Microsoft word - carskadon dement 2011-fixed.docx
Carskadon, M.A., & Dement, W.C. (2011). Monitoring and staging human sleep. In M.H. Kryger, T.
Roth, & W.C. Dement (Eds.),
Principles and practice of sleep medicine, 5th edition, (pp 16-26). St.
Louis: Elsevier Saunders.
Chapter 2 – Normal Human Sleep : An Overview Mary A. Carskadon, William C. Dement Abstract
Normal human sleep comprises two states—rapid eye movement (REM) and non–REM (NREM) sleep—that alternate cyclically across a sleep episode. State characteristics are well defined: NREM sleep includes a variably synchronous cortical electroencephalogram (EEG; including sleep spindles, K-complexes, and slow waves) associated with low muscle tonus and minimal psychological activity; the REM sleep EEG is desynchronized, muscles are atonic, and dreaming is typical. A nightly pattern of sleep in mature humans sleeping on a regular schedule includes several reliable characteristics: Sleep begins in NREM and progresses through deeper NREM stages (stages 2, 3, and 4 using the classic definitions, or stages N2 and N3 using the updated definitions) before the first episode of REM sleep occurs approximately 80 to 100 minutes later. Thereafter, NREM sleep and REM sleep cycle with a period of approximately 90 minutes. NREM stages 3 and 4 (or stage N3) concentrate in the early NREM cycles, and REM sleep episodes lengthen across the night.
Age-related changes are also predictable: Newborn humans enter REM sleep (called active sleep
) before NREM (called quiet sleep
) and have a shorter sleep cycle (approximately 50 minutes); coherent sleep stages emerge as the brain matures during the first year. At birth, active sleep is approximately 50% of total sleep and declines over the first 2 years to approximately 20% to 25%. NREM sleep slow waves are not present at birth but emerge in the first 2 years. Slow-wave sleep (stages 3 and 4) decreases across adolescence by 40% from preteen years and continues a slower decline into old age, particularly in men and less so in women. REM sleep as a percentage of total sleep is approximately 20% to 25% across childhood, adolescence, adulthood, and into old age except in dementia. Other factors predictably alter sleep, such as previous sleep-wake history (e.g., homeostatic load), phase of the circadian timing system, ambient temperature, drugs, and sleep disorders.
A clear appreciation of the normal characteristics of sleep provides a strong background and template for understanding clinical conditions in which "normal" characteristics are altered, as well as for interpreting certain consequences of sleep disorders. In this chapter, the normal young adult sleep pattern is described as a working baseline pattern. Normative changes due to aging and other factors are described with that background in mind. Several major sleep disorders are highlighted by their differences from the normative pattern.
Sleep Definitions
According to a simple behavioral definition, sleep is a reversible behavioral state of perceptual disengagement from and unresponsiveness to the environment. It is also true that sleep is a complex amalgam of physiologic and behavioral processes. Sleep is typically (but not necessarily) accompanied by postural recumbence, behavioral quiescence, closed eyes, and all the other indicators one commonly associates with sleeping. In the unusual circumstance, other behaviors can occur during sleep. These behaviors can include sleepwalking, sleeptalking, teeth grinding, and other physical activities. Anomalies involving sleep processes also include intrusions of sleep—sleep itself, dream imagery, or muscle weakness—into wakefulness, for example (Box 2-1).
Sleep Medicine Methodology and Nomenclature
In 2007, the American Academy of Sleep Medicine (AASM) published a new manual (see reference 50) for scoring sleep and associated events. This manual recommends alterations to recording methodology and terminology that the Academy will demand of clinical laboratories in the future. Although specification of arousal, cardiac, movement, and respiratory rules appear to be value added to the assessment of sleep-related events, the new rules, terminology, and technical specifications for recording and scoring sleep are not without controversy.
The current chapter uses the traditional terminology and definitions, upon which most descriptive and experimental research has been based since the 1960s.[17] Hence, where the AASM terminology uses the term N for NREM sleep stages and R for REM sleep stages, N1 and N2 are used instead of stage 1 and stage 2; N3 is used to indicate the sum of stage 3 and stage 4 (often called slow-wave sleep in human literature); R is used to name REM sleep. Another change is to the nomenclature for the recording placements. Hence, calling the auricular placements M1 and M2 (rather than A1 and A2) is unnecessary and places the sleep EEG recording terminology outside the pale for EEG recording terminology in other disciplines. Although these are somewhat trivial changes, changes in nomenclature can result in confusion when attempting to compare to previous literature and established data sets and are of concern for clinicians and investigators who communicate with other fields.
Of greater concern are changes to the core recording and scoring recommendations that the AASM manual recommends. For example, the recommended scoring montage requires using a frontal (F3 or F4) EEG placement for use with visual scoring of the recordings, rather than the central (C3 or C4) EEG placements recommended in the standard manual. The rationale for the change is that the frontal placements pick up more slow-wave activity during sleep. The consequence, however, is that sleep studies performed and scored with the frontal EEG cannot be compared to normative or clinical data and the frontal placements also truncate the ability to visualize sleep spindles. Furthermore, developmental changes to the regional EEG preclude the universal assumption that sleep slow-wave activity is a frontal event.
Other issues are present in this new AASM approach to human sleep; however, this is not the venue for a complete description of such concerns. In summary, the AASM scoring manual has not yet become the universal standard for assessing human sleep and might not achieve that status in its current form. Specifications for recording and scoring sleep are not without controversy.[51-56]
Within sleep, two separate states have been defined on the basis of a constellation of physiologic parameters. These two states, rapid eye movement (REM) and non-REM (NREM), exist in virtually all mammals and birds yet studied, and they are as distinct from one another as each is from wakefulness.
NREM (pronounced "non-REM") sleep is conventionally subdivided into four stages defined along one measurement axis, the electroencephalogram (EEG). The EEG pattern in NREM sleep is commonly described as synchronous, with such characteristic waveforms as sleep spindles, K-complexes, and high-voltage slow waves (Fig. 2-1). The four NREM stages (stages 1, 2, 3, and 4) roughly parallel a depth-of-sleep continuum, with arousal thresholds generally lowest in stage 1 and highest in stage 4 sleep. NREM sleep is usually associated with minimal or fragmentary mental activity. A shorthand definition of NREM sleep is a relatively inactive yet actively regulating brain in a movable body.
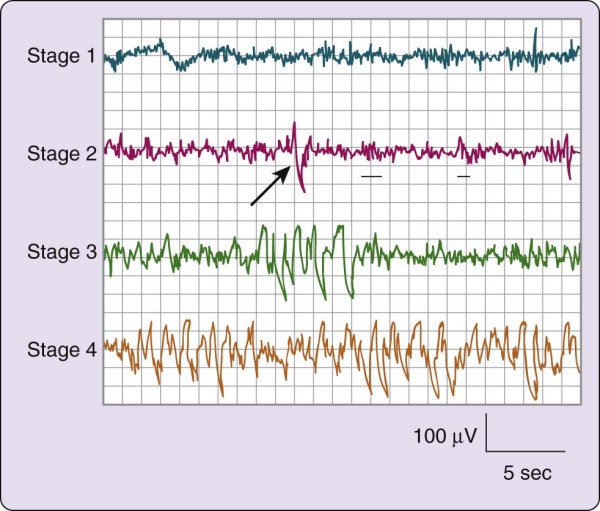
Figure 2-1 The stages of non–rapid eye movement sleep. The four electroencephalogram tracings depicted
here are from a 19-year-old female volunteer. Each tracing was recorded from a referential lead (C3/A2) recorded on a Grass Instruments Co. (West Warwick, R.I.) Model 7D polygraph with a paper speed of 10 mm/sec, time constant of 0.3 sec, and 1/2 -amplitude high-frequency setting of 30 Hz. On the second tracing, the arrow indicates a K-complex and the underlining shows two sleep spindles.
