Endo.org
The Journal of Clinical Endocrinology & Metabolism
Printed in U.S.A.
Copyright 2001 by The Endocrine Society
A Randomized Trial Comparing Levothyroxine with
Radioactive Iodine in the Treatment of Sporadic
Nontoxic Goiter
MARIA F. T. WESCHE, MONIQUE M. C. TIEL-V BUUL, PAUL LIPS,NICO J. SMITS, AND WILMAR M. WIERSINGA
Departments of Endocrinology and Metabolism (M.F.T.W., W.M.W.), Nuclear Medicine (M.M.C.T.-v.-B.),and Radiodiagnostics (N.J.S.), Academic Medical Center, University of Amsterdam,1105 AZ Amsterdam; and Department of Endocrinology (P.L.), AcademicHospital of the Free University, 1007 MB Amsterdam, The Netherlands
A and 16 of 28 patients in group B (
P ⫽ 0.00001). In responders, goiter
A randomized clinical trial was performed in consecutive patients
reduction in group A (46%) was greater than in group B (22%;
P ⬍
with sporadic nontoxic nodular goiter to compare efficacy and side
0.005). In group A, 45% of patients developed hypothyroidism. In
effects of iodine-131 (131I) therapy with suppressive levothyroxine
group B, 10 patients experienced thyrotoxic symptoms, requiring
(L-thyroxine) treatment. Sixty-four patients were randomized after
discontinuation of treatment in 2 (in 1 because of atrial fibrillation).
stratification for sex and menopausal age to receive 131I (4.44 MBq/g
Markers of bone formation and bone resorption increased signifi-
thyroid; group A) or suppressive L-thyroxine treatment aiming at TSH
cantly in group B, related to a mean decrease of 3.6% of BMD at the
values between 0.01 and 0.1 mU/L (group B). The main outcome
lumbar spine after 2 yr (from 1.09 ⫾ 0.22 to 1.05 ⫾ 0.23 g/cm2;
P ⬍
measurements after 2 yr were goiter size by ultrasound, serum thy-
0.001), both in pre- and postmenopausal women. No changes in BMD
roid function tests, markers of bone turnover, and bone mineral den-
were observed in group A. In conclusion, 131I therapy is more effective
sity (BMD). Fifty-seven patients completed the trial. Goiter size was
and better tolerated than L-thyroxine treatment in patients with
reduced after 2 yr by 44% in group A and by 1% in group B (
P ⬍ 0.001).
sporadic nontoxic goiter. Suppressive L-thyroxine treatment results
Nonresponders (goiter reduction ⬍13%) were 1 of 29 patients in group
in significant bone loss. (
J Clin Endocrinol Metab 86: 998 –1005, 2001)
nign enlargement of the thyroid gland of unknown cause,
the last few years, have attracted much attention (16–23).
in euthyroid subjects living in an area without endemic goiter
Because 131I treatment has never been compared directly
(1). The natural history of the disease is characterized by a
with l-thyroxine treatment, we performed a randomized
gradual increase of goiter size, under simultaneous develop-
clinical trial comparing the efficacy, tolerability, and safety of
ment of increasing thyroid nodularity and thyroid autonomy
l-thyroxine with 131I therapy in the treatment of sporadic
(2). Thus, large multinodular goiters may arise, often with ob-
nontoxic nodular goiter. The very study design—via the
structive signs and symptoms. Hyperthyroidism is observed in
instantaneous induction of a suppressed TSH in the patients
9–10% of the patients after a follow-up of 12 yr (3, 4). Treatment
randomized to receive l-thyroxine—allowed us to evaluate
options are surgery, levothyroxine (l-thyroxine), and radioac-
prospectively the effect of thyroid hormone-suppressive
tive iodine. Thyroidectomy is very effective, at the expense of
therapy on bone density in a controlled manner. The main
a low but unavoidable morbidity. Postoperative recurrence of
outcome measurements, as assessed after 2 yr, were goiter
the goiter occurs in 5–19% (5–8) and apparently cannot be
size, bone mineral density (BMD), and thyroid function.
prevented by the administration of T4 (5, 9–11). l-thyroxinetreatment in TSH-suppressive doses is not well studied in io-
Patients and Methods
dine-sufficient areas: in the placebo-controlled randomized
clinical trial of Berghout
et al. (12), a goiter reduction of only 25%was demonstrated in 59% of patients with SNG, irrespective of
One hundred consecutive patients with SNG, referred because of
baseline TSH value. Moreover, a suppressed TSH constitutes a
goiter, were included. The diagnosis of SNG was ascertained by ultra-sound (nodular goiter), 99mTc-pertechnetate thyroid scintigraphy (in-
risk for developing atrial fibrillation in patients 60 yr of age and
homogenous uptake), and by fine-needle aspiration cytology indicating
older (13) and for bone loss, especially in postmenopausal
the benign (dysplastic) nature of the goiter. None had been treated for
women (14, 15). Thus, it is understandable that the good results
goiter in the preceding 2 yr, and all patients lived in an iodine-sufficient
of iodine-131 (131I) therapy of SNG, as reported in several open
region (the mean 24-h urinary iodine excretion of healthy Dutch adultsis 147 g; Refs. 12 and 24). Exclusion criteria were severe obstructivesymptoms and signs (n ⫽ 2), cardiac disorders precluding l-thyroxine
Received April 17, 2000. Revision received August 15, 2000. Rerevi-
treatment (n ⫽ 3), pregnancy (-wish) or breastfeeding precluding radio-
sion received October 16, 2000. Accepted October 30, 2000.
iodine treatment (n ⫽ 3), and inability to complete follow-up (n ⫽ 2). Of
Address correspondence and requests for reprints to: Prof. Dr. W. M.
the 90 eligible patients, 64 gave informed consent to enter the trial (Fig.
