Vasiliadis-books.gr
Cambridge University Press978-0-521-88109-8 - Surgical and Medical Management of Male InfertilityMarc Goldstein and Peter N. SchlegelExcerptMore information
Section 1
Anatomy and Physiology of the Male Reproductive System
Anatomy and physiology of the male
The male reproductive hormonal axis
known as the adenohypophysis, in contrast communicates via
bloodborne factors secreted by the hypothalamus. These neu
Maintenance of normal reproductive function is depend
ropeptides are transported by the portal blood system to the
ent on the coordinated release of hormones in the hyp o
anterior lobe where they stimulate synthesis and secretion of
thalamic−pituitary−testis cascade. Gonadotropinreleasing
adenohypophyseal hormones.
Including the gonadotropes, LH
hormone (GnRH) is released in a pulsatile pattern into the pitu
and FSH, the anterior pituitary also secretes other glycoprotein
itary portal blood system from neuroendocrine cel s in the basal
hormones, corticotropin-related peptides, and somatomam-
hypothalamus and acts to stimulate gonadotropes in the anter
motropin hormones. Prolactin and growth hormone have a sig
ior pituitary to synthesize and release two peptide hormones,
nificant contribution to male reproductive function.
folliclestimulating hormone (FSH) and luteinizing hormone
(LH) into the circulation. Once in the bloodstream these hor
mones reach the testis, where
LH stimulates testosterone pro-
Steroid feedback loop
duction by the Leydig cells in the interstitium while FSH
Testosterone provides negative feedback suppression of the
supports spermatogenesis in the seminiferous epithelium by
release of GnRH through androgen receptors in the pituitary
stimulation of the Sertoli cells. A focused network of negative
and hypothalamic neurons. Testosterone is also readily metab
feedback relationships finesse testosterone secretion and sperm
olized to dihydrotestosterone and estradiol by 5αreductase
production. This cascade is maintained by steroid and peptide
and aromatase, respectively, in the testis and peripheral tissues.
feedback within the testis as well as the hypothalamic and pitu
Both testosterone and estrogen play important roles in the
itary gland (see Figure 1.1).
regulation of reproductive function at the cellular and tis-
sue levels. This can be demonstrated clinical y by individuals
with genetic mutations resulting in partial or complete loss of
The hypothalamus is located in the lower aspect of the third
function in androgen and estrogen receptors with increased
ventricle of the brain. It is a complex region in the brain, which
pituitary release of LH [1]. Stimulation of the Sertoli cell by
responds to different signals generated external y and intern
FSH results in production of inhibin, a glycoprotein hormone,
al y as it is richly connected by neural projections to other
which suppresses FSH secretion by gonadotropes. Regulation
parts of the brain including the amygdala as well as the olfac
of gonadotropin secretion exists for different steroids. While
tory bulbs. The output of GnRH is influenced by three different
the negative effect of testosterone on LH secretion is primarily
rhythms including seasonal, circadian, and pulsatile, leading to
mediated by the androgen itself, the effect of testosterone on
peak levels during the spring, the early morning hours as well
FSH secretion is mediated by estradiol [2].
as every 90 to 120 minutes. The precursors of GnRH neurons
migrate to their position in the hypothalamus from the olfac
Development of the male reproductive axis
tory placode during embryonic development.
Testosterone, dihydrotestosterone, and müllerian inhibiting
substance from the fetal testes are important determinants
of sexual phenotype. Sertoli cell precursors secrete müllerian
The pituitary body is found in the hypophyseal fossa inferior to
inhibiting substance (also known as antimüllerian hormone)
the hypothalamus. It is divided into two distinct lobes: anterior
which prevents the development of the female reproductive
and posterior. The posterior lobe is known as the neurohypo
tract structures leading to a male phenotype [3]. Secretion of
physis and is stimulated by neurons from the hypothalamus to
testosterone by the fetal Leydig cel s stimulates differentiation
secrete both oxytocin and vasopressin. The anterior lobe, also
of the wolffian duct system, which later develops into the vas
Surgical and Medical Management of Male Infertility, Marc Goldstein and Peter N. Schlegel. Published by Cambridge University Press. Cambridge
University Press 2013.
in this web service Cambridge University Press
Cambridge University Press978-0-521-88109-8 - Surgical and Medical Management of Male InfertilityMarc Goldstein and Peter N. SchlegelExcerptMore information
Section 1: Anatomy and Physiology
Tunica albuginea
Figure 1.2 Schematical drawing of the human testis showing seminiferous
tubules, epididymis, and vas deferens.
made up of three distinct layers surrounds the parenchyma of
Figure 1.1 Diagram of the hypothalamic−pituitary−testis axis.
the testis: the tunica vaginalis, tunica albuginea, and the tunica
vasculosa. The testes receive their blood supply from the tes
ticular arteries, the cremasteric arteries, and the deferential
deferens, epididymis, and sex accessory glands.
