, Infexime and Xone.
A majority of the ileal perforations occured during the time period February to May (Figure 4).
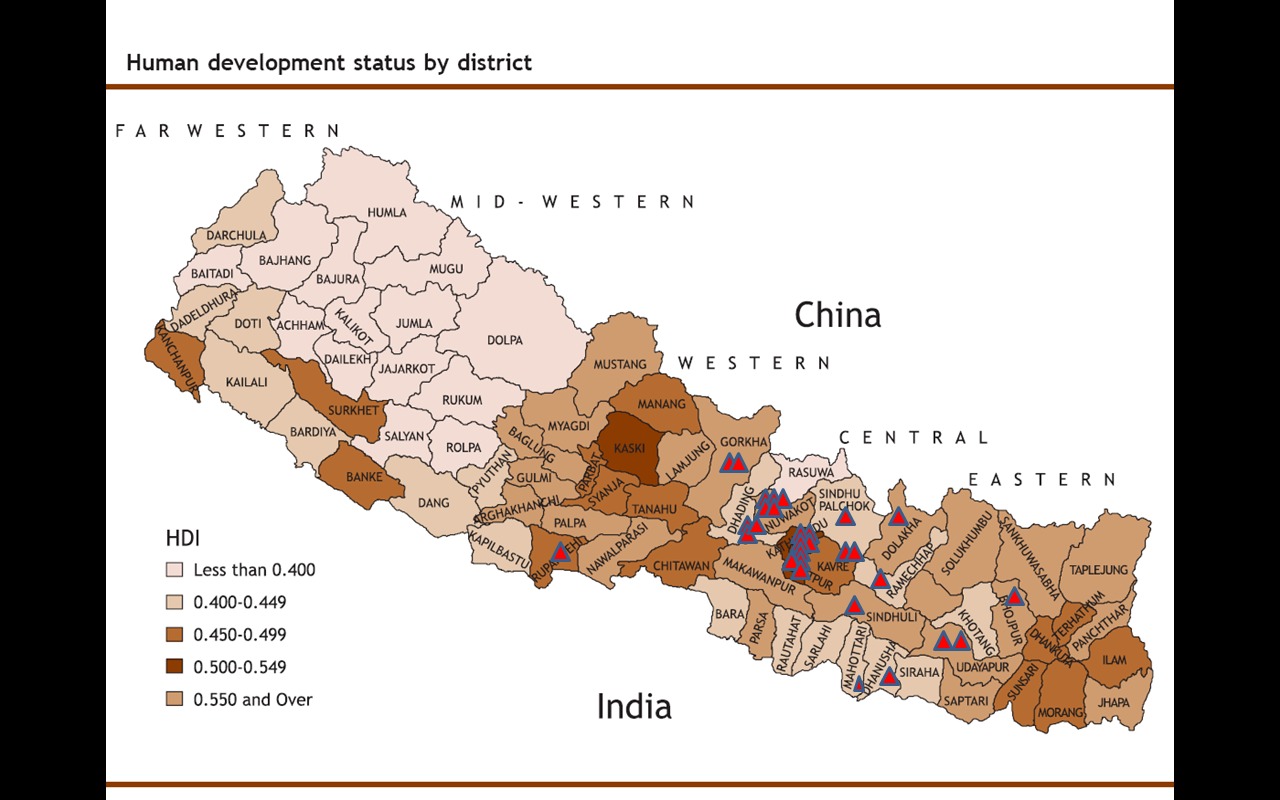
Six patients were residents of Kathmandu, whereas 24 patients came from other districts. (Figure 5)
The most common occupation was labouring and farming. (Table 6)
Table 6. Means of living
No. of patients (%)
Unemployed / Unknown
Fig 5. Geographic distribution of the patients
6. Case report
A majority of the patients with typhoid fever perforation present with a solitary perforation.
However, a reperforation of the ileum is seen in some cases and one study reported a reperforation
rate of 12 % [25]. It is important to consider the possibility that the recurrent peritonitis is truly a
persistent peritonitis due to missed perforations.
This case report demonstrates a case of re-perforation in a nepali women with typhoid fever.
A 35-year old nepali woman was admitted to the Tribhuvan University Teaching Hospital on April
27th 2010 feeling generally unwell, with a 12 day history of fever, abdominal pain, anorexia and
loose stool. She also suffered from nausea and vomiting. Her temperature was 39.0 C, blood
pressure 100/ 70 mmHg and pulse rate 108 bpm. Upon examination the patient was ill-looking but
oriented. Her abdomen was normal shaped but showed guarding, board-like rigidity and rebound
tenderness during palpation. Bowel sounds were absent on ascultation.