Wiersinga, Department of Endocrinology and Metabolism F5.171, Ac-
1), which was approved by the local medical ethics committee. None of
ademic Medical Center, University of Amsterdam, Meibergdreef 9, 1105
them used medication affecting bone metabolism, except four premeno-
AZ Amsterdam, The Netherlands.
pausal women who continued to use oral contraceptives during the
and bone turnover were determined in fasting blood samples and 2-hfasting urine samples, collected between 0800 and 1000 h.
Thyroid volume was measured by ultrasonography using a contact
B-scanner (Searle Pho/Sonic-SM, Siemens AG, Munich, Germany) witha 5.0-MHz, 14-mm transducer (focal length, 4.5 cm). Transverse scans ofthe thyroid were obtained in supine position at 5-mm intervals fromcaudal to cranial with hyperextension of the neck. The sum of all partialvolumes equals the total thyroid volume. All determinations were per-formed by one radiologist (N.J.S.), who was blinded to the given treat-ment. The accuracy and precision of the method has been reportedearlier (24). A significant decrease in thyroid volume was defined as adecrease greater than 13% (
i.e. the mean ⫹ 2 sd of the coefficient ofvariation).
BMD of the lumbar spine, femoral neck, and trochanter was measured
by dual-energy x-ray absorptiometry, using a Norland XR26 (NorlandCorp., Fort Atkinson, WI; coefficients of variation: 2.4% for the lumbarspine, 2.3% for the femoral neck, and 2.4% in the trochanteric region, asmeasured in 51 volunteers). Z-scores were calculated for comparisonwith a reference population. During the study, the Norland densitom-eter was replaced by a Hologic 2000 (Hologic, Inc., Waltham, MA); BMDwas, thus, measured on two different densitometers in 20 patients. In 14of them we measured BMD on both densitometers on the same day at1-yr follow-up and found a correlation coefficient of 0.99 at the lumbarspine. We calculated Norland values from Hologic values with theformula y (Norland value) ⫽ ⫺0.04 ⫹ 1.06x (Hologic value), obtainedby regression analysis. The same procedure was applied to the femoralneck (r ⫽ 0.90, y ⫽ 0.07 ⫹ 0.99x) and trochanter (r ⫽ 0.85, y ⫽ 0.10 ⫹0.88x).
Plasma T4 and T3 were measured by in-house RIAs, FT4 by fluoro-
immunoassay using the Delfia technique (Ultra; Wallac Oy, Turku,Finland), and TSH by an immunochemiluminometric assay (Behring,Amsterdam, The Netherlands; functional sensitivity, 0.01 mU/L). Thereference values were: T4, 60 –160 nmol/L; T3, 1.3–2.7 nmol/L; FT4,
10 –22 pmol/L; and TSH, 0.4 – 4.0 mU/L. Autoantibodies against thyroid
FIG. 1. Flow diagram of patients with SNG selected for the trial.
peroxidase (TPO) and thyroglobulin were measured by chemilumines-cence immunoassays (LUMI-test; Brahms, Berlin, Germany). Serum
whole duration of the study. According to a sample size calculation, at
TSH-binding inhibiting immunoglobulins were measured by TRAK as-
least 44 patients were required to have an 80% chance of detecting a
say (Brahms) in patients who developed a suppressed TSH after 131I
difference of 50% (
P ⬍ 0.05) in reduction of goiter size between both
treatment. Serum osteocalcin was measured by RIA (INCSTAR Corp.,
Stillwater, MN), bone alkaline phosphatase (BAP) by Alkphase-B (Metra
Patients were randomized to 131I treatment (group A) or l-thyroxine
Biosystems, Mountain View, CA), insulin-like growth factor I (IGF-I) by
treatment (group B), after stratification for sex and menopausal status.
immunoradiometric assay (Diagnostic Systems Laboratories, Webster,
Premenopause was defined as having regular periods, perimenopause
TX), sex hormone-binding globulin (SHBG) also by immunoradiometric
as having irregular periods with at least one period in the last year, and
assay (Farmos Diagnostica, Turku, Finland), and 25-hydroxyvitamin D
postmenopause as no periods for at least 1 yr. Stratified randomization
by competitive protein binding assay (TNO, Zeist, The Netherlands).
was done independently of treating physicians, by preparing envelopesfor each stratum with a block size of four.