REM sleep, by contrast, is defined by EEG activation, muscle atonia, and episodic bursts of rapid eye movements. REM sleep usually is not divided into stages, although tonic and phasic types of REM sleep are occasionally distinguished for certain research purposes. The distinction of tonic versus phasic is based on short-lived events such as eye movements that tend to occur in clusters separated by episodes of relative quiescence. In cats, REM sleep phasic activity is epitomized by bursts of ponto-geniculo-occipital (PGO) waves, which are accompanied peripherally by rapid eye movements, twitching of distal muscles, middle ear muscle activity, and other phasic events that correspond to the phasic event markers easily measurable in human beings. As described in Chapter 141, PGO waves are not usually detectable in human beings. Thus, the most commonly used marker of REM sleep phasic activity in human beings is, of course, the bursts of rapid eye movements (Fig. 2-2); muscle twitches and cardiorespiratory irregularities often accompany the REM bursts. The mental activity of human REM sleep is associated with dreaming, based on vivid dream recall reported after approximately 80% of arousals from this state
of sleep.[1] Inhibition of spinal motor neurons by brainstem mechanisms mediates suppression of postural motor tonus in REM sleep. A shorthand definition of REM sleep, therefore, is an activated brain in a paralyzed body.
Figure 2-2 Phasic events in human rapid eye movement (REM) sleep. On the left side is a burst of several
rapid eye movements (out-of-phase deflections in right outer canthus [ROC]/A1 and left outer canthus [LOC]/A2).
On the right side, there are additional rapid eye movements as well as twitches on the electromyographic (EMG)
lead. The interval between eye movement bursts and twitches illustrates tonic REM sleep.
Sleep Onset
The onset of sleep under normal circumstances in normal adult humans is through NREM sleep. This fundamental principle of normal human sleep reflects a highly reliable finding and is important in considering normal versus pathologic sleep. For example, the abnormal entry into sleep through REM sleep can be a diagnostic sign in adult patients with narcolepsy.
Definition of Sleep Onset
The precise definition of the onset of sleep has been a topic of debate, primarily because there is no single measure that is 100% clear-cut 100% of the time. For example, a change in EEG pattern is not always associated with a person's perception of sleep, yet even when subjects report that they are still awake, clear behavioral changes can indicate the presence of sleep. To begin a consideration of this issue, let us examine the three basic polysomnographic measures of sleep and how they change with sleep onset. The electrode placements are described in Chapter 141.
The electromyogram (EMG) may show a gradual diminution of muscle tonus as sleep approaches, but rarely does a discrete EMG change pinpoint sleep onset. Furthermore, the presleep level of the EMG, particularly if the person is relaxed, can be entirely indistinguishable from that of unequivocal sleep (Fig. 2-3).
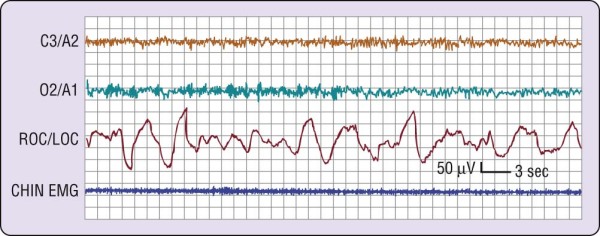
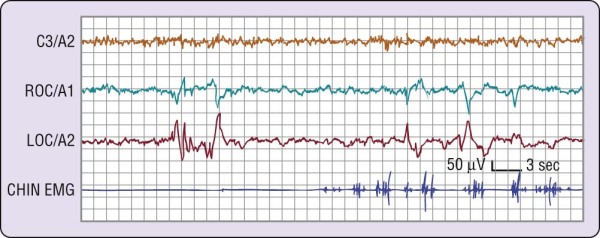
Figure 2-3 The transition from wakefulness to stage 1 sleep. The most marked change is visible on the two
electroencephalographic (EEG) channels (C3/A2 and O2/A1), where a clear pattern of rhythmic alpha activity (8 cps) changes to a relatively low-voltage, mixed- frequency pattern at about the middle of the figure. The level of electromyographic (EMG) activity does not change markedly. Slow eye movements (right outer canthus [ROC]/left outer canthus [LOC]) are present throughout this episode, preceding the EEG change by at least 20 seconds. In general, the change in EEG patterns to stage 1 as illustrated here is accepted as the onset of sleep.
Electrooculogram
As sleep approaches, the electrooculogram (EOG) shows slow, possibly asynchronous eye movements (see Fig. 2-3) that usually disappear within several minutes of the EEG changes described next. Occasionally, the onset of these slow eye movements coincides with a person's perceived sleep onset; more often, subjects report that they are still awake.
Electroencephalogram
In the simplest circumstance (see Fig. 2-3), the EEG changes from a pattern of clear rhythmic alpha (8 to 13 cycles per second [cps]) activity, particularly in the occipital region, to a relatively low-voltage, mixed-frequency pattern (stage 1 sleep). This EEG change usually occurs seconds to minutes after the start of slow eye movements. With regard to introspection, the onset of a stage 1 EEG pattern may or may not coincide with perceived sleep onset. For this reason, a number of investigators require the presence of specific EEG patterns—the K-complex or sleep spindle (i.e., stage 2 sleep)—to acknowledge sleep onset. Even these stage 2 EEG patterns, however, are not unequivocally associated with perceived sleep.[2] A further complication is that sleep onset often does not occur all at once; instead, there may be a wavering of vigilance before "unequivocal" sleep ensues (Fig. 2-4). Thus, it is difficult to accept a single variable as marking sleep onset. As Davis and colleagues[3] wrote many years ago (p. 35):
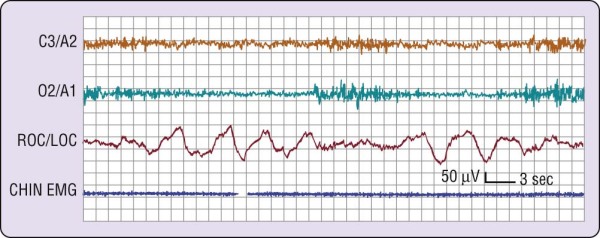
Figure 2-4 A common wake- to-sleep transition pattern. Note that the electroencephalographic pattern
changes from wake (rhythmic alpha) to stage 1 (relatively low-voltage, mixed-frequency) sleep twice during this attempt to fall asleep. EMG, electromyogram; LOC, left outer canthus; ROC, right outer canthus.