Analysis of blood showed Na+ 136.0 mEq/L, K+ 3.9 mEq, HbsAg reactive, creatinine 69.0 mmol/L,
WBC 4200 cmm with the differential count neutrophils 62 % and lymphocytes 33 %.
An abdominal ultrasound was carried out but proved inconclusive. Gynecologic disease was ruled
out by a consulting gynecologist.
Based on clinical findings and laboratory results, ileal perforation or perforated appendicitis was
suspected. After resucitation with intravenous fluids and administration of intravenous Xone and
Metron, an exploratory laparotomy was carried out. A solitary 1 x 0.5 cm perforation was localized
in the antimesenteric border of the distal ileum, 35 cm proximal to the ileo-caecal junction.
Multiple enlarged mesenteric lymphnodes were found in the distal ileum and two litres of dirty fluid
was present in the peritoneal cavity and pelvis . The peritoneal fluid was collected and the diseased
bowel segment was resected with wedge resection and a two – layer anastomosis. The enlarged
mesenteric lymphnodes were extracted.
The patients condition improved and was planned for discharge. However, upon stich removal on
May 10th 2010, profuse serosanguinous discharge from the wound was noticed and bowelloops
were visable. The patient was diagnosed with wound dehiscence and rushed to the operation theater
where an exploratory laparotomy was performed. A new perforation measuring 1 x 1 cm was found
on the antimesenteric border of the ileum, 25 cm from the ileo- caecal junction and 10 cm distal to
the previous anastomosis. Multiple interloop abscesses and adhesions as well as an abscess in the
subcutaneous plane were present. A peritoneal washing was performed and the interloop abscesses
and adhesions released. The perforation was repaired in a two – layer anastomosis and the previous
repair site strengthened with a seromuscular suture. The wound was then closed with a tension
suture. Pus was sent for culture and sensitivity test, showing Escheria Coli sensitive to Amikacin
only, whereafter treatment with intravenous Amikcain was initiated.
The postoperative recovery was uneventful and the patient was discharged with oral antibiotics on
May 17th 2010, 20 days after hospital admission.
In this study, a majority of the patients with ileal perforation were males. This finding in consistent
with previous reports where a high male : female ratio amongst patients with typhoid fever
perforation was documented [1] [6] . It does however remain unclear why typhoid fever perforation
is more commonly seen in males than in females. In a study by Santillana et al [25], immune
mechanisms and genetic predisposition factors were suggested as possible underlying factors. It has
been proposed that individuals with genetic governance of the immune response to the S. Typhi
antigen could undergo an exaggerated hyperplastic response of the Peyer's patches, leading to an
increased risk of perforation. The male predominance would then be explained by a predisposing
genetic pattern. Additional support for the genetic predisposition in the pathogenes of typhoid
perforation is given by a case report written by Vyas et al who reported typhoid fever perforation in
two identical twin brothers aged 21 years in India [26].
It is also possible that men spend more time outside of the home which increases the risk of
exposure to S. Typhi. genetic predisposition factors have
Typhoid fever perforation is generally seen in the second and third decade of young adolescents
[27, 28]. As previously suggested, multiple exposures to S. Typhi may be required for the activation
of the enhanced immune system producing ileal perforation. This theory is further supported by the
virtual elimination of typhoid fever perforation in developed countries where the disease is seen in
returning travellers, making multiple exposures to the bacteria almost impossible.
The requirement of multiple exposures would explain the increasing incidence of ileal perforation
parallell with age.
In this study, the number of patients in the age group 17 - 39 years constitute a majority of the total
number of patients. A possible explanation for the age-dependent incidence is that people in this age
group might be in touch with the environment more often and eat more food outside of the home,
thereby increasing the exposure to S. Typhi. The young and the elederly may spend more time at
home whereas people of working age naturally spend more time outside the home.
The finding of a peak incidence in perforations in the second week of disease agrees with reports
from the world literature [6] [25]. This suggests that time is required for the bacteria to invade the
tissue and also for the immune system to contribute to the pathogenesis of the intestinal lesions.
Clearly, patients seek health care late in the course of their illness. This delay might be explained
by several factors. Febrile disease is very common in Nepal, which makes it difficult to identify and
differentiate between harmful diseases. It is therefore likely that the disease is often initially
misdiagnosed as a cold and self-medicated with over the counter medicine or traditional treatment.
Moreover, a large part of the population has a poor access to health care with insufficient
infrastructure leading to transport difficulties. Many of the patients included in this study had
traveled two-three days to reach the TUTH.