In patients randomized to 131I, pretreatment thyroidal radioiodine
uptake was measured with a tracer activity of 3.7 MBq Na 131I. The
Differences in (baseline) values between groups were analyzed by
therapeutic 131I dose (aiming at 4.44 MBq/mL thyroid tissue) was cal-
Student's
t test, Mann-Whitney
U test, or 2 test (to compare percentages
culated by the following formula: 131I dose (MBq) ⫽ [4.44 (MBq) ⫻
between groups), where appropriate. Changes in (outcome-) variables
100/24 h uptake (%)] ⫻ TV (thyroid volume; mL, measured by ultra-
were analyzed by ANOVA using repeated measurements (and applying
sonography). Patients were hospitalized for 131I treatment for 2– 6 days.
log transformation, where appropriate). To compare the series of
After 131I therapy, l-thyroxine was given if TSH increased above 4.0
changes between the two treatment groups, multivariate ANOVA was
mU/L, aiming at TSH values in the normal range.
performed. Correlation tests were performed by single linear regression
In patients randomized to l-thyroxine, the initial dose of l-thyroxine
analysis, using a PC software program (SPSS, Inc., Chicago, IL). For
was 2.5 g/kg body weight, aiming at TSH values between 0.01 and 0.1
calculations, undetectable serum concentrations were considered as cor-
mU/L. If pretreatment TSH was already suppressed, free T
responding to one half the functional sensitivity (
i.e. 0.005 mU/L for
were aimed at 20 –22 pmol/L. Dose adjustments of l-thyroxine (25 g
TSH). The level of significance was taken as ␣ ⫽ 0.05.
at a time) were performed on the basis of TSH values, or of clinical signsand symptoms of thyrotoxicosis. l-thyroxine tablets were taken late in
the evening.
Randomization and treatment
Patients were evaluated at 0, 1.5, 3, 6, 9, 12, 18, and 24 months; the
main outcome measurements were obtained before treatment and after
Initial clinical and laboratory data of the eligible but not
randomized patients and the two randomization groups are
given in Table 1. No differences were found between thethree groups. Patients were accrued in the period 1993–1996.
At each visit at the outpatient clinic, complaints related to the goiter
We included one patient below the age of 30 yr, a woman of
were noted, together with signs and symptoms of thyrotoxicosis or
29 yr old, who was operated on before (subtotal thyroidec-
hypothyroidism. The daily calcium intake was determined by a dieti-cian. Serum thyroid function tests were obtained at every visit. At
tomy) and had complaints of a goiter of 139 mL at study
baseline and at 1 and 2 yr of follow-up, markers of tissue thyrotoxicosis
entry. Fourteen patients older than 60 yr were included, of
WESCHE
ET AL.
Vol. 86 • No. 3
TABLE 1. Initial clinical characteristics and laboratory data of 90 patients with SNG
Randomization groups
Eligible, not randomized
L-thyroxine therapy
Complaints of goiter
Recent goiter growth
Duration of goiter (yr)
Thyroid grade I/II/III
Thyroid volume (mL)
Uni/multinodular goiter
0.8 (⬍0.01–2.2)
0.6 (⬍0.01–2.2)
Values as mean (SD) or as median (range).
whom six were randomized to receive l-thyroxine and eight
median increase in thyroid volume of 16% after 2 yr (Fig. 2).
to receive 131I; slightly low baseline TSH values (⬎0.1– 0.39
Intention-to-treat analysis resulted in comparable results: in
mU/L) were observed in three and two of these subjects,
group A, a median decrease in goiter size of 41% at 2 yr was
respectively; none had a TSH value of 0.1 mU/L or less.
found, whereas in group B goiter size decreased with 5% at
The goiter caused complaints in 81% of the 64 randomized
2 yr of treatment (
P ⬍ 0.0001). Compliance with l-thyroxine
patients: discomfort in the neck in 69%, cosmetic complaints
treatment was good as judged by laboratory evaluation (Fig.
in 31%, fear of malignancy in 27%, and dyspnea in 13%. In
3). Taking together all time points, TSH values were below
five patients the goiter extended to the retrosternal compart-
0.01 mU/L in 38%, between 0.01 and 0.1 mU/L in 46%, and
ment, which part could not be measured by ultrasonography.
between 0.1 and 1.0 mU/L in 16%. No significant differences
During follow-up, three patients of group A were excluded:
in TSH values were observed between responders and non-
two because, by mistake, the 131I dose was calculated on the
responders to l-thyroxine treatment.