Events of early
arteries. The testicular artery arises from the abdominal aorta
testis differentiation are controlled by the sex-determining
just below the renal artery and becomes a component of the
region on the Y chromosome (SRY) gene. The SRY gene acts
spermatic cord above the internal inguinal ring as well as being
synergistical y with other transcription factors to initiate male
intimately associated with a network of anastomotic veins,
sex differentiation [4].
which eventual y form the pampiniform plexus. A counter cur
Aging of the male reproductive system
rent heat exchange in the spermatic cord provides blood to the
testis that is 2 to 4°C lower than the rectal temperature in a
Epidemiologic studies have demonstrated that with age, circulat
normal male [11]. The testicular arteries penetrate the tunica
ing levels of testosterone progressively decline [5]. This decline
albuginea and then travel inferiorly along the posterior surface
in androgen levels is associated with modifications in body
of the testicle and eventual y ascend onto the anterior surface
composition, diminished energy, muscle strength and physical
with several branches that course into the parenchyma. The
function, reduced sexual function, and depressed and decreased
location of these vessels should be considered as they may be
cognitive function [6]. This agerelated decline of testosterone
injured during biopsy or orchidopexy. The medial and lateral
levels is complex involving both intrinsic and extrinsic factors
aspects of the superior pole have the lowest density of super
to the Leydig cel . The brown Norway rat has become a well
ficial vessels compared with the inferior and anterior portions
established model for human reproductive aging, and studies
of the testicle.
have reported that the number of Leydig cel s per testis remains
The testicle is divided into compartments by septa, which
unchanged compared to younger controls [7]. These findings
are projections of the tunica albuginea. Each septum separates
suggest that functional changes to the Leydig cel s rather than
the seminiferous tubules, as well as the interstitial tissue which
their loss account for the reduction in testosterone [8]. High tes
is composed of Leydig cel s, blood vessels, lymphatics, mast
ticular concentrations of testosterone are essential to maintain
cel s, nerves, and macrophages.
The seminiferous tubules are
spermatogenesis and men older than age 40 years have been
the site of germ cell production. They are looped tubules con
shown to have significantly lower fecundity [9]. Further studies
tinuous at their ends with the rete testis, a network of collecting
are likely to center on investigation of the intracel ular molecu
tubes that eventual y coalesce to form efferent ducts that pro
lar mechanisms leading to decreased Leydig cell steroidogen
vide a conduit for collecting the testicular fluid and spermato
esis. These may lead to the discovery of potential methods by
zoa to the caput epididymis. The seminiferous tubule is made
which to ameliorate the decline of testosterone synthesis in the
up primarily of Sertoli cel s, germ cel s, and peritubular myoid
aging male, which may have an impact on male infertility.
cel s (see Figure 1.2).
Macroscopic anatomy of the testis
The Leydig cell
The ovoid testis in young healthy males has a volume of 15 to
Leydig cel s can be identified by their location in the intersti
25 mL and a longitudinal length of 4.5 to 5.1 cm [10]. A capsule
tium of the testis. They are distinguished by the presence of a
in this web service Cambridge University Press
Cambridge University Press978-0-521-88109-8 - Surgical and Medical Management of Male InfertilityMarc Goldstein and Peter N. SchlegelExcerptMore information
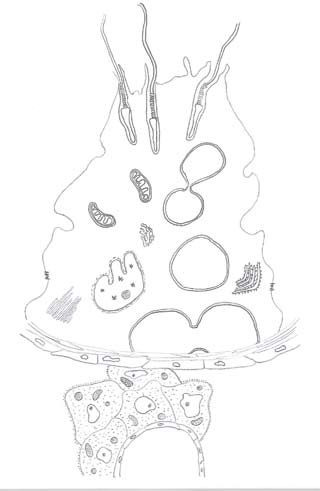
Chapter 1: Anatomy and physiology
Figure 1.3 A diagrammatic representation of the
seminiferous tubule and the interstitium of the
Tight junctionalcomplex
round nucleus, prominent nucleolus, and Reinke crystals in the
this provides an arrangement such that each germ cell is sup
cytoplasm. Numerous gap junctions allow direct communica
ported by a number of adjacent Sertoli cel s [13]. The Sertoli cell
tions between Leydig cel s. The Leydig cell is responsible for
has several distinct functions that facilitate the maturation
the majority of androgen steroid production. The potency of
of the germ cells. First, it provides a physical scaffold upon
the steroid hormones secreted by Leydig cel s is reflected by
which the germ cells develop and migrate towards the lumen
the small percentage of the human testis occupied by Leydig
of the tubule. Second, the Sertoli cell forms the blood−testis
cel s [12]. Circulating levels of testosterone in the serum dra
barrier with specialized tight junctions that exist between
matical y fluctuate in a life cycle. The maximal concentration
these cells. Third, Sertoli cells create the focused microenvir-
of testosterone is reached during the second and third decade
onment essential for germ cell maturation. These distinctive
of life, reaches a plateau, and then starts to decline thereafter.
functions also encompass phagocytosis, fluid secretion, and
Leydig cell function is regulated by pituitary hormones, para
production of a variety of molecules (see Figure 1.3).
crine factors secreted by cel s within the seminiferous tubules,
The quest for a Sertoli cell product as a marker of Sertoli cell
as well as autoregulatory factors.
function that is helpful in the evaluation of an infertile patient
The Sertoli cell
has yet to be elucidated. Androgenbinding protein was one of
the first extracel ular protein markers identified in the 1970s
The Sertoli cell is a nondividing somatic cell of epithelial ori
and since then a myriad of other secretory products have been
gin that rests on the basement membrane and forms the wall
discovered as wel . Inhibin is a glycoprotein hormone secreted
of the tubule. An irregularly shaped nucleus, prominent nucle
primarily by the Sertoli cel s and suppresses FSH secretion.