Is "falling asleep" a unitary event? Our observations suggest that it is not. Different functions, such as sensory awareness, memory, self-consciousness, continuity of logical thought, latency of response to a stimulus, and alterations in the pattern of brain potentials all go in parallel in a general way, but there are exceptions to every rule. Nevertheless, a reasonable consensus exists that the EEG change to stage 1, usually heralded or accompanied by slow eye movements, identifies the transition to sleep, provided that another EEG sleep pattern does not intervene. One might not always be able to pinpoint this transition to the millisecond, but it is usually possible to determine the change reliably within several seconds.
Behavioral Concomitants of Sleep Onset
Given the changes in the EEG that accompany the onset of sleep, what are the behavioral correlates of the wake-to-sleep transition? The following material reviews a few common behavioral concomitants of sleep onset. Keep in mind that "different functions may be depressed in different sequence and to different degrees in different subjects and on different occasions" (p. 35).[3]
Simple Behavioral Task
In the first example, volunteers were asked to tap two switches alternately at a steady pace. As shown in Figure 2-5, this simple behavior continues after the onset of slow eye movements and may persist for several seconds after the EEG changes to a stage 1 sleep pattern.[4] The behavior then ceases, usually to recur only after the EEG reverts to a waking pattern. This is an example of what one may think of as the simplest kind of "automatic" behavior pattern. Because such simple behavior can persist past sleep onset and as one passes in and out of sleep, it might explain how impaired, drowsy drivers are able to continue down the highway.
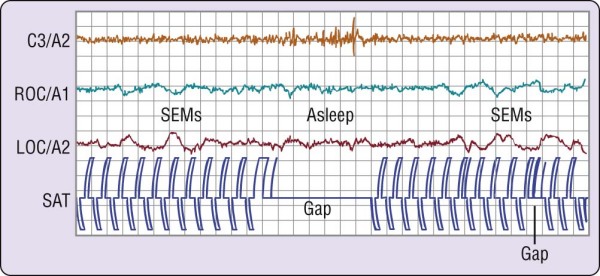
Figure 2-5 Failure to perform a simple behavioral task at the onset of sleep. The volunteer had been deprived
of sleep overnight and was required to tap two switches alternately, shown as pen deflections of opposite polarity on the channel labeled SAT. When the electroencephalographic (EEG; C3/A2) pattern changes to stage 1 sleep, the behavior stops, returning when the EEG pattern reverts to wakefulness. LOC, left outer canthus; ROC, right outer canthus; SEMs, slow eye movements. (From Carskadon MA, Dement WC. Effects of total sleep loss on sleep tendency. Percept Mot Skills 1979;48:495-506.
Visual Response
A second example of behavioral change at sleep onset derives from an experiment in which a bright light is placed in front of the subject's eyes, and the subject is asked to respond when a light flash is seen by pressing a sensitive microswitch taped to the hand.[5] When the EEG pattern is stage 1 or stage 2 sleep, the response is absent more than 85% of the time. When volunteers are queried afterward, they report that they did not see the light flash, not that they saw the flash but the response was inhibited. This is one example of the perceptual disengagement from the environment that accompanies sleep onset.
Auditory Response
In another sensory domain, the response to sleep onset is examined with a series of tones played over earphones to a subject who is instructed to respond each time a tone is heard. One study of this phenomenon showed that reaction times became longer in proximity to the onset of stage 1 sleep, and responses were absent coincident with a change in EEG to unequivocal sleep.[6] For responses in both visual and auditory modalities, the return of the response after its sleep-related disappearance typically requires the resumption of a waking EEG pattern.
Olfactory Response
When sleeping humans are tasked to respond when they smell something, the response depends in part on sleep state and in part on the particular odorant. In contrast to visual responses, one study showed that responses to graded strengths of peppermint (strong trigeminal stimulant usually perceived as pleasant) and pyridine (strong trigeminal stimulant usually perceived as extremely unpleasant) were well maintained during initial stage 1 sleep.[7] As with other modalities, the response in other sleep stages was significantly poorer: Peppermint simply was not consciously smelled in stages 2 and 4 NREM sleep or in
REM sleep; pyridine was never smelled in stage 4 sleep, and only occasionally in stage 2 NREM and in REM sleep.[7] On the other hand, a tone successfully aroused the young adult participants in every stage. One conclusion of this report was that the olfactory system of humans is not a good sentinel system during sleep.
Response to Meaningful Stimuli
One should not infer from the preceding studies that the mind becomes an impenetrable barrier to sensory input at the onset of sleep. Indeed, one of the earliest modern studies of arousability during sleep showed that sleeping human beings were differentially responsive to auditory stimuli of graded intensity.[8] Another way of illustrating sensory sensitivity is shown in experiments that have assessed discriminant responses during sleep to meaningful versus nonmeaningful stimuli, with meaning supplied in a number of ways and response usually measured as evoked K-complexes or arousal. The following are examples.
• A person tends to have a lower arousal threshold for his or her own name versus someone else's
name.[9] In light sleep, for example, one's own name spoken softly will produce an arousal; a
similarly applied nonmeaningful stimulus will not. Similarly, a sleeping mother is more likely to hear her own baby's cry than the cry of an unrelated infant.
• Williams and colleagues[10] showed that the likelihood of an appropriate response during sleep was
improved when an otherwise nonmeaningful stimulus was made meaningful by linking the absence of response to punishment (a loud siren, flashing light, and the threat of an electric shock).
From these examples and others, it seems clear that sensory processing at some level does continue after the onset of sleep. Indeed, one study has shown with functional magnetic resonance imaging that regional brain activation occurs in response to stimuli during sleep and that different brain regions (middle temporal gyrus and bilateral orbitofrontal cortex) are activated in response to meaningful (person's own name) versus nonmeaningful (beep) stimuli.[11]
Hypnic Myoclonia
What other behaviors accompany the onset of sleep? If you awaken and query someone shortly after the stage 1 sleep EEG pattern appears, the person usually reports the mental experience as one of losing a direct train of thought and of experiencing vague and fragmentary imagery, usually visual.[12] Another fairly common sleep-onset experience is hypnic myoclonia, which is experienced as a general or localized muscle contraction very often associated with rather vivid visual imagery. Hypnic myoclonias are not pathologic events, although they tend to occur more commonly in association with stress or with unusual or irregular sleep schedules.
The precise nature of hypnic myoclonias is not clearly understood. According to one hypothesis, the onset of sleep in these instances is marked by a dissociation of REM sleep components, wherein a breakthrough of the imagery component of REM sleep (hypnagogic hallucination) occurs in the absence of the REM motor inhibitory component. A response by the individual to the image, therefore, results in a movement or jerk. The increased frequency of these events in association with irregular sleep schedules is consistent with the increased probability of REM sleep occurring at the wake-to-sleep transition under such conditions (see later). Although the usual transition in adult human beings is to NREM sleep, the REM portal into sleep, which is the norm in infancy, can become partially opened under unusual circumstances.