scintigraphic instead of the ultrasonographic thyroid vol-
The decrease in goiter size in responders of group B was
ume, resulting in a much lower dose of 131I; and one because
smaller than the median decrease in thyroid volume of re-
hospital admission could not be realized due to severe illness
sponders in group A (
P ⬍ 0.005). Pretreatment goiter size was
of the patient's partner. In group B, four patients discontin-
inversely related to goiter reduction in group A (r ⫽ ⫺0.44,
ued l-thyroxine treatment prematurely: one patient because
P ⬍ 0.05) but not in group B (r ⫽ 0.01, not significant). No
of thyrotoxic symptoms not responding to a dose reduction,one because of pregnancy, one patient (55 yr old) because of
correlation was found between age or duration of goiter and
atrial fibrillation (who already had a suppressed baseline
the response to treatment in the two groups. Goiter reduction
TSH), and one because of noncompliance. Thus, data of 29
was directly related to baseline TSH in group B (r ⫽ 0.40,
P ⬍
patients in group A and 28 patients in group B were available
0.05) but not in group A. Before treatment, 17 patients had
for evaluation of treatment efficacy. In group A, the median
subclinical hyperthyroidism. Ten were treated with 131I (nine
pretreatment 131I uptake was 26% (range, 15–58) and a me-
responders and one nonresponder), and TSH normalized in
dian 131I dose of 888 MBq (range, 444 –3330) was adminis-
all; none became hypothyroid. Seven patients were treated
tered. In group B, the mean starting dose of l-thyroxine was
with l-thyroxine, without developing symptoms of thyro-
192 ⫾ 34 g/day, which at 1 yr had been reduced to 146 ⫾
toxicosis, of whom only one responded. Goiter reduction
44 g/day (
P ⬍ 0.001), corresponding to 1.89 ⫾ 0.47
after 2 yr of treatment with l-thyroxine was greater in pa-
tients with baseline TSH 0.4 mU/L or greater (medianchange, ⫺12%) than in patients with baseline TSH less than
0.4 mU/L (median change, ⫹28%;
P ⬍ 0.05).
In group A, a median decrease in goiter size of 38% at 1
In both treatment groups at 2 yr of follow-up, fewer pa-
yr and of 44% at 2 yr was found (Table 2). Twenty-eight
tients had complaints of their goiter than at baseline; after 2
patients (97%) were responders to treatment (defined as a
yr of treatment only 2 of 20 patients in group A and 9 of 19
decrease in thyroid volume ⬎13%) with a median decrease
patients in group B still had complaints of discomfort in the
in thyroid volume of 39% after 1 yr and 46% after 2 yr (Fig.
neck, 1 of 8 patients in group A and 5 of 10 patients in group
2). In group B, goiter size decreased with 7% at 1 yr and 1%
B still had cosmetic complaints, whereas dyspnea disap-
at 2 yr. Twelve patients (43%) were responders to treatment
peared in 4 of 4 patients of group A but persisted in 2 of 3
with a median decrease in thyroid volume of 23% after 1 yr
patients of group B. The remaining complaints after 2 yr of
and 22% after 2 yr. There were 16 nonresponders with a
treatment were significantly less frequent in group A than in
median decrease in thyroid volume of 1% after 1 yr and a
group B (3 of 32
vs. 16 of 32, respectively;
P ⬍ 0.05).
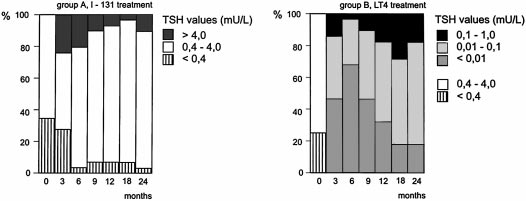
FIG. 2. Relative changes in thyroid volume after 131I treatment
(group A) or during L-thyroxine treatment (group B).
IG. 3. TSH values at baseline and during 24 months follow-up after
treatment with 131I or during L-thyroxine treatment.
Side effects
Clinical and biochemical data. One week after 131I treatment,four patients noted anterior neck tenderness and slight
symptoms of thyrotoxicosis lasting 1–3 weeks, compatible
with mild radiation thyroiditis. No obstructive symptoms
were noted. No patient developed Graves'-like hyperthy-
roidism. Of the eight patients who developed TSH values
below 0.4 mU/L 6 weeks to 9 months after 131I treatment, de
WESCHE ET AL.
Vol. 86 • No. 3
novo serum TSH-binding inhibiting immunoglobulins ap-peared in one without symptoms (TRAK before treatment,
⬍5 U/L; after 9 months, 29 U/L; after 12 months, 20 U/L).
After 2 yr, 16 patients were euthyroid (55%), 10 patients haddeveloped hypothyroidism (35%), and 3 patients developedsubclinical hypothyroidism (10%) in the first year after 131I
treatment. Patients with a normal pretreatment TSH had a
significantly higher risk of developing hypothyroidism after
131I treatment than patients with a suppressed baseline TSH(2, P ⬍ 0.005). The presence of TPO antibodies at baseline
also carried a higher risk of developing hypothyroidism after
131I (2, P ⬍ 0.05).
In group B, 10 patients experienced symptoms of mild
thyrotoxicosis, disappearing after dose adjustment in all but
1 patient. A small but significant increase in pulse rate of 9%was observed during treatment, without changes in body
weight. Serum SHBG and IGF-I concentrations did not in-
dicate tissue thyrotoxicosis in either group because no sig-
nificant changes were found during follow-up [group A,
SHBG as mean (sd): at baseline, 58 (24) nmol/L; at 2 yr, 67
(29) nmol/L; group A, IGF-I: at baseline, 28 (12) nmol/L; at
2 yr, 27 (11) nmol/L; group B, SHBG: at baseline, 64 (45); at
2 yr, 78 (69) nmol/L; group B, IGF-I: at baseline, 22 (8)
nmol/L; at 2 yr, 23 (9) nmol/L]. Serum lipids (total choles-
terol, low-density lipoprotein cholesterol, and triglycerides)
did not change either in both treatment groups.