olus, low mitotic index, Sertoli−germ cell connections, as well
Some have proposed that serum inhibin B could possibly be
as unique tight junctional complexes between adjacent Sertoli
an independent marker of impaired testicular function, as well
cell membranes characterize this unique cel . Surface processes
as a predictor of the presence of sperm in the testes of infertile
from the Sertoli cel s extend outward to surround germ cel s as
in this web service Cambridge University Press
Cambridge University Press978-0-521-88109-8 - Surgical and Medical Management of Male InfertilityMarc Goldstein and Peter N. SchlegelExcerptMore information
Section 1: Anatomy and Physiology
The blood−testis barrier
spermatogonia and primary spermatocytes that are present at
Within the testis there exists a functional blood−testis barrier.
birth. There appears to be very little activity in further devel
This barrier, made up of specialized junctions, creates a division
opment until the onset of puberty. There are three types of
in the seminiferous epithelium between adjacent Sertoli cel s
spermatogonia: the dark type A, the pale type A, and the type
that forms a basal compartment and an adluminal compart
B spermatogonia. These cel s undergo several mitotic divisions
ment [15]. The basal compartment is accessible to bloodborne
to produce a large number of cel s that will either participate in
substances via the extracel ular spaces; however, due to the
stem cell renewal or go on to create daughter cel s, which will
occluding nature of the barrier these substances are prevented
later become spermatocytes.
from directly reaching the adluminal compartment. The adlu
Primary spermatocytes are unique in that they undergo
minal compartment contains mature germ cel s, while the basal
two successive cell divisions that produce the spermatids.
compartment contains spermatogonia and young spermato
This process, called meiosis, comprises two cell divisions fol
cytes. The different functional components of the blood−testis
lowing replication of the chromosomes, generating haploid
barrier include the tight junctional complexes between Sertoli
germ cells. It is the fusion of the haploid spermatozoon with
cel s, peritubular myoid cel s, as well as the endothelial cel s in
an equally haploid ovum that restores the diploid number of
the nearby capil aries [16]. The clinical significance of this bar
chromosomes in the cells of the embryo. There are two meiotic
rier is reflected by postpubertal protection of the adluminal
divisions involving primary and secondary spermatocytes.
(inferior) compartment of the testis from postpubertal tes
Each meiotic division is comprised of four distinct phases
ticular insults and the lack of development of antisperm anti
including prophase, metaphase, telophase, and anaphase.
bodies unless this barrier is breached.
Primary prophase I is long and subdivided into five stages:
leptotene, zygotene, pachytene, diplotene, and diakinesis.
Spermiogenesis refers to the dramatic metamorphosis that
a round spermatid undergoes to become an elongated fla-
Spermatogenesis is an elaborate process of cell differentiation
gellar cell capable of motility. This transformation includes
concluding with development of the ful y differentiated highly
the development of the acrosome, condensation of chroma
specialized haploid motile spermatozoa. Spermatogonia, the
tin, formation of the flagellum, and migration of cytoplasmic
most immature germ cel s, reside along the basement mem
organelles [18].
brane of the seminiferous tubule in the basal compartment.
The sperm head consists principally of a nucleus, which
The slow evolution of spermatogonia into highly special-
contains the condensed chromatin material as well as the
ized spermatozoa requires approximately 64 days [17]. The
acrosome. The acrosome is a membranebound organelle that
first mitotic divisions occur in the fetal testis generating the
contains the hydrolytic enzymes necessary for penetration of
Figure 1.4 Diagram of a typical mammalian
spermatozoon. Cross-sectional insets show the
orientation of the internal cell structure.
Mitochondrial sheath
in this web service Cambridge University Press
Cambridge University Press978-0-521-88109-8 - Surgical and Medical Management of Male InfertilityMarc Goldstein and Peter N. SchlegelExcerptMore information
Chapter 1: Anatomy and physiology
the egg before fertilization [19]. The flagellum forms at the
communicate directly with the pampiniform plexus. The veins
lower pole where the mitochondria coalesce and generate the
arising from the cauda and distal corpus eventual y communi
energy needed for motility. The mitochondria are arranged
cate with the deferential or cremasteric veins.
in a helical pattern surrounding a set of outer fibers and the
characteristic 9 + 2 microtubular structure of the axoneme
Functions of the epididymis
The initiation and maintenance of normal spermatogen-
The three primary functions of the epididymis are sperm mat-
esis is dependent on the synergistic effect of FSH and testos-
uration, sperm transport, and sperm storage. Maturational
changes allow sperm the capacity to become motile and fer
[20]. While germ cel s require these hormones they do
tilize as they transit from the testis, through the epididymis to
not possess receptors for either FSH or testosterone. Sertoli cel s
the vas deferens. It has been shown that as human spermato
possess both these receptors and it is thought that the actions of
zoa migrate through the epididymis they develop increased
FSH and testosterone are mediated by the Sertoli cel .
motility. In comparison to the caput epididymis the more dis
Genetic factors critical for spermatogenesis are being
tal portions of the epididymis house a higher percentage of
rapidly elucidated. Investigations in men with severely
impaired spermatogenesis led to the discovery of submicro-
spermatozoa capable of efficient motility [25]. While the exact
scopic deletions of a region of the Y chromosome
mechanisms that detail sperm maturation are not ful y under
stood, the consensus is that these processes are potentiated
These regions are referred to as the azoospermic factor (AZF)
through the interaction with the epididymis during migration
a, b, and c with distinct genes that have been deleted in azoo
into more distal regions of the duct.