Memory Near Sleep Onset
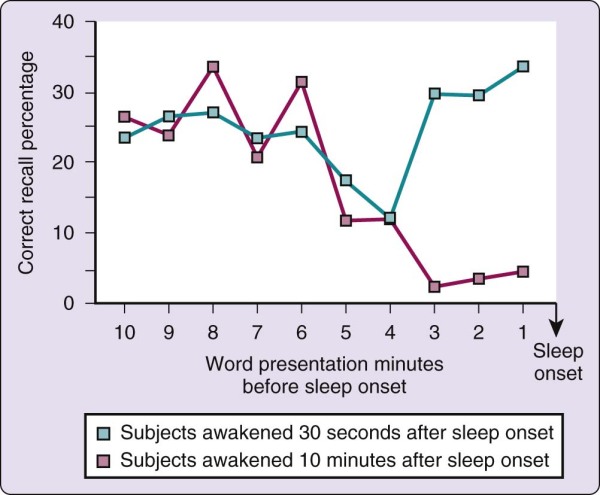
What happens to memory at the onset of sleep? The transition from wake to sleep tends to produce a memory impairment. One view is that it is as if sleep closes the gate between short-term and long-term memory stores. This phenomenon is best described by the following experiment.[13] During a presleep testing session, word pairs were presented to volunteers over a loudspeaker at 1-minute intervals. The subjects were then awakened either 30 seconds or 10 minutes after the onset of sleep (defined as EEG stage 1) and asked to recall the words presented before sleep onset. As illustrated in Figure 2-6, the 30-second condition was associated with a consistent level of recall from the entire 10 minutes before sleep onset. (Primacy and recency effects are apparent, although not large.) In the 10-minute condition, however, recall paralleled that in the 30-second group for only the 10 to 4 minutes before sleep onset and then fell abruptly from that point until sleep onset.
Figure 2-6 Memory is impaired by sleep, as shown by the study results illustrated in this graph. See text for
In the 30-second condition, therefore, both longer-term (4 to 10 minutes) and shorter-term (0 to 3 minutes) memory stores remained accessible. In the 10-minute condition, by contrast, words that were in
longer-term stores (4 to 10 minutes) before sleep onset were accessible, whereas words that were still in shorter-term stores (0 to 3 minutes) at sleep onset were no longer accessible; that is, they had not been consolidated into longer-term memory stores. One conclusion of this experiment is that sleep inactivates the transfer of storage from short- to long-term memory. Another interpretation is that encoding of the material before sleep onset is of insufficient strength to allow recall. The precise moment at which this deficit occurs is not known and may be a continuing process, perhaps reflecting anterograde amnesia. Nevertheless, one may infer that if sleep persists for approximately 10 minutes, memory is lost for the few minutes before sleep. The following experiences represent a few familiar examples of this phenomenon:
• Inability to grasp the instant of sleep onset in your memory.
• Forgetting a telephone call that had come in the middle of the night.
• Forgetting the news you were told when awakened in the night.
• Not remembering the ringing of your alarm clock.
• Experiencing morning amnesia for coherent sleeptalking.
• Having fleeting dream recall.
Patients with syndromes of excessive sleepiness can experience similar memory problems in the daytime if sleep becomes intrusive.
Learning and Sleep
In contrast to this immediate sleep-related "forgetting," the relevance for sleep to human learning—particularly for consolidation of perceptual and motor learning—is of growing interest.[14],[15] The importance of this association has also generated some debate and skepticism.[16] Nevertheless, a spate of recent research is awakening renewed interest in the topic, and mechanistic studies explaining the roles of REM and NREM sleep more precisely are under examination (see Chapter 29).
Progression of Sleep Across the Night
Pattern of Sleep in a Normal Young Adult
The simplest description of sleep begins with the ideal case, the normal young adult who is sleeping well and on a fixed schedule of about 8 hours per night (Fig. 2-7). In general, no consistent male versus female distinctions have been found in the normal pattern of sleep in young adults. In briefest summary, the normal human adult enters sleep through NREM sleep, REM sleep does not occur until 80 minutes or longer thereafter, and NREM sleep and REM sleep alternate through the night, with an approximately 90-minute cycle (see Chapter 141 for a full description of sleep stages).
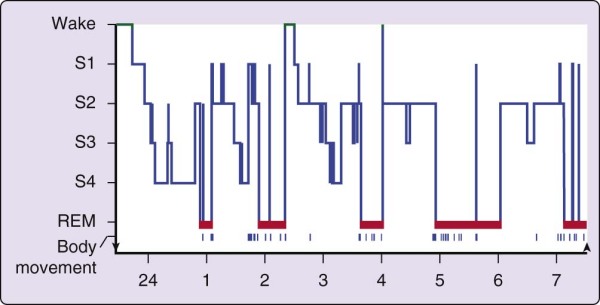
Figure 2-7 The progression of sleep stages across a single night in a normal young adult volunteer is
illustrated in this sleep histogram. The text describes the ideal or average pattern. This histogram was drawn on the basis of a continuous overnight recording of electroencephalogram, electrooculogram, and electromyogram in a normal 19-year-old man. The record was assessed in 30-second epochs for the various sleep stages. REM, rapid eye movement.
First Sleep Cycle
The first cycle of sleep in the normal young adult begins with stage 1 sleep, which usually persists for only a few (1 to 7) minutes at the onset of sleep. Sleep is easily discontinued during stage 1 by, for example, softly calling a person's name, touching the person lightly, quietly closing a door, and so forth. Thus, stage 1 sleep is associated with a low arousal threshold. In addition to its role in the initial wake-to-sleep transition, stage 1 sleep occurs as a transitional stage throughout the night. A common sign of severely disrupted sleep is an increase in the amount and percentage of stage 1 sleep.
Stage 2 NREM sleep, signaled by sleep spindles or K-complexes in the EEG (see Fig. 2-1), follows this brief episode of stage 1 sleep and continues for approximately 10 to 25 minutes. In stage 2 sleep, a more intense stimulus is required to produce arousal. The same stimulus that produced arousal from stage 1 sleep often results in an evoked K-complex but no awakening in stage 2 sleep.
As stage 2 sleep progresses, high-voltage slow-wave activity gradually appears in the EEG. Eventually, this activity meets the criteria[17] for stage 3 NREM sleep, that is, high-voltage (at least 75 µV) slow-wave (2 cps) activity accounting for more than 20% but less than 50% of the EEG activity. Stage 3 sleep usually lasts only a few minutes in the first cycle and is transitional to stage 4 as more and more high-voltage slow-wave activity occurs. Stage 4 NREM sleep—identified when the high-voltage slow-wave activity comprises more than 50% of the record—usually lasts approximately 20 to 40 minutes in the first cycle. An incrementally larger stimulus is usually required to produce an arousal from stage 3 or 4 sleep than from stage 1 or 2 sleep. (Investigators often refer to the combined stages 3 and 4 sleep as slow-wave sleep [SWS], delta sleep, or deep sleep.)
A series of body movements usually signals an "ascent" to lighter NREM sleep stages. A brief (1- or 2-minute) episode of stage 3 sleep might occur, followed by perhaps 5 to 10 minutes of stage 2 sleep
interrupted by body movements preceding the initial REM episode. REM sleep in the first cycle of the night is usually short-lived (1 to 5 minutes). The arousal threshold in this REM episode is variable, as is true for REM sleep throughout the night. Theories to explain the variable arousal threshold of REM sleep have suggested that at times, the person's selective attention to internal stimuli precludes a response or that the arousal stimulus is incorporated into the ongoing dream story rather than producing an awakening. Certain early experiments examining arousal thresholds in cats found highest thresholds in REM sleep, which was then termed deep sleep in this species. Although this terminology is still often used in publications about sleep in animals, it should not be confused with human NREM stages 3 plus 4 sleep, which is also often called deep sleep. One should also note that SWS is sometimes used (as is synchronized sleep) as a synonym for all of NREM sleep in other species and is thus distinct from SWS (stages 3 plus 4 NREM) in human beings.