Markers of bone turnover
Markers of bone formation and resorption are listed in
Table 3. Males (n ⫽ 2) and perimenopausal women (n ⫽ 4)
were excluded from evaluation as well as two other patients,
one because of hypoparathyroidism due to previous thy-
roidectomy and one because of the appearance of bone me-
tastases of breast cancer. Baseline values in group A (n ⫽ 24;
13 pre- and 11 postmenopausal women) and group B (n ⫽
25; 15 pre- and 10 postmenopausal women) did not differ.
Daily calcium intake (group A, 1110 mg; range, 418 –2440 vs.
group B, 886 mg; range, 480 –1905) and serum 25-hydroxy-
vitamin D concentrations [group A, 55 (22) nmol/L, mean
(sd), vs. group B, 53 (30) nmol/L] were similar in both
groups. Markers of bone turnover did not change in group
A, except an increase in total and BAP, although less marked
than in group B (P ⬍ 0.0005). Serum osteocalcin, total and
BAP, and urinary hydroxyproline increased in group B. After
2 yr of treatment with l-thyroxine, TSH values were in-
versely correlated with alkaline phosphatase (r ⫽ ⫺0.70, P ⬍
0.001), BAP (r ⫽ ⫺0.72, P ⬍ 0.001), osteocalcin (r ⫽ ⫺0.36, P ⫽
0.08), and the hydroxyproline to creatinine (Hp/Cr) ratio (r ⫽
0.62, P ⬍ 0.001). At 2 yr, a positive correlation was noted
between osteocalcin and the Hp/Cr ratio (r ⫽ 0.70, P ⬍ 0.001)
in group B, but not in group A.
BMD changes
Measurements of BMD are given in Table 3. No differences
between groups A and B were noted in smoking history,
alcohol and coffee intake, physical activity, body weight,
menarche, time and duration of menopause, and use of oral
contraceptives. Pretreatment values of patients of groups A
and B were not different. Z-scores in groups A and B were
than 0.1 mU/L is associated with a 3-fold higher risk for de-veloping atrial fibrillation in the next decade (13). We concludethat T4 treatment is apparently contraindicated if TSH is below0.1 mU/L, irrespective of age.
The administered dose of 131I is obviously a determinant of
goiter reduction by radioiodine treatment (23). This could notbe evaluated in the present study because all patients receivedthe same dose of 4.44 MBq/g thyroid. We calculated the 131Idose on goiter size measured by ultrasonography. Scintigraphicmeasurement of the size of nodular goiters may differ consid-erably from ultrasonographic measurements (25–27). The pre-cise method of assessing nodular goiter size should, thus, betaken into account when comparing literature data on the out-
FIG. 4. Relative changes of BMD (given as mean and SEM) at the
come of 131I therapy. In our series, the outcome of radioiodine
lumbar spine, femoral neck, and trochanter 1 and 2 yr after treatment
treatment was inversely related to goiter size: the larger the
with 131I (group A) or L-thyroxine (group B).
goiter, the smaller the relative decrease in size. To enhance theefficacy of 131I therapy and to limit the theoretical risk of cancer
not different either: lumbar spine as mean (sd), 0.22 (1.02) vs.
induction through the radiation burden of large doses of 131I,
0.19 (1.18); femoral neck, 0.34 (1.10) vs. 0.32 (0.94); and tro-
one could argue to administer radioiodine at an earlier stage
chanter, 0.23 (0.90) vs. 0.26 (0.94). Postmenopausal women
when the goiter is still smaller, allowing a lower 131I dose. The
had significantly lower BMD values than premenopausal
gain, however, must be weighted against an increased risk on
women. In group A, BMD did not change during treatment.
postradioiodine hypothyroidism, because patients with smaller
In group B, BMD at the lumbar spine was reduced by 3.6%
goiters are less likely to have suppressed TSH values, which
after 2 yr (P ⬍ 0.001; Fig. 4); the decrease was similar in pre-
in the present study protected against the development of
and postmenopausal women [from 1.19 (0.19) to 1.16 (0.19)
g/cm2 (P ⫽ 0.002; mean decrease, 2.6%) and from 0.93 (0.17)to 0.89 (0.18) g/cm2 (P ⫽ 0.003; mean decrease, 5.0%), re-spectively]. BMD of femoral neck and trochanter also de-
Side effects
creased in group B, although not significantly (Fig. 4). Pa-
The price to be paid for the good efficacy of 131I therapy is the
tients in group B with a baseline TSH below 0.4 mU/L had
rather high incidence of hypothyroidism. Our figure of 45% is
a lower baseline BMD at the lumbar spine (P ⬍ 0.05) and a
relatively high compared with some, but not all, previous stud-
larger decrease in BMD at the lumbar spine after 2 yr of
ies (17, 19, 20, 23); the differences may be explained by the fact
treatment [from 0.94 (0.19) to 0.88 (0.21) g/cm2, P ⫽ 0.002]
that in our study 131I-treated patients were given l-thyroxine as
than patients with a normal baseline TSH value [from 1.15
soon as TSH rose above 4.0 mU/L, aiming at TSH levels in the
(0.21) to 1.12 (0.20) g/cm2, P ⫽ 0.001; mean change, ⫺7.2%
normal range to prevent regrowth of the goiter. Knowing that
vs. ⫺2.2%, P ⬍ 0.05]. An inverse relationship was found
hypothyroidism may be transient after 131I treatment for hy-
between the changes in BMD at the lumbar spine and the
perthyroidism (28), we, thus, could have overestimated the
changes in BAP (r ⫽ ⫺0.42, P ⬍ 0.05) or the Hp/Cr ratio (r ⫽
percentage of patients with permanent hypothyroidism in our
0.48, P ⬍ 0.05) in group B, but not in group A.