spermic men such as the DAZ (deleted in azoospermia) gene
The transit time of the sperm in the human epididy-
found in the AZFc region. Localization of specific genes that
mis averages 12 days but is highly variable, with some
are critical for spermatogenesis remains the subject of active
sperm moving ahead through the epididymis in as little as
2 days [26–28]. Transport through the proximal epididymal
duct is principal y due to spontaneous, peristaltic contrac
tions of the smooth muscle that surrounds the epididymal
Spermatozoa acquire the capacity to become ful y motile as
duct. Other contributing factors that aid in the transport of
well as the ability to recognize and fertilize an egg within the
sperm include motile cilia as wel as the flow of the secreted
epididymis. These transformations of spermatozoa are called
testicular fluid. Sperm transport time through the epididymis
sperm maturation. Sperm motility and fertilization capacity
has also been shown to vary with age and sexual activity with
are both androgendependent processes. The loss of androgens
a direct correlation to the differences in daily rate of sperm
results in the loss of epididymal weight, as well as changes in
production [29].
the components of the epididymal fluid secretions [22]. The
In humans the major storage site of spermatozoa is the
epididymis, derived from the wolffian (mesonephric) duct, is
cauda epididymis where approximately half of the total num
an organ consisting of a single highly convoluted duct, which
ber of spermatozoa are stored [26]. It has been suggested that
the testicular sperm must pass through. It is attached to the
preservation of sperm viability and motility in humans is not as
superior and inferior pole of the testis and is closely applied
efficient as it is in other species [30]. While there are numerous
to the posterior aspect. The epididymis is divided into three
studies using experimental animals, the fate of unejaculated
major regions: caput, corpus, and cauda. The caput epididymis
sperm is still unknown.
overlies the superior pole of the testis and the cauda overlies
the inferior pole of the testis. The intervening region is referred
Ductus vas deferens
to as the corpus.
The vas deferens is a thick muscular tube that measures
The epididymis is surrounded by the visceral layer of the
approximately 30 to 40 cm from the cauda epididymis to the
tunica vaginalis, except over the posterior aspect, which is
point of fusion with the seminal vesicle and ejaculatory ducts.
attached to the scrotum and spermatic cord by a fibrofatty
Five portions have been previously described: epididymal,
connective tissue. Approximately 10 ductuli efferentes arise
scrotal, inguinal, pelvic, and ampul a. The vas deferens, like the
from the rete testis that eventually come together to form a
epididymis and seminal vesicle, is derived from the mesone
single epididymal duct. In humans, the epididymal tubule is
phric duct. The ability to propel sperm forceful y is depend
approximately 3 to 4 m in length [23]. The vascular supply to
ent on a threelayered muscular coat, with an inner and outer
the epididymis is from two sources. The caput and corpus are
longitudinal layer and a middle circular layer. While the vas
supplied from the superior and inferior epididymal branches of
deferens receives nerve fibers from both the sympathetic and
the testicular artery. The cauda is supplied from the branches of
parasympathetic nervous system, the rich supply of adrenergic
the deferential artery. This blood supply is characterized by tor
fibers contributes to the efficiency of sperm transport. The vas
tuosity of the vessels as well as a large number of anastomotic
deferens receives its blood supply from the deferential artery
communications [24]. While the venous drainage of the epi
via the inferior vesical artery, and the deferential vein accom
didymis may vary, the veins of the caput and proximal corpus
in this web service Cambridge University Press
Cambridge University Press978-0-521-88109-8 - Surgical and Medical Management of Male InfertilityMarc Goldstein and Peter N. SchlegelExcerptMore information
Section 1: Anatomy and Physiology
• Approximately 10 ductuli efferentes arise from the rete
testis that eventual y come together to form a single
• LH stimulates testosterone production by the Leydig cel s
epididymal duct. In humans, the epididymal tubule is
in the interstitium while FSH supports spermatogenesis in
approximately 3 to 4 m in length [23].
the seminiferous epithelium by stimulation of the Sertoli
• The three primary functions of the epididymis are sperm
maturation, sperm transport, and sperm storage.
• The hypothalamus is a complex region in the brain that
• The transit time of the sperm in the human epididymis
responds to different signals generated external y and
averages 12 days but is highly variable, with some sperm
internal y as it is richly connected by neural projections to
moving ahead through the epididymis in as little as 2 days
other parts of the brain including the amygdala as well as
the olfactory bulbs.
• The vas deferens is a thick muscular tube that measures
• Apart from the gonadotropes, LH and FSH, the anterior
approximately 30 to 40 cm from the cauda epididymis to
pituitary also secretes other glycoprotein hormones,
the point of fusion with the seminal vesicle and ejaculatory
corticotropinrelated peptides, and somatomammotropin
• Both testosterone and estrogen play important roles in
the regulation of reproductive function at the cel ular and
tissue levels.
• Events of early testis differentiation are controlled by the
1. Shupnik MA, Schreihofer DA. Molecular aspects of steroid
sexdetermining region on the Y chromosome (SRY) gene.
hormone action in the male reproductive axis. J Androl
• The ovoid testis in young healthy males measures 15 to
25 mL in volume and has a longitudinal length of 4.5 to
2. Hayes FJ, Pitteloud N, DeCruz S, Crowley WF Jr, Boepple PA.
5.1 cm [10].