NREM sleep and REM sleep continue to alternate through the night in cyclic fashion. REM sleep episodes usually become longer across the night. Stages 3 and 4 sleep occupy less time in the second cycle and might disappear altogether from later cycles, as stage 2 sleep expands to occupy the NREM portion of the cycle. The average length of the first NREM-REM sleep cycle is approximately 70 to 100 minutes; the average length of the second and later cycles is approximately 90 to 120 minutes. Across the night, the average period of the NREM-REM cycle is approximately 90 to 110 minutes.
Distribution of Sleep Stages across the Night
In the young adult, SWS dominates the NREM portion of the sleep cycle toward the beginning of the night (the first one third); REM sleep episodes are longest in the last one third of the night. Brief episodes of wakefulness tend to intrude later in the night, usually near REM sleep transitions, and they usually do not last long enough to be remembered in the morning. The preferential distribution of REM sleep toward the latter portion of the night in normal human adults is thought to be linked to a circadian oscillator, which can be gauged by the oscillation of body temperature.[18],[19] The preferential distribution of SWS toward the beginning of a sleep episode is not thought to be mediated by circadian processes but shows a marked response to the length of prior wakefulness.[20] The SWS pattern reflects the homeostatic sleep system, highest at sleep onset and diminishing across the night as sleep pressure wanes. Thus, these aspects of the normal sleep pattern highlight features of the two-process model of sleep as elaborated on in Chapter 37.
Length of Sleep
The length of nocturnal sleep depends on a great number of factors—of which volitional control is among the most significant in human beings—and it is thus difficult to characterize a "normal" pattern. Most young adults report sleeping approximately 7.5 hours a night on weekday nights and slightly longer, 8.5 hours, on weekend nights. The variability of these figures from person to person and from night to night, however, is quite high. Sleep length also depends on genetic determinants,[21] and one may think of the volitional determinants (staying up late, waking by alarm, and so on) superimposed on the background of a genetic sleep need. Length of prior waking also affects how much sleeps, although not in a one-for-one manner. Indeed, the length of sleep is also determined by processes associated with circadian rhythms. Thus, when one sleeps helps to determine how long one sleeps. In addition, as sleep is extended, the amount of REM sleep increases, because REM sleep depends on the persistence of sleep into the peak circadian time in order to occur.
Generalizations about Sleep in the Normal Young Adult
A number of general statements can be made regarding sleep in the normal young adult who is living on a conventional sleep-wake schedule and who is without sleep complaints:
• Sleep is entered through NREM sleep.
• NREM sleep and REM sleep alternate with a period near 90 minutes.
• SWS predominates in the first third of the night and is linked to the initiation of sleep and the length
• REM sleep predominates in the last third of the night and is linked to the circadian rhythm of body
• Wakefulness in sleep usually accounts for less than 5% of the night.
• Stage 1 sleep generally constitutes approximately 2% to 5% of sleep.
• Stage 2 sleep generally constitutes approximately 45% to 55% of sleep.
• Stage 3 sleep generally constitutes approximately 3% to 8% of sleep.
• Stage 4 sleep generally constitutes approximately 10% to 15% of sleep.
• NREM sleep, therefore, is usually 75% to 80% of sleep.
• REM sleep is usually 20% to 25% of sleep, occurring in four to six discrete episodes.
Factors Modifying Sleep Stage Distribution
Age
The strongest and most consistent factor affecting the pattern of sleep stages across the night is age (Fig. 2-8). The most marked age-related differences in sleep from the patterns described earlier are found in newborn infants. For the first year of life, the transition from wake to sleep is often accomplished through REM sleep (called active sleep in newborns). The cyclic alternation of NREM-REM sleep is present from birth but has a period of approximately 50 to 60 minutes in the newborn compared with approximately 90 minutes in the adult. Infants also only gradually acquire a consolidated nocturnal sleep cycle, and the fully developed EEG patterns of the NREM sleep stages are not present at birth but emerge over the first 2 to 6 months of life. When brain structure and function achieve a level that can support high-voltage slow-wave EEG activity, NREM stages 3 and 4 sleep become prominent.
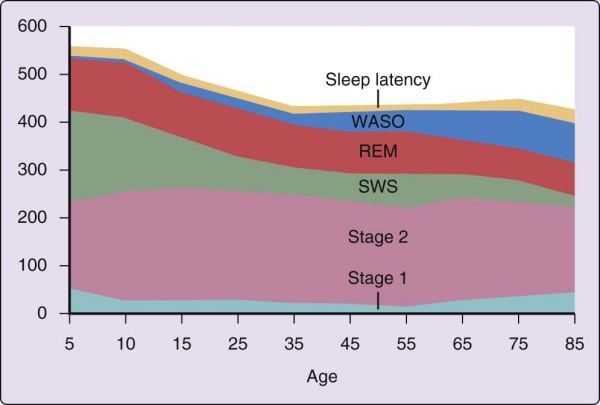
Figure 2-8 Changes in sleep with age. Time (in minutes) for sleep latency and wake time after sleep onset
(WASO) and for rapid eye movement (REM) sleep and non-REM (NREM) sleep stages 1, 2, and slow wave sleep (SWS). Summary values are given for ages 5 to 85 years. (Ohayon M, Carskadon MA, Guilleminault C, et al. Meta-analysis of quantitative sleep parameters from childhood to old age in healthy individuals: developing normative sleep values across the human lifespan. Sleep 2004;27:1255-1273.)
SWS is maximal in young children and decreases markedly with age. The SWS of young children is both qualitatively and quantitatively different from that of older adults. For example, it is nearly impossible to wake youngsters in the SWS of the night's first sleep cycle. In one study,[22] a 123-dB tone failed to produce any sign of arousal in a group of children whose mean age was 10 years. In addition, children up to midadolescence often "skip" their first REM episode, perhaps due to the quantity and intensity of slow-wave activity early in the night. A similar, although less profound qualitative difference distinguishes SWS occurring in the first and later cycles of the night in a given person. The quantitative change in SWS may best be seen across adolescence, when SWS decreases by nearly 40% during the second decade, even when length of nocturnal sleep remains constant.[23] Feinberg[24] hypothesized that the age-related decline in nocturnal SWS might parallel loss of cortical synaptic density. By midadolescence, youngsters no longer typically skip their first REM, and their sleep resembles that described earlier for young adults. By age 60 years, SWS might no longer be present, particularly in men. Women appear to maintain SWS later into life than men.
REM sleep as a percentage of total sleep is maintained well into healthy old age; the absolute amount of REM sleep at night has been correlated with intellectual functioning[25] and declines markedly in the case of organic brain dysfunctions of the elderly.[26]
Arousals during sleep increase markedly with age. Extended wake episodes of which the individual is aware and can report, as well as brief and probably unremembered arousals, increase with aging.[27] The latter type of transient arousals may occur with no known correlate but are often associated with occult sleep disturbances, such as periodic limb movements during sleep (PLMS) and sleep-related respiratory irregularities, which also become more prevalent in later life.[28],[29]
Perhaps the most notable finding regarding sleep in the elderly is the profound increase in interindividual variability,[30] which thus precludes generalizations such as those made for young adults.