study. Determinants of postradioiodine hypothyroidism in ourseries were baseline TSH and the presence of TPO antibodies,
as reported before (29). Other side effects of 131I therapy were
transient in nature and limited in number.
The present study demonstrates that radioiodine treatment
Among the patients of group B receiving T4, 10 developed
is far more effective in reducing the size of SNGs than sup-
thyrotoxic symptoms, necessitating premature discontinua-
pressive doses of l-thyroxine: in the 131I-treated patients, there
tion of T4 in 2 (in 1 because of atrial fibrillation). The ad-
were 97% responders with a decrease in goiter size of 46%
ministration of TSH-suppressive doses of T4 did not change
compared with 43% responders in the l-thyroxine-treated pa-
serum concentrations of SHBG, lipids, and IGF-I. These
tients in whom goiter size decreased by 22%. The observed
markers for the effect of thyroid hormones on peripheral
effect size of both treatment modalities is in good agreement
tissues, thus, did not indicate tissue thyrotoxicosis in the
with earlier studies: a reduction in goiter size of 40–60% has
liver. Our findings are in disagreement with two previous
been reported in 80–100% of the patients treated with 131I in
cross-sectional studies reporting lower total and (low-
open studies (19–23) and of 25% in 59% of the patients treated
density lipoprotein) cholesterol values in spontaneous sub-
with l-thyroxine in a placebo-controlled trial (12).
clinical hyperthyroidism (30) and in l-thyroxine-treated pa-
Chances for goiter reduction on T4 treatment were less if
tients with suppressed TSH compared with controls (31). A
pretreatment TSH was already suppressed. Moreover, patients
longer exposure time in these studies than the 2 yr in the
with a suppressed baseline TSH value had a larger decrease in
present study might explain the discrepancy.
BMD at the lumbar spine after 2 yr of treatment, and one patient
In bones, however, the consequences of induced subclinical
of 55 yr developed atrial fibrillation after 15 months of l-thy-
hyperthyroidism maintained for 2 yr were striking. Stratifica-
roxine treatment. Our observations extend on an earlier finding
tion for sex and menopausal age at randomization allowed us
that among people 60 yr of age and older, a serum TSH of less
to compare BMD and bone turnover in two homogeneous
WESCHE ET AL.
Vol. 86 • No. 3
groups composed of pre- and postmenopausal women, who
though hypothyroidism develops in approximately half of
had received radioiodine or T4 treatment. BMD and markers of
the patients, it is easily treated with T4, not requiring TSH-
bone turnover did not change in the 131I-treated group, except
suppressive doses. The efficacy of 131I can be enhanced by
for an increase of total and BAP, which remains unexplained.
treatment at an earlier stage when the goiter is still smaller;
In the T4-treated group, the increase of BAP was larger than
this will also add to the long-term safety of radioactive io-
after 131I (P ⬍ 0.005) and was accompanied by an increase in
dine, by allowing the application of a lower dose of 131I.
serum osteocalcin and urinary hydroxyproline, indicating a riseof both bone formation and bone resorption, which were related
to the fall of TSH. The inverse relationship between serum TSHand bone turnover has been noted before (32). The increased
1. Hennemann G. 1979 Non-toxic goitre. Clin Endocrinol Metab. 8:167–179.
bone turnover was associated with a decrease of BMD at the
2. Berghout A, Wiersinga WM, Smits NJ, Touber JL. 1990 Interrelationships
between age, thyroid volume, thyroid nodularity, and thyroid function in
lumbar spine, which was present both in pre- and postmeno-
patients with sporadic nontoxic goiter. Am J Med. 89:602– 608.
pausal women (2.6% and 5.0%, respectively); a decrease of
3. Wiener JD, de Vries AA. 1979 On the natural history of Plummer's disease.
Clin Nucl Med. 4:181–190.
BMD at the femoral neck and trochanter was also observed but
4. Elte JW, Bussemaker JK, Haak A. 1990 The natural history of euthyroid
failed to reach significance, probably due to a small sample size.
multinodular goitre. Postgrad Med J. 66:186 –190.