Importance of inhibin B in the regulation of FSH secretion in the
human male. J Clin Endocrinol Metab 2001;86:5541–6.
• A counter current heat exchange in the spermatic cord
provides blood to the testis that is 2 to 4°C lower than
3. Lee MM, Donahoe PK. Mullerian inhibiting substance: a gonadal
hormone with multiple functions. Endocr Rev 1993;14:152–64.
rectal temperature in a normal male.
• The medial and lateral aspects of the superior pole have
4. Sekido R, Lovel Badge R. Sex determination involves synergistic
action of SRY and SF1 on a specific Sox9 enhancer. Nature
the lowest density of superficial vessels compared with the
inferior and anterior portions of the testicle.
5. Harman SM, Metter EJ, Tobin JD, Pearson J, Blackman MR.
• The seminiferous tubules are the site of germ cell
Longitudinal effects of aging on serum total and free testosterone
levels in healthy men: Baltimore Longitudinal Study of Aging.
• The Sertoli cell has several distinct functions that facilitate
J Clin Endocrinol Metab 2001;86:724–31.
the maturation of the germ cel s. First, it provides a
6. Matsumoto AM. Andropause: clinical implications of the decline
physical scaffold upon which the germ cel s develop and
in serum testosterone levels with aging in men. J Gerontol A Biol
migrate towards the lumen of the tubule. Second, the
Sci Med Sci 2002;57:M76–99.
Sertoli cell forms the blood−testis barrier with specialized
7. Wang C, Leung A, SinhaHikim AP. Reproductive aging in the
tight junctions that exist between these cel s. Third, Sertoli
male brownNorway rat: a model for the human. Endocrinology
cel s create the focused microenvironment essential for
germ cell maturation.
8. Midzak AS, Chen H, Papadopoulos V, Zirkin BR. Leydig cell
• The slow evolution of spermatogonia into highly
aging and the mechanisms of reduced testosterone synthesis. Mol
specialized spermatozoa requires approximately 64 days
9. Ford WC, North K, Taylor H, et al. Increasing paternal age is
• Spermiogenesis refers to the dramatic metamorphosis
associated with delayed conception in a large population of fertile
couples: evidence for declining fecundity in older men. The
that a round spermatid undergoes to become an elongated
ALSPAC Study Team (Avon Longitudinal Study of Pregnancy
flagel ar cell capable of motility.
and Childhood). Hum Reprod 2000;15:1703–8.
• The initiation and maintenance of normal spermatogenesis
10. Winter JS, Faiman C. Pituitarygonadal relations in male children
is dependent on the synergistic effect of FSH and
and adolescents. Pediatr Res 1972;6:126–35.
testosterone [20].
11. Agger P. Scrotal and testicular temperature: its relation to sperm
• Investigations in men with severely impaired
count before and after operation for varicocele. Fertil Steril
spermatogenesis led to the discovery of submicroscopic
deletions of a region of the Y chromosome [21].
12. Kaler LW, Neaves WB. Attrition of the human Leydig cell
• Spermatozoa acquire the capacity to become ful y motile
population with advancing age. Anat Rec 1978;192:513–8.
as well as the ability to recognize and fertilize an egg
13. Nagano T. Some observations on the fine structure of the Sertoli
within the epididymis.
cell in the human testis. Z Zellforsch Mikrosk Anat 1966;73:89–106.
in this web service Cambridge University Press
Cambridge University Press978-0-521-88109-8 - Surgical and Medical Management of Male InfertilityMarc Goldstein and Peter N. SchlegelExcerptMore information
Chapter 1: Anatomy and physiology
14. von Eckardstein S, Simoni M, Bergmann M, et al. Serum inhibin
21. Girardi SK, Mielnik A, Schlegel PN. Submicroscopic
B in combination with serum folliclestimulating hormone (FSH)
deletions in the Y chromosome of infertile men. Hum Reprod
is a more sensitive marker than serum FSH alone for impaired
spermatogenesis in men, but cannot predict the presence of
22. Cohen J, Ooms MP, Vreeburg JT. Reduction of fertilizing
sperm in testicular tissue samples. J Clin Endocrinol Metab
capacity of epididymal spermatozoa by 5 alphasteroid reductase
15. Fawcett DW. Observations on the organization of the interstitial
23. Turner TT, D'Addario D, Howards SS. Further observations on
tissue of the testis and on the occluding cell junctions in the
the initiation of sperm motility. Biol Reprod 1978;19:1095–101.
seminiferous epithelium. Adv Biosci 1973;10:83–99.
24. Macmil an EW. The blood supply of the epididymis in man.
16. Dym M, Fawcett DW. The blood–testis barrier in the rat and the
Br J Urol 1954;26:60–71.
physiological compartmentation of the seminiferous epithelium.
Biol Reprod 1970;3:308–26.
25. Bedford JM, Calvin H, Cooper GW. The maturation of spermatozoa
in the human epididymis. J Reprod Fertil Suppl 1973;18:199–213.
17. Clermont Y. Kinetics of spermatogenesis in mammals:
seminiferous epithelium cycle and spermatogonial renewal.