Furthermore, the expenses of hospital treatment may impede typhoid patients from seeking
professional medical care or wait to the point where complications such as ileal perforation have
already occured. Finally, the people may be accustumed to social hardship and withstand the
disease symtoms for a longer period of time, thereby delaying the time to appropriate treatment.
Leukopenia is a common laboratory finding in patients with uncomplicated typhoid fever. A study
by Khan et al, in which five of 21 patients had intestinal perforation, the rate of leukopenia was
higher amongst patients with no complications [29]. Santillana et al reported leukocytosis in 59.5 %
of a total of 96 patients diagnosed with typhoid fever perforation [25].
In this study, 15 out of 29 patients showed leukocytosis, whereas only three patients showed
leukopenia. The likely explanation is that the ileal perforation leads to secondary peritonitis and
consequently, a rise in the white blood cell count.
Fever ( >38.0 °C) was not a striking admission finding. All patients presented with a history of
prolonged fever, but only 10 patients had an elevated body temperature upon examination. It is
likely that a majority of the patients had recieved antipyretic drugs before hospital admission.
The vast majority of the patients presented with a solitary perforation. However, when searching the
literature, multiple perforations are not infrequently reported [30, 31].
Out of the 30 patients enrolled in this study, two patients (7 %) deceased due to complications
following the ileal perforation. This number is lower than the typical mortality rates of 20- 40 %
that are most commonly reported [24] [31, 32, 33] . There is, however, a wide range in these
figures. Studies conducted in countries with better economic conditions, including South Korea [34]
and South Africa [35] have showed mortality rates as low as 9 %. These outcomes have been
achieved by the addition of close electrolyte and blood gas monitoring, intensive care nursing,
central venous pressure monitoring and the use of total parenteral nutrition.
A possible explantion for the low mortality in this study is the fact that most patients were referred
from other health centres. Furthermore, patients with a rapid and agressive progress of typhoid
fever may not reach the hospital in time and decease at home. These patients can therefore not be
included and consequently, the overall mortality in typhoid fever perforation may be higher than
indicated in this study.
Multiple studies have previously highlighted the seasonal variation of typhoid fever. Karkey et al
reported an incidence peak in Nepal during the monsoon months from June to August [4]. This is
an association that is well known amongst physicians in Nepal. However, in this study, the majority
of ileal perforations took place during February to May. This is the period of spring with limited
rainfall and not a typical time for the onset of typhoid fever.
One explanation for the high incidence of ileal perforation during this time period could be that
physicians misdiagnose the disease since it is not commonly seen in this time of year. Therefore, the
disease may remain undiagnosed and consequently untreated, increasing the risk for ileal
Early administration of efficient antimicrobial treatment is essential for the successful treatment of
typhoid fever. In this study, eight of 29 patients had recieved previous antibiotic treatment
prescribed by physicians prior to admission to the TUTH. Previous studies have suggested that an
adequate antibiotic treatment will prevent the development of ileal perforation in patients with
typhoid fever [6] [24].This is further illustrated by the fact that ileal perforation among patients
with typhoid fever has been extremely rare in developed countries during the era of antibiotic use
[24] [6]. However, for these eight patients, the chosen antimicrobial treatment could not prevent the
development of intestinal perforation. This may be due to he administration of an inappropriate
antibiotic, antibiotic resistance or to a delay in administration.
It is important to highlight the assocation between inadequate antibiotic treatment and intestinal
perforation, since this is a factor that is potentially modifiable.
Out of the 30 patients included in the study, six patients were residents of Kathmandu whereas 24
patients came from smaller villages on the countryside.
It appears, that a majority of the patients that received care at the TUTH were residents of areas
with a higher standard of living. There were no patients from the far west of Nepal, which is the
most economically deprived area of the country. Therefore, it is reasonable to assume, that these
patients are treated at regional hospitals which are not properly equipped. There were no patients
from the southern districts close to the indian border. One possible explanation is that these patients
may seek medical care at indian hospitals instead of coming to Kathmandu.
As previously stated, antibiotic resistance is becoming a major problem and treatment is becoming
increasingly difficult, leading to patients taking longer to recover, suffering more complications and
continuing to spread the disease to their family and to their community.
In Nepal, many cases of typhoid fever may be self-treated, with patients using unprescribed
antibiotics from medical shops. A wide range of medications are available prior to consultation with
a physician, which makes it difficult to know if the selected antimicrobial treatment is appropriate.
Furthermore, a majority of patients diagnosed with typhoid fever are treated as out-patients, which
makes it difficult to control the complaince to treatment.
In this study, most patients recieved treatment with intravenous Xone and Metronidazol and had a
good outcome after the surgical treatment. This indicates that the antibiotic treatment with third
generation cephalosporins was adequate and that resistance development did not pose a problem for
these patients.