Prior Sleep History
A person who has experienced sleep loss on one or more nights shows a sleep pattern that favors SWS during recovery (Fig. 2-9). Recovery sleep is also usually prolonged and deeper—that is, having a higher arousal threshold throughout—than basal sleep. REM sleep tends to show a rebound on the second or subsequent recovery nights after an episode of sleep loss. Therefore, with total sleep loss, SWS tends to be preferentially recovered compared with REM sleep, which tends to recover only after the recuperation of SWS.
Figure 2-9 The upper histogram shows the baseline sleep pattern of a normal 14-year-old female volunteer.
The lower histogram illustrates the sleep pattern in this volunteer for the first recovery night after 38 hours without sleep. Note that the amount of stage 4 sleep on the lower graph is greater than on baseline, and the first rapid eye movement (REM) sleep episode is markedly delayed.
Cases in which a person is differentially deprived of REM or SWS—either operationally, by being awakened each time the sleep pattern occurs, or pharmacologically (see later)—show a preferential rebound of that stage of sleep when natural sleep is resumed. This phenomenon has particular relevance in a clinical setting, in which abrupt withdrawal from a therapeutic regimen can result in misleading diagnostic findings (e.g., sleep-onset REM periods [SOREMPs] as a result of a REM sleep rebound) or could conceivably exacerbate a sleep disorder (e.g., if sleep apneas tend to occur preferentially or with greater intensity in the rebounding stage of sleep).
Chronic restriction of nocturnal sleep, an irregular sleep schedule, or frequent disturbance of nocturnal sleep can result in a peculiar distribution of sleep states, most commonly characterized by premature REM sleep, that is, SOREMPs. Such episodes can be associated with hypnagogic hallucinations, sleep paralysis, or an increased incidence of hypnic myoclonia in persons with no organic sleep disorder.
Although not strictly related to prior sleep history, the first night of a laboratory sleep evaluation is commonly associated with a disruption of the normal distribution of sleep states, characterized chiefly by a delayed onset of REM sleep.[31] Often this delay takes the form of skipping the first REM episode of the night. In other words, the NREM sleep stages progress in a normal fashion, but the first cycle ends with an episode of stage 1 or a brief arousal instead of the expected brief REM sleep episode. In addition, REM sleep episodes are often disrupted, and the total amount of REM sleep on the first night in the sleep laboratory is also usually reduced from the normal value.
Circadian Rhythms
The circadian phase at which sleep occurs affects the distribution of sleep stages. REM sleep, in particular, occurs with a circadian distribution that peaks in the morning hours coincident with the trough of the core body temperature rhythm.[18],[19] Thus, if sleep onset is delayed until the peak REM phase of the circadian rhythm—that is, the early morning—REM sleep tends to predominate and can even occur at the onset of sleep. This reversal of the normal sleep-onset pattern is commonly seen in a normal person who acutely undergoes a phase shift, either as a result of a work shift change or as a change resulting from jet travel across a number of time zones. Studies of persons sleeping in environments free of all cues to time have shown that the timing of sleep onset and the length of sleep occur as a function of circadian phase.[32],[33] Under these conditions, sleep distribution with reference to the circadian body temperature phase position shows that sleep onset is likeliest to occur on the falling limb of the temperature cycle. A secondary peak of sleep onsets, corresponding to afternoon napping, also occurs; the offset of sleep occurs most often on the rising limb of the circadian body temperature curve.[34]
Extremes of temperature in the sleeping environment tend to disrupt sleep. REM sleep is commonly more sensitive to temperature-related disruption than is NREM sleep. Accumulated evidence from human beings and other species suggests that mammals have only minimal ability to thermoregulate during REM sleep; in other words, the control of body temperature is virtually poikilothermic in REM sleep.[35] This inability to thermoregulate in REM sleep probably affects the response to temperature extremes and suggests that such conditions are less of a problem early during a night than late, when REM sleep tends to predominate. It should be clear, as well, that such responses as sweating or shivering during sleep under ambient temperature extremes occur in NREM sleep and are limited in REM sleep.
The distribution of sleep states and stages is affected by many common drugs, including those typically prescribed in the treatment of sleep disorders as well as those not specifically related to the pharmacotherapy of sleep disorders and those used socially or recreationally. Whether changes in sleep stage distribution have any relevance to health, illness, or psychological well-being is unknown; however, particularly in the context of specific sleep disorders that differentially affect one sleep stage or another, such distinctions may be relevant to diagnosis or treatment. A number of generalizations regarding the effects of certain of the more commonly used compounds on sleep stage distribution can be made.
• Benzodiazepines tend to suppress SWS and have no consistent effect on REM sleep.
• Tricyclic antidepressants, monoamine oxidase inhibitors, and certain selective serotonin reuptake
inhibitors tend to suppress REM sleep. An increased level of motor activity during sleep occurs with
certain of these compounds, leading to a pattern of REM sleep without motor inhibition or an increased incidence of PLMS. Fluoxetine is also associated with rapid eye movements across all sleep stages ("Prozac eyes").
• Withdrawal from drugs that selectively suppress a stage of sleep tends to be associated with a
rebound of that sleep stage. Thus, acute withdrawal from a benzodiazepine compound is likely to produce an increase of SWS; acute withdrawal from a tricyclic antidepressant or monoamine
oxidase inhibitor is likely to produce an increase of REM sleep. In the latter case, this REM rebound could result in abnormal SOREMPs in the absence of an organic sleep disorder, perhaps leading to an incorrect diagnosis of narcolepsy.
• Acute presleep alcohol intake can produce an increase in SWS and REM sleep suppression early
in the night, which can be followed by REM sleep rebound in the latter portion of the night as the
alcohol is metabolized. Low doses of alcohol have minimal effects on sleep stages, but they can increase sleepiness late at night.[36],[37]
• Acute effects of marijuana (tetrahydrocannabinol [THC]) include minimal sleep disruption,
characterized by a slight reduction of REM sleep. Chronic ingestion of THC produces a long-term suppression of SWS.[38]
Sleep disorders, as well as other nonsleep problems, have an impact on the structure and distribution of sleep. As suggested before, these distinctions appear to be more important in diagnosis and in the consideration of treatments than for any implications about general health or illness resulting from specific sleep stage alterations. A number of common sleep-stage anomalies are commonly associated with sleep disorders.
Narcolepsy
Narcolepsy is characterized by an abnormally short delay to REM sleep, marked by SOREMPs. This abnormal sleep-onset pattern occurs with some consistency, but not exclusively; that is, NREM sleep onset can also occur. Thus, the preferred diagnostic test consists of several opportunities to fall asleep across a day (see Chapter 143). If REM sleep occurs abnormally on two or more such opportunities, narcolepsy is extremely probable. The occurrence of this abnormal sleep pattern in narcolepsy is thought
to be responsible for the rather unusual symptoms of this disorder. In other words, dissociation of components of REM sleep into the waking state results in hypnagogic hallucinations, sleep paralysis, and, most dramatically, cataplexy.