26. Amann RP, Howards SS. Daily spermatozoal production and
Physiol Rev 1972;52:198–236.
epididymal spermatozoal reserves of the human male. J Urol
18. de Kretser DM, Kerr JB, Paulsen CA. Evaluation of the
ultrastructural changes in the human Sertoli cell in testicular
27. Johnson L, Varner DD. Effect of daily spermatozoan production
disorders and the relationship of the changes to the levels of
but not age on transit time of spermatozoa through the human
serum FSH. Int J Androl 1981;4:129–44.
epididymis. Biol Reprod 1988;39:812–7.
19. McMaster R, Yanagimachi R, Lopata A. Penetration of
28. Rowley MJ, Teshima F, Heller CG. Duration of transit of
human eggs by human spermatozoa in vitro. Biol Reprod
spermatozoa through the human male ductular system. Fertil
20. Simoni M, Gromoll J, Hoppner W, et al. Mutational analysis
29. Curtis SK, Amann RP. Testicular development and
of the folliclestimulating hormone (FSH) receptor in normal
establishment of spermatogenesis in Holstein bul s. J Anim Sci
and infertile men: identification and characterization of two
discrete FSH receptor isoforms. J Clin Endocrinol Metab
30. Bedford JM. The status and the state of the human epididymis.
Hum Reprod 1994;9:2187–99.
in this web service Cambridge University Press
Cambridge University Press978-0-521-88109-8 - Surgical and Medical Management of Male InfertilityMarc Goldstein and Peter N. SchlegelExcerptMore information
Section 1
Section 2
History and physical examination
of the infertile male
2 Moshe Wald
days near the time of ovulation, ensuring the presence of viable
sperm in the female reproductive tract during the critical 12- to
The evaluation of the infertile male consists of a variety of com-
24-hour period in which the oocyte is within the fal opian tube
ponents, which include a detailed medical, surgical, develop-
and is capable of being fertilized [2]. While exceedingly frequent
mental, and reproductive history, as well as a careful physical
intercourse may result in inadequate numbers of sperm being
examination, semen analyses and possibly other laboratory
deposited in the vagina, conversely, ovulation could be missed
tests, all performed in concert with the evaluation of the female
with infrequent sexual activity. The use of vaginal lubricants
partner. The history and physical examination, along with
during intercourse should also be determined, as some of these
appropriately obtained semen analyses, represent the core of
substances, such as Astroglide® (BioFilm, Inc., Vista, CA), K-Y
the evaluation of the infertile male. In fact, the need for dif-
Jel y® (McNeil-PPC, Inc., Skillman, NJ), Surgilube® (Fougera,
ferent laboratory tests is determined by the history and phys-
Melvil e, NY), and saliva, have been reported to negatively affect
ical examination findings. For example, a cystic fibrosis screen,
sperm motility [3,4,5]. Decreased libido, as wel as erectile or
which is not part of the routine male infertility evaluation,
ejaculatory dysfunction should be noted, as these could be asso-
should be ordered when a vas deferens can not be palpated
ciated with hypogonadism or other systemic disorders. While a
on physical examination, a finding concerning for congenital
history of absent or noticeably low ejaculate volume could also
absence of the vas deferens (CBAVD). This chapter will provide
be part of the clinical picture of hypogonadism, it also suggests
a comprehensive review of the history and physical examin-
the possibility of other conditions, including retrograde ejacula-
ation of the infertile male as well as the indications and recom-
tion, ejaculatory duct obstruction, or congenital absence of the
mended timing for the performance of each component.
vas deferens.
Genitourinary infections
Obtaining a thorough medical and reproductive history that
Information regarding any previous urinary tract infections or
explores all aspects potential y related to fertility is a key com-
sexually transmitted diseases should be obtained. A history of
ponent of the evaluation of the infertile male [1]. While the
prostatitis may lead to ejaculatory duct obstruction, and previ-
detailed history focuses on the male partner, pertinent infor-
ous pyospermia may represent an inflammatory process with an
mation regarding the reproductive status of the female partner
adverse effect on sperm production. However, the direct causa-
and the couple's fertility efforts should also be gathered.
tive relationship of these conditions to infertility has not been
confirmed [2,6]. A history of previous epididymitis should also
Sexual and reproductive history
be noted, given its possible sequelae of epididymal obstruction.
Duration of infertility and previous fertility should be determined,
Mumps orchitis and other forms of viral orchitis may
including details of any prior pregnancies achieved. Frequency
develop in post-pubertal patients. In cases of previous mumps
of sexual intercourse and masturbation should be recorded, as
infection, it is important to confirm that the disease involved
wel as the timing of coitus. It is important to determine whether
the testicles, as only 10–30% of pubertal patients who acquire
the couple attempts to time intercourse with ovulation, and
mumps develop mumps orchitis [7]. Bilateral involvement has
whether this is done in an effective manner that could optimize
been reported in 20% to 60% of cases [2].
the chances of conception. As sperm remain viable within the
cervical mucus and crypts for 48 hours or longer, the timing of
Childhood illnesses and developmental history
sexual intercourse does not have to coincide exactly with ovula-
Delayed or absent puberty may indicate an endocrine dis
tion, but most experts recommend vaginal intercourse every 2
order or an androgen receptor abnormality [8]. A history
Surgical and Medical Management of Male Infertility, Marc Goldstein and Peter N. Schlegel. Published by Cambridge University Press. Cambridge
University Press 2013.