Two meta-analyses of published studies (all cross-sectional) on
5. Hegedu¨s L, Hansen JM, Hansen BM, Hjalgrim H, Gervil M. 1991 Is pro-
phylactic thyroxine therapy after operation for non-toxic goiter justified? Thy-
suppressive thyroid hormone therapy showed significant bone
roid. 1(Suppl 1):2.
loss in postmenopausal, but not in premenopausal, women (14,
6. Berghout A, Wiersinga WM, Drexhage HA, et al. 1989 The long-term outcome
15). The authors recommended a large, double-blind, placebo-
of thyroidectomy for sporadic non-toxic goitre. Clin Endocrinol. 31:193–199.
7. Geerdsen JP, Hee P. 1982 Nontoxic goitre. I. Surgical complications and
controlled trial (in patients with benign nodules, receiving sup-
long-term prognosis. Acta Chir Scand. 148:221–224.
8. Vagn Nielsen O, Iverson O, Bagge E. 1963 Surgical treatment of non-toxic
4 treatment for at least 2 yr), to get definitive answers.
goitre. II. Follow-up on patients operated upon during the period 1945–1950.
Our trial seems to meet this ideal study design to a large extent
Dan Med Bull. 10:68 –70.
and demonstrates that prolonged subclinical hyperthyroidism
9. Hegedus L, Hansen JM, Veiergang D, Karstrup S. 1987 Does prophylactic
has an adverse effect on bone mass not only in post- but also
thyroxine treatment after operation for non-toxic goitre influence thyroid size?Br Med J. 294:801– 803.
in premenopausal women. Two other recent prospective, al-
10. Bistrup C, Nielsen JD, Gregersen G, Franch P. 1994 Preventive effect of
though nonrandomized, trials are in agreement with our find-
levothyroxine in patients operated for non-toxic goitre: a randomized trial of
ings. A longitudinal study in premenopausal women indicated
one hundred patients with nine years follow-up. Clin Endocrinol. 40:323–327.
11. Berglund J, Aspelin P, Bondeson A, et al. 1998 Rapid increase in volume of
spinal bone loss of 0.2 ⫾ 1.2% per year in controls, significantly
the remnant after hemithyroidectomy does not correlate with serum concen-
less than 2.6 ⫾ 1.9% in patients on suppressive T
tration of thyroid stimulating hormone. Eur J Surg. 164:257–262.
12. Berghout A, Wiersinga WM, Drexhage HA, Smits NJ, Touber JL. 1990 Com-
(sub)total thyroidectomy for goiter or cancer (33). Another
parison of placebo with l-thyroxine alone or with carbimazole for treatment
study in nontoxic goiter patients on suppressive therapy with
of sporadic non-toxic goitre. Lancet. 336:193–197.
13. Sawin CT, Geller A, Wolf PA, et al. 1994 Low serum thyrotropin concentra-
4 also suggests reduction of BMD relative to age-matched
tions as a risk factor for atrial fibrillation in older persons. N Engl J Med.
controls, in both pre- and postmenopausal women (34). One
331:1249 –1252.
may question the appropriateness of the radioiodine-treated
14. Faber J, Galloe AM. 1994 Changes in bone mass during prolonged subclinical
patients as a control group in our study. One could argue that
hyperthyroidism due to l-thyroxine treatment: a meta-analysis. Eur J Endo-crinol. 130:350 –356.
the preserved BMD in the radioiodine group is explained by
15. Uzzan B, Campos J, Cucherat M, Nony P, Boissel JP, Perret GY. 1996 Effects
correction of slight thyroid hormone excess (35) and that the
on bone mass of long term treatment with thyroid hormones: a meta-analysis.
J Clin Endocrinol Metab. 81:4278 – 4289.
observed bone loss in the T4-treated patients is due to natural
16. Keiderling W, Emrich D, Hauswaldt C, Hoffmann G. 1964 Ergebnisse der
history. However, a decrease in BMD at the lumbar spine of
radio-jodverkleinerungstherapie euthyreoter strumen. Dtsch Med Wochen-
2.6% in premenopausal women treated with l-thyroxine is far
schr. 89:453– 457.
17. Kay TW, d'Emden MC, Andrews JT, Martin FI. 1988 Treatment of non-toxic
more than expected by aging with 2 yr, and the absence of any
multinodular goiter with radioactive iodine. Am J Med. 84:19 –22.
change in BMD in our radioiodine-treated patients was found
18. Verelst J, Bonnyns M, Glinoer D. 1990 Radioiodine therapy in voluminous
irrespective of baseline TSH levels, both in pre- and postmeno-
multinodular non-toxic goitre. Acta Endocrinol. 122:417– 421.
19. Hegedus L, Hansen BM, Knudsen N, Hansen JM. 1988 Reduction of size of
pausal women.
thyroid with radioactive iodine in multinodular non-toxic goitre. Br Med J.
297:661– 662.
20. Nygaard B, Hegedus L, Gervil M, Hjalgrim H, Soe-Jensen P, Hansen JM. 1993
Radioiodine treatment of multinodular non-toxic goitre. Br Med J. 307:828–832.