Other conditions in which a short REM sleep latency can occur include infancy, in which sleep-onset REM sleep is normal; sleep reversal or jet lag; acute withdrawal from REM-suppressant compounds; chronic restriction or disruption of sleep; and endogenous depression.[39] Reports have indicated a relatively high prevalence of REM sleep onsets in young adults[40] and in adolescents with early rise times.[41] In the latter, the REM sleep onsets on morning (8:30 AM and 10:30 AM) naps were related to a delayed circadian phase as indicated by later onset of melatonin secretion.
Sleep Apnea Syndromes
Sleep apnea syndromes may be associated with suppression of SWS or REM sleep secondary to the sleep-related breathing problem. Successful treatment of this sleep disorder, as with nocturnal continuous positive airway pressure, can produce large rebounds of SWS or REM sleep (Fig. 2-10).
Figure 2-10 These sleep histograms depict the sleep of a 64-year-old male patient with obstructive sleep
apnea syndrome. The let graph shows the sleep pattern before treatment. Note the absence of slow-wave sleep, the preponderance of stage 1 (S1), and the very frequent disruptions. The right graph shows the sleep pattern in this patient during the second night of treatment with continuous positive airway pressure (CPAP). Note that sleep is much deeper (more SWS) and more consolidated, and rapid eye movement (REM) sleep in particular is abnormally increased. The pretreatment REM percentage of sleep was only 10%, versus nearly 40% with treatment. (Data supplied by G. Nino-Murcia, Stanford University Sleep Disorders Center, Stanford, Calif.)
Sleep Fragmentation
Fragmentation of sleep and increased frequency of arousals occur in association with a number of sleep disorders as well as with medical disorders involving physical pain or discomfort. PLMS, sleep apnea syndromes, chronic fibrositis, and so forth may be associated with tens to hundreds of arousals each night. Brief arousals are prominent in such conditions as allergic rhinitis,[42],[43] juvenile rheumatoid arthritis,[44] and Parkinson's disease.[45] In upper airway resistance syndrome,[46] EEG arousals are important markers because the respiratory signs of this syndrome are less obvious than in frank obstructive sleep apnea syndrome, and only subtle indicators may be available.[47] In specific situations, autonomic changes, such as transient changes of blood pressure,[48] can signify arousals; Lofaso and colleagues[49] indicated that autonomic changes are highly correlated with the extent of EEG arousals. Less well studied is the possibility that sleep fragmentation may be associated with subcortical events not visible in the cortical EEG signal. These disorders also often involve an increase in the absolute amount of and the proportion of stage 1 sleep.
Clinical Pearls
The clinician should expect to see less slow-wave sleep (stages 3 and 4) in older persons, particularly men.
Clinicians or colleagues might find themselves denying mid-night communications (nighttime calls) because of memory deficits that occur for events proximal to sleep onset. This phenomenon might also account for memory deficits in excessively sleepy patients.
Many medications (even if not prescribed for sleep) can affect sleep stages, and their use or discontinuation alters sleep. Thus, REM-suppressant medications, for example, can result in a rebound of REM sleep when they are discontinued.
Certain patients have sleep complaints (insomnia, hypersomnia) that result from attempts to sleep or be awake at times not in synchrony with their circadian phase.
Patients who wake with events early in the night might have a disorder affecting NREM sleep; patients who wake with events late in the night may have a disorder affecting REM sleep.
When using sleep restriction to build sleep pressure, treatment will be more effective if sleep is scheduled at the correct circadian phase. The problem of napping in patients with insomnia is that naps diminish the homeostatic drive to sleep.
References
1. Dement W, Kleitman N: The relation of eye movements during sleep to dream activity: an objective
method for the study of dreaming. J Exp Psychol 1957; 53:339-346.
2. Agnew HW, Webb WB: Measurement of sleep onset by EEG criteria. Am J EEG
Technol 1972; 12:127-134.
3. Davis H, Davis PA, Loomis AL, et al: Human brain potentials during the onset of sleep. J
Neurophysiol 1938; 1:24-38.
4. Carskadon MA, Dement WC: Effects of total sleep loss on sleep tendency. Percept Mot
Skills 1979; 48:495-506.
5. Guilleminault C, Phillips R, Dement WC: A syndrome of hypersomnia with automatic
behavior. Electroencephalogr Clin Neurophysiol 1975; 38:403-413.
6. Ogilvie RD, Wilkinson RT: The detection of sleep onset: behavioral and physiological
convergence. Psychophysiology 1984; 21:510-520.
7. Carskadon MA, Herz R: Minimal olfactory perception during sleep: why odor alarms will not work for
humans. Sleep 2004; 27:402-405.
8. Williams HL, Hammack JT, Daly RL, et al: Responses to auditory stimulation, sleep loss and the EEG
stages of sleep. Electroencephalogr Clin Neurophysiol 1964; 16:269-279.
9. Oswald I, Taylor AM, Treisman M: Discriminative responses to stimulation during human
sleep. Brain 1960; 83:440-453.
10. Williams HL, Morlock HC, Morlock JV: Instrumental behavior during
sleep. Psychophysiology 1966; 2:208-216.
11. Portas CM, Krakow K, Allen P, et al: Auditory processing across the sleep-wake cycle: simultaneous
EEG and fMRI monitoring in humans. Neuron 2000; 28:991-999.