in this web service Cambridge University Press
Cambridge University Press978-0-521-88109-8 - Surgical and Medical Management of Male InfertilityMarc Goldstein and Peter N. SchlegelExcerptMore information
Chapter 2: History and physical examination
of gynecomastia may be associated with testis cancer, hyper-
syndrome, which also includes situs inversus [18]. Frequent
prolactinemia, or estrogen abnormalities [9]. While unilateral
respiratory infections associated with azoospermia suggests the
cryptorchidism has been reported to only slightly decrease fer-
possibility of Young's syndrome, in which epididymal obstruc-
tility, bilateral cryptorchidism results in a significant reduction
tion is caused by inspissation of secretions [19]. A personal
in fertility [10,11].
or familial history of cystic fibrosis (CF) is of importance, as
almost all male patients with clinical CF have bilateral congeni-
Past surgical history
tal absence of the vas deferens [20,21].
Various surgical procedures could potential y disrupt the
Prolactinoma or other pituitary tumors should be suspected
physiologic regulation of different functions of the male repro-
with a history of severe headaches, galactorrhea, or impaired
ductive tract, as well as damage the anatomic integrity of this
visual fields. Anosmia associated with male infertility should
system at different sites along its course. Surgery or trauma of
raise the possibility of Kallmann syndrome, a congenital form
the brain or pituitary could impair the hormonal regulation of
of hypogonadotropic hypogonadism.
spermatogenesis and testicular testosterone production. Pelvic
or retroperitoneal surgery may affect erectile and ejaculatory
Medications, recreational drugs, and gonadotoxin
function. For example, retroperitoneal lymph node dissection
for testis cancer may involve sympathetic nerve injury, resulting
in failure of emission or retrograde ejaculation. Bladder neck
Certain medications, including nitrofurantoin, cimetidine,
surgery may also result in retrograde ejaculation. Inguinal her-
and sulfasalazine, have been reported to impair spermat o
nia repair may be associated with damage to the vas deferens,
genesis [2]. A similar effect has been attributed to certain rec-
by inadvertent direct injury or compromising its blood supply.
reational drugs, including cocaine [22,23] and marijuana [24],
Additional y, the vas deferens may be entrapped in dense fibro-
as well as to anabolic steroid and chronic alcohol abuse [25].
sis associated with hernia repair using mesh, leading to vasal
Additionally, the androgenic effect of steroids may cause
obstruction. Final y, scrotal surgery such as hydrocelectomy,
hypogonadotropic hypogonadism, which is usually but not
spermatocelectomy or orchidopexy for torsion may result
always reversible after the discontinuation of these agents
in injury and obstruction of the vas deferens and/or the epi-
[26]. While the effect of cigarette smoking on spermatogenesis
didymis. Testicular trauma or torsion may result in testicular
is unclear, it has been suggested that smoking could possibly be
atrophy or scarring. Furthermore, these events may lead to the
a cofactor in male patients with other causes of infertility [27].
formation of antisperm antibodies, possibly due to the disrup-
Occupational or environmental exposure to pesticides or
tion of the blood−testis barrier.
other toxic chemicals should be noted, as these substances
may have a deleterious effect on sperm production or func-
Systemic medical illnesses
tion. Additional y, a history of excessive heat exposure, either
occupational or secondary to the frequent use of saunas and
Erectile dysfunction, retrograde ejaculation, and other ejacu-
hot tubs is of relevance, as experimental hyperthermia and the
latory abnormalities may develop in patients with diabetes
frequent use of hot tubs have been shown to cause impaired
mellitus or multiple sclerosis. Many other systemic disorders
semen quality and spermatogenesis [2].
could have a negative effect on spermatogenesis. A febrile ill-
ness, even if associated with a disease that does not directly
involve the genitourinary tract, could cause spermatogenesis
impairment for up to 3 months [12]. End-stage renal disease
The family history of the infertile male should focus on the
has been reported to be associated with male infertility [2].
phenotype of the maternal uncles, as the androgen receptor
Men with testicular cancer or lymphoma may have fertility
gene, as well as multiple other genes affecting male reproduc-
difficulties even before initiation of treatment, as low sperm
tion, is located on the X chromosome.
concentrations have been reported in 60% or more of patients
at the time of diagnosis [13,14,15]. Obviously, chemotherapy
or radiotherapy administered for these conditions or other
cancers may impair spermatogenesis. While these treatments
General examination
may result in permanent azoospermia, return of spermatogen-
The physical examination of the infertile male should not be
esis is possible under certain circumstances, although it may
limited to a genital examination, and should include a detailed
take up to 4 to 5 years to occur after completion of treatment
general examination, which can reveal identifiable abnormal-
[15,16,17]. Spermatogenesis recovery following radiation ther-
ities that may be associated with infertility and its underlying
apy or chemotherapy varies, depending on the specific agents
causes. The patient's habitus should be noted, as alterations of
used, doses, and duration of treatment [17].
the normal male appearance may be associated with chromo-
A history of frequent or chronic respiratory tract infections
somal or endocrine disorders that have an impact on fertility
in the setting of male infertility and lack of sperm motility
as well as on other health issues. For example, a eunuchoid
should raise the suspicion for immotile cilia (or Kartagener's)
appearance could be associated with Klinefelter syndrome or
in this web service Cambridge University Press

Cambridge University Press978-0-521-88109-8 - Surgical and Medical Management of Male InfertilityMarc Goldstein and Peter N. SchlegelExcerptMore information
Section 2: Evaluation
Figure 2.1 Calipers used for measuring long and
short testicular axis.