21. Huysmans DA, Hermus AR, Corstens FH, Barentsz JO, Kloppenborg PW.
The side effects of suppressive T
1994 Large, compressive goiters treated with radioiodine. Ann Intern Med.
4 therapy are not negli-
gible, because suppressed TSH levels (defined as ⬍0.1
22. Wesche MF, Tiel-v Buul MM, Smits NJ, Wiersinga WM. 1995 Reduction in
mU/L) increase the risk for atrial fibrillation and the ob-
goiter size by 131I therapy in patients with non-toxic multinodular goiter. Eur
served bone loss constitutes a risk factor for fractures. Be-
J Endocrinol. 132:86 – 87.
23. de Klerk JM, van Isselt JW, van Dijk A, et al. 1997 Iodine-131 therapy in
cause reduction of goiter size during T4 treatment is lost on
sporadic nontoxic goiter. J Nucl Med. 38:372–376.
discontinuation of the drug (12), continuous treatment is
24. Berghout A, Wiersinga WM, Smits NJ, Touber JL. 1987 Determinants of
thyroid volume as measured by ultrasonography in healthy adults in a non-
necessary; long-term exposure to suppressed TSH values
iodine deficient area. Clin Endocrinol. 26:273–280.
raises concern as to the safety of this treatment modality for
25. Igl W, Lukas P, Leisner B, et al. 1981 Sonographic determination of thyroid
nontoxic goiter. Taken together with the modest efficacy of
volume. Comparison with other methods. Nuklearmedizin. 20:64 –71.
26. Tannahill AJ, Hooper MJ, England M, Ferriss JB, Wilson GM. 1978 Mea-
T4, we do not recommend it any longer for the treatment of
surement of thyroid size by ultrasound, palpation and scintiscan. Clin Endo-
nontoxic nodular goiter, also because an attractive nonsur-
crinol. 8:483– 486.
gical alternative is available: radioactive iodine. 131I therapy
27. Wesche MFT, Tiel-v Buul MM, Smits NJ, Wiersinga WM. 1998 Ultrasono-
graphic versus scintigraphic measurement of thyroid volume in patients re-
is simple, devoid of major side effects, and effective. Al-
ferred for 131I therapy. Nucl Med Commun. 19:341–346.
28. Yoshida K, Aizawa Y, Kaise N, Fukazawa H, Kiso Y, Sayama N. 1998 Role
density and bone turnover in postmenopausal women on thyroxine. Clin
of thyroid-stimulating blocking antibody in patients who developed hypo-
thyroidism within one year after 131I treatment for Graves' disease. Clin En-
33. Pioli G, Pedrazzoni M, Palummeri E, et al. 1992 Longitudinal study of bone
loss after thyroidectomy and suppressive thyroxine therapy in premenopausal
29. Nygaard B, Knudsen JH, Hegedus L, Scient AV, Hansen JE. 1997 Thyrotropin
women. Acta Endocrinol. 126:238 –242.
receptor antibodies and Graves' disease, a side effect of 131I treatment in
34. De Rosa G, Testa A, Giacomini D, Carrozza C, Astazi P, Caradonna P. 1997
patients with nontoxic goiter. J Clin Endocrinol Metab. 82:2926 –2930.
Prospective study of bone loss in pre- and post-menopausal women on
30. Parle JV, Franklyn JA, Cross KW, Jones SR, Sheppard MC. 1992 Circulating
l-thyroxine therapy for non-toxic goitre. Clin Endocrinol. 47:529 –535.
lipids and minor abnormalities of thyroid function. Clin Endocrinol. 37:411–414.
35. Faber J, Jensen IW, Petersen L, Nygaard B, Hegedus L, Siersbaek-Nielsen K.
31. Franklyn JA, Daykin J, Betteridge J, et al. 1993 Thyroxine replacement therapy
1998 Normalization of serum thyrotrophin by means of radioiodine treatment
and circulating lipid concentrations. Clin Endocrinol. 38:453– 459.
in subclinical hyperthyroidism: effect on bone loss in postmenopausal women.
32. Guo CY, Weetman AP, Eastell R. 1997 Longitudinal changes of bone mineral
Clin Endocrinol. 48:285–290.
Source: http://www.endo.org/club/wesche.pdf
Departamento de Estado de Estados Unidos / Oficina de Programas de Información Internacional La Oficina de Programas de Información Internacional del Departamento de Estado de Estados Unidos publica cinco periódicos electrónicos — Perspectivas Económicas, Cuestiones Mundiales, Temas de la Democracia, Agenda de la Política Exterior de Estados Unidos y Sociedad y Valores Estadounidenses— que analizan los principales temas que
Diagnosis,Therapy and Prophylaxis of Fungal Diseases Inflammatory Tinea capitis: a 12-year study and a review of theliterature Ine s Zaraa,1,2 Abdelmohti Hawilo,1 Amina Aounallah,1 Sondes Trojjet,1 Dalenda El Euch,1Mourad Mokni1,2 and Amel Ben Osman1,21Department of Dermatology, La Rabta Hospital Tunis, Tunisia and 2University of Medicine, El Manar Faculty Tunis, Tunis, Tunisia