12. Foulkes D: The psychology of sleep. New York, Charles Scribner's Sons, 1966.
13. Wyatt JK, Bootzin RR, Anthony J, et al: Does sleep onset produce retrograde amnesia?. Sleep Res 1992; 21:113. 14. Maquet P: The role of sleep in learning and memory. Science 2001; 294:1048-1052. 15. Stickgold R, Hobson JA, Fosse R, et al: Sleep, learning and dreams: Off-line memory reprocessing. Science 2001; 294:1052-1057. 16. Siegel J: The REM sleep-memory consolidation hypothesis. Science 2001; 294:1058-1063. 17. A manual of standardized terminology, techniques and scoring system for sleep stages of human subjects. In: Rechtschaffen A, Kales A, ed. Los Angeles: UCLA Brain Information Service/Brain Research Institute; 1968. 18. Czeisler CA, Zimmerman JC, Ronda JM, et al: Timing of REM sleep is coupled to the circadian rhythm of body temperature in man. Sleep 1980; 2:329-346. 19. Zulley J: Distribution of REM sleep in entrained 24 hour and free-running sleep-wake cycles. Sleep 1980; 2:377-389. 20. Weitzman ED, Czeisler CA, Zimmerman JC, et al: Timing of REM and stages 3 + 4 sleep during temporal isolation in man. Sleep 1980; 2:391-407. 21. Karacan I, Moore CA: Genetics and human sleep. Psychiatr Ann 1979; 9:11-23. 22. Busby K, Pivik RT: Failure of high intensity auditory stimuli to affect behavioral arousal in children during the first sleep cycle. Pediatr Res 1983; 17:802-805. 23. Carskadon MA, Dement WC: Sleepiness in the normal adolescent. In: Guilleminault C, ed. Sleep and its disorders in children, New York: Raven Press; 1987:53-66. 24. Feinberg I: Schizophrenia: caused by a fault in programmed synaptic elimination during adolescence?. J Psychiatr Res 1983; 17:319-334. 25. Prinz P: Sleep patterns in the healthy aged: relationship with intellectual function. J Gerontol 1977; 32:179-186. 26. Prinz PN, Peskind ER, Vitaliano PP, et al: Changes in the sleep and waking EEGs of nondemented and demented elderly subjects. J Am Geriatr Soc 1982; 30:86-93. 27. Carskadon MA, Brown ED, Dement WC: Sleep fragmentation in the elderly: relationship to daytime sleep tendency. Neurobiol Aging 1982; 3:321-327. 28. Ancoli-Israel S, Kripke DF, Mason W, et al: Sleep apnea and nocturnal myoclonus in a senior population. Sleep 1981; 4:349-358. 29. Carskadon MA, Dement WC: Respiration during sleep in the aging human. J Gerontol 1981; 36:420-423. 30. Williams RL, Karacan I, Hursch CJ: EEG of human sleep: clinical applications. New York, John Wiley & Sons, 1974. 31. Agnew HW, Webb WB, Williams RL: The first-night effect: an EEG study of sleep. Psychophysiology 1966; 2:263-266. 32. Czeisler CA, Weitzman ED, Moore-Ede MC, et al: Human sleep: Its duration and organization depend on its circadian phase. Science 1980; 210:1264-1267. 33. Zulley J, Wever R, Aschoff J: The dependence of onset and duration of sleep on the circadian rhythm of rectal temperature. Pflugers Arch 1981; 391:314-318. 34. Strogatz SH: The mathematical structure of the human sleep-wake cycle. New York, Springer-Verlag, 1986. 35. Parmeggiani PL: Temperature regulation during sleep: a study in homeostasis. In: Orem J, Barnes CD, ed. Physiology in sleep, New York: Academic Press; 1980:98-143. 36. Van Reen E, Jenni O, Carskadon MA: Effects of alcohol on sleep and the sleep electroencephalogram in healthy young women. Alcohol Clin Exp Res 2006; 30(6):974-981. 37. Rupp TL, Acebo C, Van Reen E, Carskadon MA: Effects of a moderate evening dose of alcohol. I. Sleepiness. Alcohol Clin Exp Res 2007; 31(8):1358-1364. 38. Freemon FR: The effect of chronically administered delta-9-tetrahydrocannabinol upon the polygraphically monitored sleep of normal volunteers. Drug Alcohol Depend 1982; 10:345-353. 39. Kupfer DJ: REM latency: a psychobiologic marker for primary depressive disease. Biol Psychiatry 1976; 11:159-174. 40. Bishop C, Rosenthal L, Helmus T, et al: The frequency of multiple sleep onset REM periods among subjects with no excessive daytime sleepiness. Sleep 1996; 19:727-730. 41. Carskadon MA, Wolfson AR, Acebo C, et al: Adolescent sleep patterns, circadian timing, and sleepiness at a transition to early school days. Sleep 1998; 21:871-881.
42. Lavie P, Gertner R, Zomer J, et al: Breathing disorders in sleep associated with "microarousals" in patients with allergic rhinitis. Acta Otolaryngol 1981; 92:529-533. 43. Craig TJ, Teets S, Lehman EB, et al: Nasal congestion secondary to allergic rhinitis as a cause of sleep disturbance and daytime fatigue and the response to topical nasal corticosteroids. J Allergy Clin Immunol 1998; 101:633-637. 44. Zamir G, Press J, Tal A, et al: Sleep fragmentation in children with juvenile rheumatoid arthritis. J Rheumatol 1998; 25:1191-1197. 45. Stocchi F, Barbato L, Nordera G, et al: Sleep disorders in Parkinson's disease. J Neurol 1998; 245(Suppl. 1):S15-S18. 46. Guilleminault C, Stoohs R, Clerk A, et al: From obstructive sleep apnea syndrome to upper airway resistance syndrome—consistency of daytime sleepiness. Sleep 1992; 15(6 Suppl.):S13-S16. 47. Hosselet JJ, Norman RG, Ayappa I, et al: Detection of flow limitation with a nasal cannula/pressure transducer system. Am J Respir Crit Care Med 1998; 157:1461-1467. 48. Pitson DJ, Stradling JR: Autonomic markers of arousal during sleep in patients undergoing investigation for obstructive sleep apnoea, their relationship to EEG arousals, respiratory events and subjective sleepiness. J Sleep Res 1998; 7:53-59. 49. Lofaso F, Goldenberg F, Dortho MP, et al: Arterial blood pressure response to transient arousals from NREM sleep in nonapneic snorers with sleep fragmentation. Chest 1998; 113:985-991. 50. Iber C, Ancoli-Israel S, Quan SF: The AASM manual for the scoring of sleep and associated events: rules, terminology, and technical specifications. for the American Academy of Sleep Medicine1st ed. Westchester, Ill, American Academy of Sleep Medicine, 2007. 51. Moser D, Anderer P, Gruber G, et al: Sleep classification according to AASM and Rechtschaffen & Kales: effects on sleep scoring parameters. Sleep 2009; 32:139-149. 52. Danker-Hopfe H, Anderer P, Zeitlhofer J, et al: Interrater reliability for sleep scoring according to the Rechtschaffen & Kales and the new AASM standard. J Sleep Res 2009; 18:74-84. 53. Parrino L, Ferri R, Zucconi M, Fanfulla F: Commentary from the Italian Association of Sleep Medicine on the AASM manual for the scoring of sleep and associated events: for debate and discussion. Sleep Med 2009; 10:799-808. 54. Grigg-Damberger MM: The AASM scoring manual: a critical appraisal. Curr Opin Pulm Med 2009; 15:540-549. 55. Novelli L, Ferri R, Bruni O: Sleep classification according to AASM and Rechtschaffen and Kales: effects on sleep scoring parameters of children and adolescents. J Sleep Res 2010; 19:238-247. 56. Miano S, Paolino MC, Castaldo R, Villa MP: Visual scoring of sleep: A comparison between the Rechtschaffen and Kales criteria and the American Academy of Sleep Medicine criteria in a pediatric population with obstructive sleep apnea syndrome. Clin Neurophysiol 2010; 121:39-42.
Source: http://readinglists.northumbria.ac.uk/link?url=http%3A%2F%2Fapsychoserver.psych.arizona.edu%2Fjjbareprints%2Fpsyc501a%2Freadings%2FCarskadon%2520Dement%25202011.pdf&sig=0d288f2713f539ed222a0904ee8a6b502856ccca45715a3444a03c1f2979d1e9
ContentsIntroduction What is Alzheimer's? 02 visit: www.alzheimersresearchuk.org This introductory booklet aims to provide an overview of Alzheimer's disease. It is for anyone who wants to know more about the disease, including people living with Alzheimer's, their carers, friends and family. The information here does not replace any advice that doctors, pharmacists or nurses may give you. It provides background information which we hope you will find helpful.
Allicin and methicillin-resistant Staphylococcus aureus Antibacterial activity of a new, stable, aqueousextract of allicin against methicillin-resistantStaphylococcus aureus R R. CUTLER* and P. WILSON† *University of East London, School of Health and Bioscience, Stratford Campus,Romford Road, London E15 4LZ; and Department of Medical Microbiology,