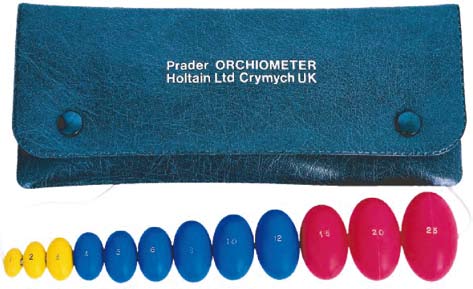
Figure 2.2 Orchometer used for measuring
testicular volume. Image reproduced with kind
permission from Prader.
hypogonadotropic hypogonadism. Additional y, abnormalities
warts, sores, herpetic-like lesions, and any urethral discharge.
of the secondary sex characteristics and changes in the pattern
The penis should be examined for any curvature or plaques,
of virilization, such as lack of temporal pattern balding, may
which could suggest Peyronie's disease. The possible presence
also indicate a congenital endocrine disorder. Other pertinent
of severe chordee should also be noted. The location of the
findings on the general physical examination include gyne-
urethral meatus should be determined, since significant hypo-
comastia, which is suggestive of either an imbalance between
spadias, as well as severe penile curvatures and chordee could
estrogen and androgen levels or increased prolactin levels, as
interfere with proper deposition of semen in the vagina.
well as situs inversus, which may be part of Kartagener's syn-
The examination of the scrotum should be performed
drome, a congenital disorder associated with immotile cilia
with the patient both supine and standing in a warm room to
leading to absent sperm motility.
allow for relaxation of the cremaster muscle. Use of a heating
pad to relax the scrotum prior to examination is very effect-
Genital examination
ive without overheating the examiner or the patient. The testi
cles should be carefully palpated to assess their consistency
A careful genital examination is a critical part of the evaluation
and to rule out the presence of an intratesticular mass. The
of the infertile male. This examination can allow for identifi-
dimensions of the testicles should be measured, using either
cation of the cause of infertility, such as in cases of bilateral y
calipers (Figure 2.1) for determination of the long and short
absent vasa deferentia or clinical y evident varicoceles, and may
testicular axis, or an orchidometer for assessment of testicu
also direct the clinician toward the next steps of the evaluation
lar volume (Figure 2.2) [28]. Testicular measurement by cali-
that are required for a given scenario. For example, the absence
pers should be done careful y, to avoid painful squeezing of the
of any palpable vas deferens on both sides suggests the diagno-
testicles. Interestingly, variations in the normal range for tes-
sis of congenital bilateral absence of the vas deferens (CBAVD),
ticular dimensions between different ethnic groups have been
a condition closely associated with cystic fibrosis, and should
reported. While the normal adult testis is greater than 4 × 3 cm
prompt genetic testing for cystic fibrosis.
in its greatest dimensions or greater than 20 mL in volume for
The entire genital area should be inspected for any find-
Caucasians and African-Americans [29], Asian men normal y
ings concerning for sexual y transmitted diseases, such as
have smaller testicles but higher sperm production per cubic
in this web service Cambridge University Press
Source: http://www.vasiliadis-books.gr/Vasiliadis-books/wp-content/uploads/2015/12/Surgical-and-Medical-Management-of-Male-Infertility-%CE%9A%CE%AC%CE%BD%CF%84%CE%B5-%CE%BA%CE%BB%CE%B9%CE%BA-%CE%B3%CE%B9%CE%B1-%CE%BD%CE%B1-%CE%B4%CE%B5%CE%AF%CF%84%CE%B5-%CE%B1%CF%80%CF%8C%CF%83%CF%80%CE%B1%CF%83%CE%BC%CE%B1-%CF%84%CE%BF%CF%85-%CE%B2%CE%B9%CE%B2%CE%BB%CE%AF%CE%BF%CF%85.pdf
How Does Psychotherapy Influence Personality?A Theoretical Integration John D. MayerUniversity of New Hampshire A given type of psychotherapy (e.g., psychodynamic) is associated with aset of specific change techniques (e.g., interpreting defenses, identifyingrelationship themes). Different change techniques can be conceived of asinfluencing different parts of personality (e.g., interpreting defense increasesconscious awareness). An integrated model of personality is presented.Then, change techniques from different theoretical perspectives are assignedby judges to areas of personality the techniques are believed to influence.The results suggest that specific change techniques can be reliably sortedinto the areas of personality. Thinking across theoretical perspectives leadsto important new opportunities for assessment, therapy outcome research,and communication with patients concerning personality change. ©2004Wiley Periodicals, Inc. J Clin Psychol 60: 1291–1315, 2004.
EUROPEAN HEART NETWORK TABLE OF CONTENTS ROUND UP THE USUAL SUSPECTS! EXECUTIVE SUMMARY AND RECOMMENDATIONS THE STRESS CONCEPT Stressors and stress Stress physiology Stress in relation to the pathogenesis of cardiovascular disease Stress and heart disease - how important is stress according to the patients and according to their doctors?