As previously stated, a majority of the patients came from villages outside the city of Kathmandu,.
In these areas, the acess and exposition to antibiotics is limitied, especially when compared to the
situation in Kathmandu. It is likely that self-medication is more common in the city, whereas
traditional medicine has a bigger impact in the villages. This might be the explanation for the
overall successful antibiotic treatment in these patients. It is however important to control the
prescribing and over the counter sales of antibiotics and to monitor antibiotic susceptibility patterns
The two interventions for the control of typhoid fever are treatment of the already infected and
preventive measures. Ultimately, resources should be allocated towards prevention strategies that
reduce the transmission of the disease. Prevention is based on ensuring access to safe water and by
promoting safe food handling practices. The setting up and maintenance of the needed modern
water system is essential as well as collection and treatment of sewage, especially during the
monsoon season. Behaviour change with regard to hygiene is a cornerstone in the prevention of
typhoid fever. In order to raise public awarness of typhoid fever and the recommended prevention
measures, health education is important.
Many challenges remain for the control and management of typhoid fever in Nepal and the rest of
the developing world. Especially when taking the young age of patients with typhoid fever
perforation in consideration, the typical morbidity and mortality is unacceptably high. It also is
important to study typhoid fever from a broader context of society and acknowledge poverty as a
determinant for health.
Typhoid fever remains a major health problem in Nepal. The leading cause of mortality in typhoid
fever is ileal perforation, which is a complication that possibly could be prevented. Identification
and appropriate antibiotic treatment of typhoid fever would reduce morbidity and mortality. Early
and appropriate surgical intervention is vital for the survival of typhoid fever perforation.
A majority of the patients in this study were young males and this finding is consistent with
previous reports.
This study was supported by grants from the Minor Field Study Programme of the Swedish
International Development Agency (SIDA) and the University of Gothenburg.
I would like to adress warm and grateful thanks to all whom in different ways have contributed to
the development and realization of this project.
Firts of all I want to thank my supervisors Dr. Göran Kurlberg, Dr. Yogendra Singh and Dr. Bikhal
Ghimire for their input and assistance throughtout the planning, researching and writing of this
My family and my partner deserve special recognition for supporting me in all circumstances and
providing me with a foundation to feel comfortable in new environments.
Finally, I am very thankful to the Nepali people – for their.kindness and welcoming attitudes, and
not least for sharing their personal experiences of typhoid fever with me.
1. Gedik, E., et al., Risk factors affecting morbidity in typhoid enteric perforation. Langenbecks
Arch Surg, 2008. 393 (6): p. 973-7.
2. Crump, J.A. and E.D. Mintz, Global trends in typhoid and paratyphoid Fever. Clin Infect Dis,
2010. 50(2): p. 241-6.
3. Kothari, A., A. Pruthi, and T.D. Chugh, The burden of enteric fever. J Infect Dev Ctries, 2008.
2(4): p. 253-9.
4. Karkey, A., et al., Kathmandu, Nepal: still an enteric fever capital of the world. J Infect Dev
Ctries, 2008. 2(6): p. 461-5.
5. Raffatellu, M., et al., Clinical pathogenesis of typhoid fever. J Infect Dev Ctries, 2008. 2(4): p.
6. Hosoglu, S., et al., Risk factors for enteric perforation in patients with typhoid Fever. Am J
Epidemiol, 2004. 160(1): p. 46-50.
7. Zhang, X.L., V.T. Jeza, and Q. Pan, Salmonella typhi: from a human pathogen to a vaccine
vector. Cell Mol Immunol, 2008. 5(2): p. 91-7.
8. World Health Organization, Treatment of Typhoid Fever, Background document: the diagnosis,
treatment and prevention of typhoid fever. Department of Vaccines and Biologicals, 2003: p. 19-23.
9. Crump, J., S. Luby, and E. Mintz (2004) The global burden of typhoid fever. Bull World Health
Organ 82, 346–353.
10. Ochiai, R.L., et al., A study of typhoid fever in five Asian countries: disease burden and
implications for controls. Bull World Health Organ, 2008. 86(4): p. 260-8.
11. Maskey, A.P., et al., Emerging trends in enteric fever in Nepal: 9124 cases confirmed by blood
culture 1993-2003. Trans R Soc Trop Med Hyg, 2008. 102(1): p. 91-5.
12. Baker, S., M. Favorov, and G. Dougan, Searching for the elusive typhoid diagnostic. BMC
Infect Dis, 2010. 10: p. 45.
13. Bhutta, Z.A., Current concepts in the diagnosis and treatment of typhoid fever. BMJ, 2006.
333(7558): p. 78-82.
14. Crump, J.A., et al., Estimating the incidence of typhoid fever and other febrile illnesses in
developing countries. Emerg Infect Dis, 2003. 9(5): p. 539-44.
15. House, D., et al., Typhoid fever: pathogenesis and disease. Curr Opin Infect Dis, 2001. 14(5): p.
16. Raman, T.S., et al., Diazo test in typhoid fever. Indian Pediatr, 1994. 31(2): p. 201-4.
17. Khan, M., et al., The early diagnosis of typhoid fever prior to the Widal test and bacteriological
culture results. Acta Trop, 1998. 69(2): p. 165-73.
18. Pohan, H.T., Clinical and laboratory manifestations of typhoid fever at Persahabatan Hospital,
Jakarta. Acta Med Indones, 2004. 36(2): p. 78-83.
19. Akgun, Y., et al., Typhoid enteric perforation. Br J Surg, 1995. 82(11): p. 1512-5.
20. Mock, C.N., J. Amaral, and L.E. Visser, Improvement in survival from typhoid ileal perforation.
Results of 221 operative cases. Ann Surg, 1992. 215(3): p. 244-9.
21. Gibney, E.J., Typhoid perforation. Br J Surg, 1989. 76(9): p. 887-9.
22. Bank, T.W., "Nepal at a glance", Nepal: Publications & Reports.
ccessed 25.5.2010).
23. Organization, W.H., Nepal: Health Profile. (accessed
24. Butler, T., et al., Typhoid fever complicated by intestinal perforation: a persisting fatal disease
requiring surgical management. Rev Infect Dis, 1985. 7(2): p. 244-56.
25. Santillana, M., Surgical complications of typhoid fever: enteric perforation. World J Surg, 1991.
15(2): p. 170-5.
26. Vyas, I.D., M.G. Purohit, and H.L. Patel, Simultaneous typhoid ileal perforation in identical
twin brothers. Br J Clin Pract, 1980. 34(8-9): p. 256-7.
27. Sumer, A., et al., Outcome of surgical treatment of intestinal perforation in typhoid fever. World
J Gastroenterol, 2010. 16(33): p. 4164-8.
28. Shukla, V.K., et al., Enteric perforation--single-layer closure. Dig Dis Sci, 2004. 49(1): p. 161-
29. Khan, M., et al., Risk factors predicting complications in blood culture-proven typhoid fever in
adults. Scand J Infect Dis, 2000. 32(2): p. 201-5.
30. Tade, A.O., B.A. Ayoade, and A.A. Olawoye, Pattern of presentation and management of
typhoid intestinal perforation in Sagamu, South-West Nigeria: a 15 year study. Niger J Med, 2008.
17(4): p. 387-90.
31. Agbakwuru, E.A., et al., A review of typhoid perforation in a rural African hospital. West Afr J
Med, 2003. 22(1): p. 22-5.
32. Kayabali, I., I.H. Gokcora, and M. Kayabali, A contemporary evaluation of enteric perforations
in typhoid fever: analysis of 257 cases. Int Surg, 1990. 75(2): p. 96-100.
33. van Basten, J.P. and R. Stockenbrugger, Typhoid perforation. A review of the literature since
1960. Trop Geogr Med, 1994. 46(6): p. 336-9.
34. Kim, J.P., S.K. Oh, and F. Jarrett, Management of ileal perforation due to typhoid fever. Ann
Surg, 1975. 181(1): p. 88-91.
Keenan, J.P. and G.P. Hadley, The surgical management of typhoid perforation in children.
Br J Surg, 1984. 71(12): p. 928-9.
Kurlberg G. and B. Frisk, Factors reducing mortality in typhoid ileal perforation, Trans R
Soc Trop Med Hyg., 1991 Nov-Dec;85(6):793-5.
Source: http://www.goteborgsuniversitet.se/digitalAssets/1345/1345073_isabel-vigmo-2010.pdf
Magnus Hirsch, Markus von Fuchs and Margret Knitter SKW Schwarz Rechtsanwälte Trademarks A Global Guide SKW Schwarz:independent and business-minded The Law Firm Areas of Work SKW Schwarz is an independent German law firm. Banking and Finance We advise companies of all sizes ranging from owner-
Treatment Options for GERD or Acid Reflux Disease A Review of the Research for Adults Is This Information Right for Me? A doctor said that you have gastroesophageal reflux disease (GERD), a chronic disease that causes ongoing pain and burning in the chest and throat (heartburn). GERD is also called "acid You want to know what research says about your options.













